If the reaction N2(g)+3H2(g)=2NH3(g) is at equilibrium, what direction will the reaction shift if NH3 gas...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
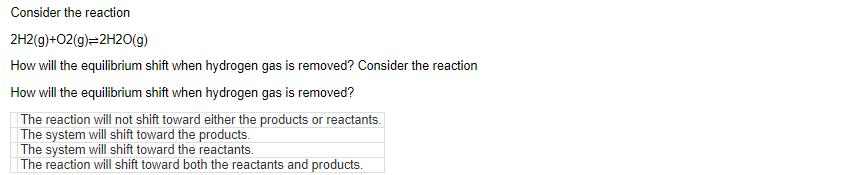
If the reaction N2(g)+3H2(g)=2NH3(g) is at equilibrium, what direction will the reaction shift if NH3 gas is added? If the reaction is at equilibrium, in what direction will the reaction shift if gas is added? The reaction will remain unchanged. The reaction will shift to produce both reactants and products. The reaction will shift to produce products. The reaction will shift to produce reactants. If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? The reaction will shift toward both reactants and products. The reaction will shift toward the reactants. The reaction will not shift toward products or reactants. The reaction will shift toward the products. Consider the reaction 2H2(g)+O2(g)=2H2O(g) How will the equilibrium shift when hydrogen gas is removed? Consider the reaction How will the equilibrium shift when hydrogen gas is removed? The reaction will not shift toward either the products or reactants. The system will shift toward the products. The system will shift toward the reactants. The reaction will shift toward both the reactants and products. Consider the exothermic reaction Fe203(s)+2Al(s) A1203(s)+2Fe(s) In what direction will the equilibrium shift when heat is removed? Consider the exothermic reaction In what direction will the equilibrium shift when heat is removed? The reaction will not shift toward either the products or reactants. The equilibrium will shift toward the reactants. The reaction will shift toward both the reactants and products. The equilibrium will shift toward the products. If the reaction N2(g)+3H2(g)=2NH3(g) is at equilibrium, what direction will the reaction shift if NH3 gas is added? If the reaction is at equilibrium, in what direction will the reaction shift if gas is added? The reaction will remain unchanged. The reaction will shift to produce both reactants and products. The reaction will shift to produce products. The reaction will shift to produce reactants. If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? If an endothermic reaction is at equilibrium, how will the equilibrium shift when heat is added to the reaction? The reaction will shift toward both reactants and products. The reaction will shift toward the reactants. The reaction will not shift toward products or reactants. The reaction will shift toward the products. Consider the reaction 2H2(g)+O2(g)=2H2O(g) How will the equilibrium shift when hydrogen gas is removed? Consider the reaction How will the equilibrium shift when hydrogen gas is removed? The reaction will not shift toward either the products or reactants. The system will shift toward the products. The system will shift toward the reactants. The reaction will shift toward both the reactants and products. Consider the exothermic reaction Fe203(s)+2Al(s) A1203(s)+2Fe(s) In what direction will the equilibrium shift when heat is removed? Consider the exothermic reaction In what direction will the equilibrium shift when heat is removed? The reaction will not shift toward either the products or reactants. The equilibrium will shift toward the reactants. The reaction will shift toward both the reactants and products. The equilibrium will shift toward the products.
Expert Answer:
Answer rating: 100% (QA)
ANSWER Concept and reason The concepts used to solve this question are to determine effect of the ch... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The following reaction is at equilibrium: N2 + 3H2 + 2NH3 The Keq at a particular temperature is 13.7. If the equilibrium [N2] is 0.055 M and the equilibrium [H2] is 1.62 M, what is the equilibrium...
-
The following reaction is at equilibrium: 4HBr + O2 + 2H2O + 2Br2 If [HBr] is 0.100 M, [O2] is 0.250 M, and [H2O] is 0.0500 M at equilibrium, what is [Br2] at equilibrium if the Keq is 0.770?
