What is the Ht concentration for an aqueous solution with pOH = 4.27 at 25 ...
Fantastic news! We've Found the answer you've been seeking!
Question:
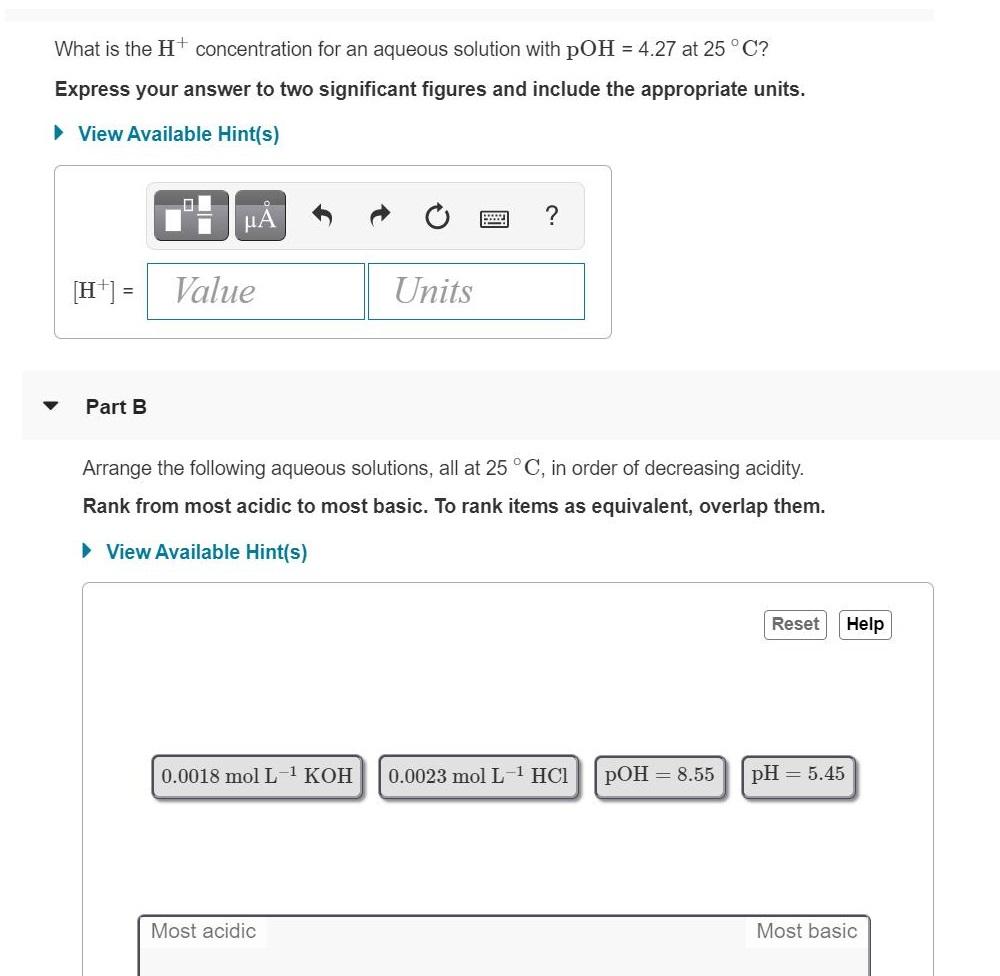
Transcribed Image Text:
What is the Ht concentration for an aqueous solution with pOH = 4.27 at 25 ° C? Express your answer to two significant figures and include the appropriate units. • View Available Hint(s) [H*] = Value Units Part B Arrange the following aqueous solutions, all at 25 °C, in order of decreasing acidity. Rank from most acidic to most basic. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Help 0.0018 mol L1 KOH 0.0023 mol L-1 HCl pOH = 8.55 pH = 5.45 Most acidic Most basic What is the Ht concentration for an aqueous solution with pOH = 4.27 at 25 ° C? Express your answer to two significant figures and include the appropriate units. • View Available Hint(s) [H*] = Value Units Part B Arrange the following aqueous solutions, all at 25 °C, in order of decreasing acidity. Rank from most acidic to most basic. To rank items as equivalent, overlap them. • View Available Hint(s) Reset Help 0.0018 mol L1 KOH 0.0023 mol L-1 HCl pOH = 8.55 pH = 5.45 Most acidic Most basic
Expert Answer:

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemistry questions
-
Rank the following compounds in order of decreasing acidity of the indicated hydrogen: O 0 CH CCH2CH2CCH3 CH CCH2CH2CH2CCH3 CH CCH2CCH3
-
Arrange the members of each group in order of decreasing basicity: (a) Ammonia, aniline, methylamine (b) Acetanilide, aniline, N-methylaniline (c) 2,4-Dichloroaniline, 2,4-dimethylaniline,...
