Identify the oxidation numbers of each element in following molecules. FeO42- Identify the oxidation numbers of...
Fantastic news! We've Found the answer you've been seeking!
Question:
![]()
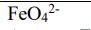
Transcribed Image Text:
Identify the oxidation numbers of each element in following molecules. FeO42- Identify the oxidation numbers of each element in following molecules. FeO42-
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The income statement, balance sheet, and statement of cash flows are the three main financial statements that every business uses and are essential for a manager to review on a monthly basis. If the...
-
The Lewises are a married couple with a combined income of $46,700. They have one child with a health condition, and can claim the following deductions: $6,000 in property tax $7,000 in mortgage...
-
A rock is thrown at a 60.0 angle above the horizontal across level ground. It is thrown from a height of 2.00 m above the ground with a speed of 20.0 m/s and experiences no appreciable air...
-
Determine the force in member GC of the truss and state if this member is in tension or compression. Units Used: kip = 103 lb Given: F1 = 1000 lb F2 = 1000 lb F3 = 1000 lb F4 = 1000 lb a = 10 ft =...
-
The loaves of rye bread distributed to local stores by a certain bakery have an average length of 30 centimeters and a standard deviation of 2 centimeters. Assuming that the lengths are normally...
-
A ray traveling through a large vat filled with medium 1 , index of refraction \(n_{1}=1.3\), is incident on a flat layer (thickness \(d=20 \mathrm{~mm}\) ) of medium 2, index of refraction...
-
Little, Inc., reported earnings of \($159,000\) for 2016, and at the end of the year, had the following securities outstanding: 1. 60,000 shares of common stock. (The year-end share price was \($25\)...
-
The data below present key ratios for six well-known U.S. corporations: Apple, Boeing, Citigroup, Facebook, McDonalds, and Walmart. However, the companies are not listed in order, and their names...
-
Madison Manufacturing is considering a new machine that costs $ 3 5 0 , 0 0 0 and would reduce pre - tax manufacturing costs by $ 1 1 0 , 0 0 0 annually. Madison would use the 3 - year MACRS method...
-
Low Crop Prices Drag Farm Towns Down with Them: Low wheat, corn, and soybean prices mean another bad year for many Midwest farmers. In response to the above newspaper headline, what government action...
-
In a topological space (X,t) a subset Ac X is called: pre open if Ac int(cl(A))) "ACA " ii) semi open if Ac c(int(A))) "ACA-". iii) a- open if Acint(cl(int(A))) "ACA iv) B-open if Accl(int(cl(A)))...
-
Consider the grammar below: G=({S,A,B),(a,b,c},S,P) S ScS aSS | aA | Bb AaAla BBb|A a) Write the leftmost derivation for the string aacabb b) Write the rightmost derivation for the string aacabb
-
Ellen came to the US as an EB five preference worker. She's making the final preparations for hiring 12 individuals to work 40 hours per week. However, she is currently reviewing all job...
-
Solve, the system of equations below, algebraically. (x-3)+(y+2)=16 2x+2y=10
-
Jan exchanges stock that she owns in Rocky Corporation with a value of $21,500 for stock in Lucy Corporation. The stock that she received from Lucy had a value of $20,000 when received. In addition,...
-
The data for an asset that is being considered: Salvage value at 5 years=$3,000 Rebuild cost at 3 years=$23,000 Annual net cash flow=$22,000 per year What is the ROR for this asset?
-
Compute for the magnitude of the combined moment (in N-m) of the three forces indicated in the figure with point B as the reference point. B 0.5 m 500 N 250 N 1 m 0.2 m 1 m 1 m 30 A 300 N
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
Water is added to 25.0mL of a 0.866 M KNO3 solution until the volume of the solution is exactly 500mL. What is the concentration of the nal solution?
-
What are the empirical formulas of the following compounds? (a) C2N2, (b) C6H6, (c) C9H20, (d) P4O10, (e) B2H6
-
Antimony pentafluoride, SbF5, reacts with XeF4 and XeF6 to form ionic compounds, XeF3+SbF6- and XeF5+ SbF6-. Describe the geometries of the cations and anion in these two compounds?
-
The chapter highlighted one way in which the Procter \& Gamble Company demonstrates its commitment to being a good corporate citizen. Go to Procter \& Gamble's Website and navigate to the section on...
-
Below are income statements and balance sheets for the Peyton Company for 2019 and 2018: Required Calculate the following ratios for the Peyton Company for 2019 and 2018 and discuss your findings: 1....
-
Which of the following information system functions is generally responsible for the design, coding, and debugging of computer programs? (a) technical support (b) programming (c) operations (d)...

Study smarter with the SolutionInn App