-
Consider the reaction of hydrogen gas with oxygen gas: Assuming complete reaction, which of the diagrams shown next represents the amounts of reactants and products left after the reaction? 2H2(g) +...
-
Q-1 A motor car shaft consists of a steel tube 30 mm internal diameter and 4 mm thick. The engine develops 10 kW at 2000 r.p.m. Find the maximum shear stress in the tube when the power is transmitted...
-
Refer to Problem M7-45. (a) If this is solved with the simplex algorithm, how many constraints and how many variables (including slack, surplus, and artificial variables) would be used? (b) If the...
-
An article in the Wall Street Journal, discussing large high-tech firms such as Amazon, Microsoft, and Google, stated, Todays high-tech giants may not be monopolies in the most classic sense....
-
For a sample of size n = 15, the following values were obtained: b0 = 3.71, b1 = 8.38, se = 1.13, (x x)2 = 7.71, x = 13.16. Construct a 95% prediction interval for an individual response when x = 8.
-
In 2011, Alliant Corporation acquired Centerpoint Inc. for $300 million, of which $50 million was allocated to goodwill . At the end of 2013, management has provided the following information for a...
-
moodle.uowplatform.edu.au FINAL ALTERNATIVE ASSESSMENT: FEBRUARY 2021 SESSION QUESTION 1 (25 MARKS) The following is a predictions of accounting data of ColdStor Company after careful study of the...
-
1.Identify the three countries with the most favorable collection experience as measured by their receivables turnover ratios. RTR US = RTR Saudi Arabia = RTR UK = 2. Which country appears to have...
-
1. Assume I won 5 million dollars in a lottery that pays installments of 1 million dollars a year for five years or a lump sum of less than 5 million dollars. If I take the installments, my first...
-
When employees leave an organization, they will have to be replaced and the replacement will need to be hired and trained. Estimates vary, but in order to recruit and get an employee up to speed in...
-
Can both the traditional deficit-reduction approach to training and the social media style of training be useful in the same organization? Describe.
-
The vast majority of organizations have some kind of presence on Facebook or Twitter. Others have LinkedIn, Pinterest, and Instagram accounts, which are an integral part of their overall social media...
-
Illiterate workers can suffer from embarrassment and fear that keeps them from admitting their problem. Instead, they may cope by asking questions, observing others, and relying on informal...
-
Would you conclude that an organization that offers greater pay improves employee satisfaction with their pay, their job, and life in general? Review the papers by Easterlin and Stevenson and Wolfers...
-
Consider the attached layout for a lift that involves putting a 9,000 lb heavy elevator hoisting machine onto the roof of a new building that is 118 ft high. A Terex HC 165 hydraulic crawler crane...
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
Which of the indicators in Fig. could be used for doing the titrations in Exercises 66 and 68? Fig Pheadl Red
-
Isocyanic acid (HNCO) can be prepared by heating sodium cyanate in the presence of solid oxalic acid according to the equation 2NaOCN(s) + H2C2O4(s) 2HNCO(l) + Na2C2O4(s)
-
Draw the Lewis structure of O2F2. Assign oxidation states and formal charges to the atoms in O2F2. The com-pound O2F2 is a vigorous and potent oxidizing and fluo-rinating agent. Are oxidation states...
-
A plane gate of uniform thickness holds back a depth of water as shown. Find the minimum weight needed to keep the gate closed. 0=30 L=3m Water w=2m P3.45
-
Obtain an expression for the kinetic energy flux, \(\int\left(V^{2} / 2 ight)\) \(ho \vec{V} \cdot d \vec{A}\), through cross section (1) of the control volume shown. x CV Width = w P4.12 h V
-
The velocity distribution for laminar flow in a long circular tube of radius \(R\) is given by the one-dimensional expression, \[\vec{V}=u \hat{i}=u_{\max }\left[1-\left(\frac{r}{R} ight)^{2} ight]...

Study smarter with the SolutionInn App


