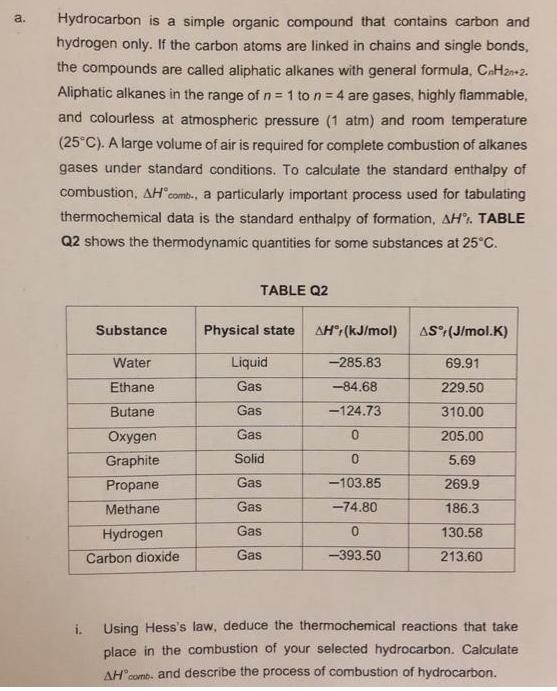
Hydrocarbon is a simple organic compound that contains carbon and a. hydrogen only. If the carbon...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Hydrocarbon is a simple organic compound that contains carbon and a. hydrogen only. If the carbon atoms are linked in chains and single bonds, the compounds are called aliphatic alkanes with general formula, CaH2n+2. Aliphatic alkanes in the range ofn = 1 to n = 4 are gases, highly flammable, and colourless at atmospheric pressure (1 atm) and room temperature (25°C). A large volume of air is required for complete combustion of alkanes gases under standard conditions. To calculate the standard enthalpy of combustion, AH comb., a particularly important process used for tabulating thermochemical data is the standard enthalpy of formation, AH. TABLE Q2 shows the thermodynamic quantities for some substances at 25°C. TABLE Q2 Substance Physical state AH" (kJ/mol) AS (J/mol.K) Water Liquid -285.83 69.91 Ethane Gas -84.68 229.50 Butane Gas -124.73 310.00 Oxyg Gas 205.00 Graphite Solid 5.69 Propane Gas -103.85 269.9 Methane Gas -74.80 186.3 Hydrogen Gas 130.58 Carbon dioxide Gas -393.50 213.60 i. Using Hess's law, deduce the thermochemical reactions that take place in the combustion of your selected hydrocarbon. Calculate AH"comb. and describe the process of combustion of hydrocarbon. ii. Calculate the standard entropy change of the reaction and explain the spontaneity of the reaction with the decrease of temperature. [5 marks) b. The purpose of electroplating is to prevent corrosion. Electroplating is a process of coating an inferior object with a superior metal through electrolysis. Electroplating process requires expensive and efficient treatment of waste. Eventually, due to contamination, the quality of the coated objects drops. "Spoon is an inexpensive metal, but when coated with a superior metal they become valuable" By using your knowledge in electrolysis, design an experiment to discuss the statement. Provide your arguments clearly on how to improve the quality of the coated metal formed by electroplating. [10 marks] Hydrocarbon is a simple organic compound that contains carbon and a. hydrogen only. If the carbon atoms are linked in chains and single bonds, the compounds are called aliphatic alkanes with general formula, CaH2n+2. Aliphatic alkanes in the range ofn = 1 to n = 4 are gases, highly flammable, and colourless at atmospheric pressure (1 atm) and room temperature (25°C). A large volume of air is required for complete combustion of alkanes gases under standard conditions. To calculate the standard enthalpy of combustion, AH comb., a particularly important process used for tabulating thermochemical data is the standard enthalpy of formation, AH. TABLE Q2 shows the thermodynamic quantities for some substances at 25°C. TABLE Q2 Substance Physical state AH" (kJ/mol) AS (J/mol.K) Water Liquid -285.83 69.91 Ethane Gas -84.68 229.50 Butane Gas -124.73 310.00 Oxyg Gas 205.00 Graphite Solid 5.69 Propane Gas -103.85 269.9 Methane Gas -74.80 186.3 Hydrogen Gas 130.58 Carbon dioxide Gas -393.50 213.60 i. Using Hess's law, deduce the thermochemical reactions that take place in the combustion of your selected hydrocarbon. Calculate AH"comb. and describe the process of combustion of hydrocarbon. ii. Calculate the standard entropy change of the reaction and explain the spontaneity of the reaction with the decrease of temperature. [5 marks) b. The purpose of electroplating is to prevent corrosion. Electroplating is a process of coating an inferior object with a superior metal through electrolysis. Electroplating process requires expensive and efficient treatment of waste. Eventually, due to contamination, the quality of the coated objects drops. "Spoon is an inexpensive metal, but when coated with a superior metal they become valuable" By using your knowledge in electrolysis, design an experiment to discuss the statement. Provide your arguments clearly on how to improve the quality of the coated metal formed by electroplating. [10 marks]
