Identify the oxidation numbers of each element in following molecules. Cu(NH3)42+ Identify the oxidation numbers of...
Fantastic news! We've Found the answer you've been seeking!
Question:
![]()
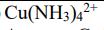
Transcribed Image Text:
Identify the oxidation numbers of each element in following molecules. Cu(NH3)42+ Identify the oxidation numbers of each element in following molecules. Cu(NH3)42+
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The income statement, balance sheet, and statement of cash flows are the three main financial statements that every business uses and are essential for a manager to review on a monthly basis. If the...
-
The Lewises are a married couple with a combined income of $46,700. They have one child with a health condition, and can claim the following deductions: $6,000 in property tax $7,000 in mortgage...
-
A rock is thrown at a 60.0 angle above the horizontal across level ground. It is thrown from a height of 2.00 m above the ground with a speed of 20.0 m/s and experiences no appreciable air...
-
According to Hostels.com. The average room rate for a New York City hotel was $204 in 2011. Suppose the Chamber of Commerce of New York City would like to test if this rate has changed recently by...
-
Given a standard normal distribution, value of k such that (a) P(Z < k) = 0.0427; (b) P(Z > /,) = 0.2946; (c) P(-0.93 < Z < k) = 0.7235.
-
For a randomly selected passenger on the Titanic, which of the following statements refers to a conditional probability? a. There is an \(8 \%\) chance that the passenger was booked in third class...
-
Presented below is a list of accounting events for the Longo Corporation. Classify each of the events as an operating (O) activity event, an investing (I) activity event, a financing (F) activity...
-
Cepeda Company uses a standard cost accounting system. During January, the company reported the following manufacturing variances. In addition, 8,000 units of product were sold at $8.00 per unit....
-
An $80,000 loan is amortized by monthly payments over 25 years. The interest rate charged is 10% compounded semi-annually. How much interest will be paid during the first three years?
-
On January 1, 2021, Gundy Enterprises purchases an office for $360,000, paying $60,000 down and borrowing the remaining $300,000, signing a 7%, 10-year mortgage. Installment payments of $3,483.25 are...
-
Share some of the basic details of the lives of the key founders of Daoism and Confucianism?
-
An industrial Oven costs $13613 and if a sales tax is of 8% is applied to the cost. Find the Amont of the TAX. FORMULA = (PRICE * TAX AMOUNT%)
-
Explain how you would evaluate a social media post using the functional perspective. Explain how you would evaluate a social media post using the dynamic perspective. Explain how you would evaluate a...
-
If a farmer is worried the price of soybeans will decrease before harvest, what are two possible hedging strategies they might employ? Be explicit about which components of cash price risk (futures...
-
of each firm: Suppose there are 6 firms in an industry. The following table gives the total sales Total Firm # Sales 1 200 2 90 3 80 4 60 5 40 6 30 Calculate the HHI for this industry: HHI =
-
For the point P(10,20) and Q(15,23), find the distance d(P.Q) and the coordinates of the midpoint M of the segment PQ. What is the distance? (Simplify your answer. Type an exact answer, using...
-
5. The two 12 kg slender rods are pin connected and released from rest at the position 0 = 60 (fig. 5). The spring is initially extended and has an unstretched length of 1.5 m. Neglect the mass of...
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
A solution made by dissolving 0.875 g of Co(NH3)4Cl3 in 25.0 g of water freezes at 20.56C. Calculate the number of moles of ions produced when 1 mole of Co(NH3)4Cl3 is dissolved in water and suggest...
-
The density of ethanol, a colorless liquid that is commonly known as grain alcohol, is 0.798 g/mL. Calculate the mass of 17.4 mL of the liquid.
-
To detect bombs that may be smuggled onto airplanes, the Federal Aviation Administration (FAA) will soon require all major airports in the United States to install thermal neutron analyzers. The...
-
ANSI X3.5-1970-the information system flowcharting standards published by the American National Standards Institute-defines four groups of flowchart symbols and illustrates conventions regarding...
-
Which type of diagram emphasizes the physical description of a system? (a) analytic flowchart (b) logical data flow diagram (DFD) (c) HIPO (d) IPO
-
Structured systems analysis can best be referred to as ( ___________________ ). (a) a process of increasingly complex sets of controls (b) a process of successive refinement (c) a procedure for...

Study smarter with the SolutionInn App



