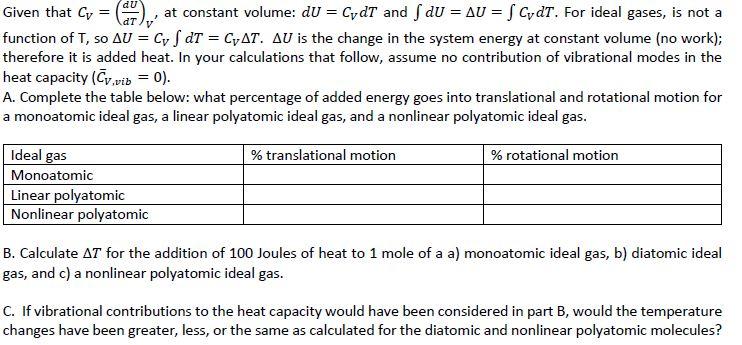
Given that Cv = ( at constant volume: dU = CydT and f dU = AU...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Given that Cv = ( at constant volume: dU = CydT and f dU = AU = f CydT. For ideal gases, is not a function of T, so AU = Cyf dT = CAT. AU is the change in the system energy at constant volume (no work); therefore it is added heat. In your calculations that follow, assume no contribution of vibrational modes in the heat capacity (Cv.vib = 0). A. Complete the table below: what percentage of added energy goes into translational and rotational motion for a monoatomic ideal gas, a linear polyatomic ideal gas, and a nonlinear polyatomic ideal gas. Ideal gas Monoatomic Linear polyatomic Nonlinear polyatomic % translational motion % rotational motion B. Calculate AT for the addition of 100 Joules of heat to 1 mole of a a) monoatomic ideal gas, b) diatomic ideal gas, and c) a nonlinear polyatomic ideal gas. C. If vibrational contributions to the heat capacity would have been considered in part B, would the temperature changes have been greater, less, or the same as calculated for the diatomic and nonlinear polyatomic molecules? Given that Cv = ( at constant volume: dU = CydT and f dU = AU = f CydT. For ideal gases, is not a function of T, so AU = Cyf dT = CAT. AU is the change in the system energy at constant volume (no work); therefore it is added heat. In your calculations that follow, assume no contribution of vibrational modes in the heat capacity (Cv.vib = 0). A. Complete the table below: what percentage of added energy goes into translational and rotational motion for a monoatomic ideal gas, a linear polyatomic ideal gas, and a nonlinear polyatomic ideal gas. Ideal gas Monoatomic Linear polyatomic Nonlinear polyatomic % translational motion % rotational motion B. Calculate AT for the addition of 100 Joules of heat to 1 mole of a a) monoatomic ideal gas, b) diatomic ideal gas, and c) a nonlinear polyatomic ideal gas. C. If vibrational contributions to the heat capacity would have been considered in part B, would the temperature changes have been greater, less, or the same as calculated for the diatomic and nonlinear polyatomic molecules?
Expert Answer:
Answer rating: 100% (QA)
A Complete the table below what percentage of added energy goes into transl ational and rot ational motion for a mon oat omic ideal gas a linear poly ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
2. Suppose money demand function is given by M-500*(0.25-r), r is real interest rate, money supply M' is 100 (billion dollars) and the required reserve ratio is 20%. a) Calculate equilibrium interest...
-
The molar heat capacity of a diatomic molecule is 29.1 J/K mol. Assuming the atmosphere contains only nitrogen gas and there is no heat loss, calculate the total heat intake (in kilojoules) if the...
-
The vibrational contribution to the molar heat capacity of a gas of nonlinear molecules is given in statistical mechanics by the formula i=1 " style="" class="fr-fic fr-dib"> where u i =...
-
Consider examples 9.2 and 9.3. Assume that social damage is quadratic as in the examples. Following section 9.2.3, assume that the regulator can only set a uniform tax. Determine the formula for the...
-
Early this year, ZeZe Inc. paid a $52,000 legal fee in connection with a dispute over ZeZes title to investment land. ZeZes auditors required the corporation to expense the payment on this years...
-
Scan the Web and identify the Internet marketing techniques of two to three companies. Start with the companys home page. What functionality does the page contain (just information, online selling...
-
Guatemalan Imports is the only company with reliable sources for its imported gifts. The company does a brisk business with specialty stores such as Pier 1 Imports. Guatemalan Imports' recent success...
-
1. How should Oreos success in social engagement be evaluated? What metrics would be most useful? Consider both the usage-specific metrics that a social media provider might supply as well as...
-
Does APPLE issues stocks in foreign countries? If yes, are there any correlations in the stock price movements?
-
An experiment was performed to improve the yield of a chemical process. Four factors were selected, and two replicates of a completely randomized experiment were run. The results are shown in the...
-
Your client was fired from her job. She claims the employer engaged in discrimination and that the employer's actions violated the law. Your client worked for the employer in Maryland. The employer...
-
Consider a theory which states that an increase in interest rates will lead to an increase in savings. How would the principle of ceteris paribus be important in investigating the predictive power of...
-
Would you expect economists to disagree less about public policy as time goes on? Why or why not? Can their differences be completely eliminated? Why or why not?
-
Does the fact that there are different schools of thought in economics reduce its validity as an academic discipline?
-
If models are not capable of representing the real world in any detail and rely too much on assumptions, then what value can they be?
-
Why do differences in values lead to disagreements among economists?
-
On January 1, 2021, Eagle Company borrows $19,000 cash by signing a four-year, 8% installment note. The note requires four equal payments of $5,737, consisting of accrued interest and principal on...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Aqueous copper(II) sulfate solution is blue in color. When aqueous potassium fluoride is added, a green precipitate is formed. When aqueous potassium chloride is added instead, a bright-green...
-
Assuming ideal behavior, calculate the density of gaseous HF at its normal boiling point (19.5C). The experimentally measured density under the same conditions is 3.10 g/L. Account for the...
-
Calculate the molecular mass or formula mass (in amu) of each of the following substances: (a) CH4, (b) NO2, (c) SO3, (d) C6H6, (e) NaI, (f) K2SO4, (g) Ca3 (PO4)2.
-
Refer to the information in S6-1 . Calculate the gross margin for the month of August. Data from S6-1 Garda's Equipment has the following items in its inventory on August 1: The company uses the...
-
Elmo's Music carries a large inventory of guitars and other musical instruments. The store uses the FIFO method and a perpetual inventory system. Company records indicate the following for a...
-
The account number 211031 most likely refers to a. Liabilities b. Current liabilities c. Accounts payable d. An individual supplier

Study smarter with the SolutionInn App