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Using the data in Appendix 3, calculate the standard entropy changes for the following reactions at 25C: (a) S(s) + O2(g) SO2(g) (b) MgCO3(s) MgO(s) + CO2(g)
-
Using the data in Appendix 3, calculate the standard entropy changes for the following reactions at 25C: (a) H2(g) + CuO(s) Cu(s) + H2O(g) (b) 2Al(s) + 3ZnO(s) Al2O3(s) + 3Zn(s) (c) CH4(g) + 2O2(g)...
-
Calculate the standard entropy change for the reaction at 25 C. Standard molar entropy values can be found in this. C,H,(g) + 50,(g) 3 CO,(g) + 4 H,0(g) ASixn J/K
-
Austin Limited is trying to determine the value of its ending inventory as of February 28, 2019, the companys year-end. The following transactions occurred, and the accountant asked your help in...
-
You are given the following information: (a) Compute the amount of net revaluations in each of the four years. (b) Compute the change in U.S.-owned assets overseas in year 1. (c) Compute the current...
-
In ten years how will the e-commerce landscape be different?
-
What are the two tests for determining whether a party may be joined in a lawsuit?
-
Phelps Company distributed the following dividends to its stockholders: (a) 450,000 shares of Bedrock Corporation stock, carrying value of investment, $975,000; fair market value, $1,350,000. (b)...
-
We are looking at Plowman's and Wilson's "Strategy and Tactics in Strategic Communication: Examining their Intersection with Social Media Use" ( International Journal of Strategic Communication, 12,...
-
A cash budget, by quarter, is given below for a retail company (000 omitted). The company requires a minimum cash balance of $5,000 to start each quarter. Required: Fill in the missing amounts in the...
-
You own 1 million shares in Locos Inc a 100% equity company that your purchased paying $10 per share. Last week the company was worth $100 million with 10 million shares outstanding. Yesterday the...
-
What is JIT-inventory system and how significant is it to reduce inventory cost?
-
List and briefly explain at least five of the reasons why going public is not a good idea.
-
Briefly explain the way by which a firm would know if it needs new financing.
-
Explain how the wholesale layout is different from both retail layout and manufacturing layout.
-
What is the safety stock? Show how its calculation can be different when the demand or lead time becomes either constant or variable.
-
Do you think sales tax should be collected on internet purchases? Why, or why not? What are the positive and negative implications of collecting sales tax on internet purchases? explain.
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
The equilibrium constant (KP) for the reaction H2(g) + CO2(g) H2O(g) + CO(g) is 4.40 at 2000 K. (a) Calculate DG for the reaction. (b) Calculate DG for the reaction when the partial pressures are...
-
Protons can be accelerated to speeds near that of light in particle accelerators. Estimate the wavelength (in nm) of such a proton moving at 2.90 108 m/s. (Mass of a proton = 1.673 10-27 kg.)
-
Calculate the molarity and the molality of an NH3 solution made up of 30.0 g of NH3 in 70.0 g of water. The density of the solution is 0.982 g/mL.
-
Give examples of non-current (or fixed) assets. In what circumstances would some of the items you have listed be current assets?
-
What is the difference between a non-current asset and a current asset?
-
You are given the following simplified statement of financial position of the Sandin Castle Company: a Calculate the current ratio. b Calculate the liquidity ratio. c After preparing the statement of...

Study smarter with the SolutionInn App


