Identify P.Q.R and S in following reactions NH.HCI (i) (ii) Ar Ar- NH.HCI OC,H, NH (excess)...
Fantastic news! We've Found the answer you've been seeking!
Question:
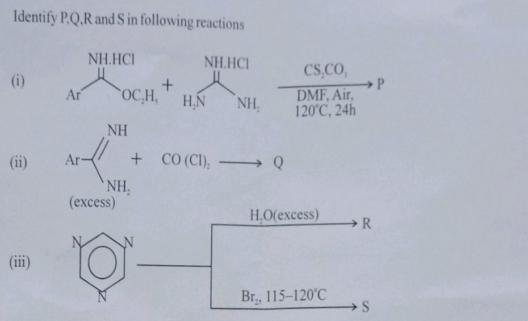
Transcribed Image Text:
Identify P.Q.R and S in following reactions NH.HCI (i) (ii) Ar Ar- NH.HCI OC,H, NH (excess) NH, H.N +CO (CI), Q - NH₂ CS,CO, DMF, Air, 120°C, 24h H₂O(excess) Br., 115-120°C R S P Identify P.Q.R and S in following reactions NH.HCI (i) (ii) Ar Ar- NH.HCI OC,H, NH (excess) NH, H.N +CO (CI), Q - NH₂ CS,CO, DMF, Air, 120°C, 24h H₂O(excess) Br., 115-120°C R S P
Expert Answer:
Answer rating: 100% (QA)
answer In the gave responses P Q R and S address different compound species included We should recog... View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these chemistry questions
-
Write the IUPAC name of the following molecule: *Lowercase letters only and DO NOT put space in between. Gray balls represent C, white balls represent H, green ball represent F. Do not include...
-
You are a bank, and your customer asks you to quote an agreed-upon rate for a 6x6 FRA. You observe the following rates: Maturity Rate 3-month 1% 6-month 2% 9-month 3% 12-month 4% What rate should you...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
client with celiac disease asks for a nutrition plan, what should the trainer recommend? Refer to a dietician 3-day food log to prescribe meal plan A templated meal plan Give a breakdown of...
-
Assume that in silver metal there is one free electron per silver atom. Compute the free electron density for silver, and compare it to the value given in Exercise 25.2.
-
Identify and describe the approach the FASB requires for reporting changes in accounting principles.
-
Selected accounts of Armstrong Consulting, Inc., a financial services business, have the following balances at December 31, 2010, the end of its first year of operations. During the year, Lori...
-
Refer to question 10. Suppose that the consultants fee is $5,000 and the utility function for the owner of Morley Properties can be approximated by the exponential utility function: U(x) = 1 e-x/R...
-
Question 1 Suppose a technician who assembles Comet computers earns $ 160 per hour. The technician works 48 hours during a week instead of scheduled time of 40 hours per week. The overtime pay scale...
-
What is meant by the term decision analysis?
-
Investment tax credits are widely practiced by a number of governments in African countries, as a way of attracting foreign direct investment. A number of civil society organizations argue that there...
-
Which of the following could be valid JDBC URL formats for an imaginary driver named magic and a database named box? A. Only first B. Only second C. Only third D. Both first and second E. Both first...
-
Which statement about the following declarations is correct? A. None of the declarations compile. B. Only the declaration of Friend contains a compiler error. C. Only the declaration of MyFriends...
-
Which properties of the Clean annotation must be true for the following to compile? (Choose three.) A. The annotation must contain exactly one element. B. The annotation must contain an element named...
-
Which is found in the java.sql package that come with the standard JDK? A. Only DerbyDriver B. Only MySqlDriver C. Only OracleDriver D. DerbyDriver, MySqlDriver, OracleDriver E. Only DerbyDriver and...
-
How many lines of the following declarations contain a compiler error? A. None B. One C. Two D. Three E. Four F. Five. import java.lang.annotation.*; enum Colors { RED, BLUE, GREEN } @Documented...
-
If the nominal risk-free rate is 8%, the expected rate of inflation is 2.5%, and the risk premium is 5%, calculate the required rate of return.
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
Explain the results of the following reactions by means of mechanisms. (a) (b) (c) Explain why hemiacetal formation may be catalyzed by either acid or base, but acetal formation is catalyzed only by...
-
If cyclobutane were flat, it would have exactly 90 C-C-C bond angles and could conceivably use pure p orbitals in its C-C bonds. What would be a possible hybridization for the carbon atoms of the...
-
The polypeptide bradykinin is a tissue hormone that can function as a potent pain-producing agent. By means of a single treatment with the Edman reagent, the N-terminal amino acid in bradykinin is...
-
A uniform circular disc is pivoted at point \(O\), as shown in Fig. 2.105. Find the natural frequency of the system. Also find the maximum frequency of the system by varying the value of \(b\). b...
-
Find the natural frequency of the traffic sign system described in Problem 2.79 in torsional vibration about the \(z\)-axis by considering the masses of both the post and the sign. The spring...
-
Solve Problem 2.99 by changing the material from steel to bronze for both the post and the sign. Material properties of bronze: \(ho\) (specific weight) \(=80.1 \mathrm{kN} / \mathrm{m}^{3}, E=111.0...

Study smarter with the SolutionInn App