-
Rank the following groups of compounds in order of decreasing solubility in water: a. CH3CH3CH3OH CH3CH3CH3CH3Cl CH3CH3CH3CH3OH HOCH3CH3CH3OH b. CH NH2 OH
-
Cassandra is retiring December 31, 2023, after many, happy years of employment with Canadian National Railway Company (CN). She is fortunate to be part of a Defined Benefit Pension Plan (DBPP). Her...
-
A sample of gas mixture from a neon sign contains 0.0856 mol Ne and 0.0254 mol Kr. What are the mass percentages of Ne and Kr in the gas mixture?
-
When metal is deformed between recrystallization temperature and melting point of the metal, it is called _________.
-
Number of classes: 6 Data set: Ages of the U.S. presidents at Inauguration Construct a frequency distribution and a frequency polygon for the data set using the indicated number of classes. Describe...
-
Would the name Hallowed receive protection as a trademark or as trade dress? Two computer science majors, Trent and Xavier, have an idea for a new video game, which they propose to call Hallowed....
-
Discuss the principles of wind energy resource assessment and optimization in onshore and offshore wind farm development, addressing topics such as wind turbine siting, wake effects, turbulence...
-
Cathy's Curls is the only hair stylist in a small town and is a profit-maximizing monopoly. It faces a demand curve for its haircuts which is given by P = 100 - 0.2 Q. The marginal cost of providing...
-
We'll watch the documentary "Making it Balance" in class. You may also view it at Making It Balance - DiscoverAudit.org Discover a Career in Public Company Auditing. After watching the film, write a...
-
During the recession of 20072009, the U.S. federal governments tax collections fell from about $2.6 trillion down to about $2.1 trillion while GDP declined by about 4 percent. Does the U.S. tax...
-
An economy is producing at full employment when AD unexpectedly shifts to the left. A new classical economist would assume that as the economy adjusts back to producing at full employment, the price...
-
A commercial bank has $100 million in checkable-deposit liabilities and $12 million in actual reserves. The required reserve ratio is 10 percent. How big are the banks excess reserves? a. $100...
-
City Bank is considering making a $50 million loan to a company named SheetOil that wants to commercialize a process for turning used blankets, pillowcases, and sheets into oil. This companys chances...
-
A DVCs population is growing 2 percent per year and its output is growing 3 percent per year. If the government wants to improve living standards over the coming decades, which of the following would...
-
How do global governance institutions, such as the World Trade Organization and International Monetary Fund, shape the contours of economic glo balization and influence policies regarding trade...
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
Vapor pressure data for chlorine are given below. a. Use these data and the ClausiusClapeyron equation (Equation 6.1-3) to estimate the heat of vaporization of chlorine (kJ/mol) and to obtain an...
-
A sedimentation process is to be used to separate pulverized coal from slate. A suspension of finely divided particles of galena (lead sulfide SG = 7.44) in water is prepared. The overall specific...
-
A storage tank containing oil (SG = 0.92) is 10.0 meters high and 16.0 meters in diameter. The tank is closed, but the amount of oil it contains can be determined from the gauge pressure at the...
-
What ideas do you have for a new business? How can you multiply the stimuli around these ideas to enhance them and identify attractive opportunities?
-
What do you really enjoy doing? What is your passion? Can your passion be a platform for a viable opportunity?
-
What do your friends and family envision you doing? What strengths and weaknesses do they observe? How do their insights help lead you to an opportunity that is right for you?

Study smarter with the SolutionInn App


