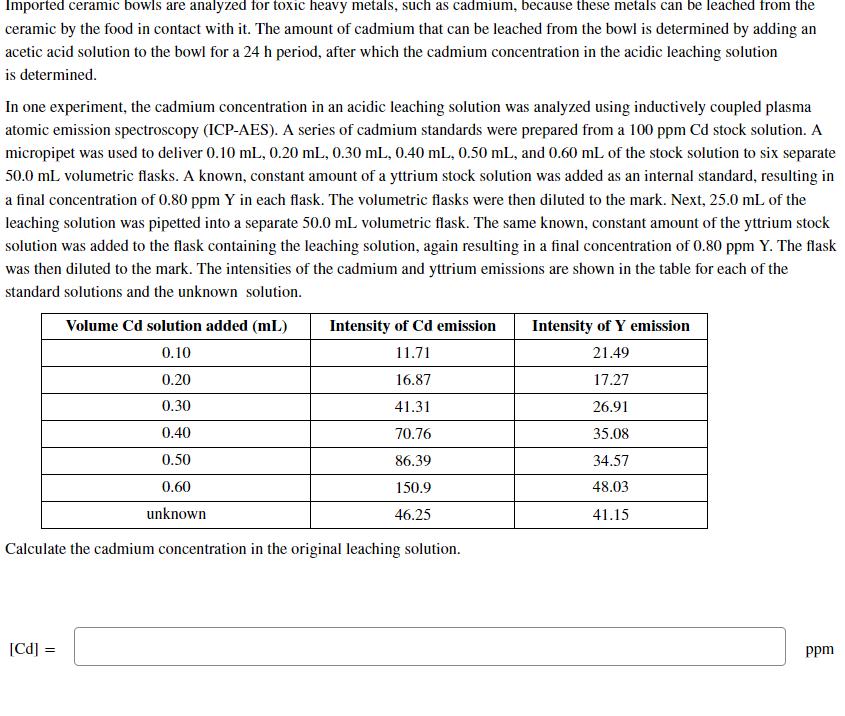
Imported ceramic bowls are analyzed for toxic heavy metals, such as cadmium, because these metals can...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Imported ceramic bowls are analyzed for toxic heavy metals, such as cadmium, because these metals can be leached from the ceramic by the food in contact with it. The amount of cadmium that can be leached from the bowl is determined by adding an acetic acid solution to the bowl for a 24 h period, after which the cadmium concentration in the acidic leaching solution is determined. In one experiment, the cadmium concentration in an acidic leaching solution was analyzed using inductively coupled plasma atomic emission spectroscopy (ICP-AES). A series of cadmium standards were prepared from a 100 ppm Cd stock solution. A micropipet was used to deliver 0.10 mL, 0.20 mL, 0.30 mL, 0.40 mL, 0.50 mL, and 0.60 mL of the stock solution to six separate 50.0 mL volumetric flasks. A known, constant amount of a yttrium stock solution was added as an internal standard, resulting in a final concentration of 0.80 ppm Y in each flask. The volumetric flasks were then diluted to the mark. Next, 25.0 mL of the leaching solution was pipetted into a separate 50.0 mL volumetric flask. The same known, constant amount of the yttrium stock solution was added to the flask containing the leaching solution, again resulting in a final concentration of 0.80 ppm Y. The flask was then diluted to the mark. The intensities of the cadmium and yttrium emissions are shown in the table for each of the standard solutions and the unknown solution. Volume Cd solution added (mL) 0.10 0.20 0.30 0.40 0.50 0.60 unknown [Cd] = Intensity of Cd emission 11.71 16.87 41.31 70.76 86.39 150.9 46.25 Calculate the cadmium concentration in the original leaching solution. Intensity of Y emission 21.49 17.27 26.91 35.08 34.57 48.03 41.15 ppm Imported ceramic bowls are analyzed for toxic heavy metals, such as cadmium, because these metals can be leached from the ceramic by the food in contact with it. The amount of cadmium that can be leached from the bowl is determined by adding an acetic acid solution to the bowl for a 24 h period, after which the cadmium concentration in the acidic leaching solution is determined. In one experiment, the cadmium concentration in an acidic leaching solution was analyzed using inductively coupled plasma atomic emission spectroscopy (ICP-AES). A series of cadmium standards were prepared from a 100 ppm Cd stock solution. A micropipet was used to deliver 0.10 mL, 0.20 mL, 0.30 mL, 0.40 mL, 0.50 mL, and 0.60 mL of the stock solution to six separate 50.0 mL volumetric flasks. A known, constant amount of a yttrium stock solution was added as an internal standard, resulting in a final concentration of 0.80 ppm Y in each flask. The volumetric flasks were then diluted to the mark. Next, 25.0 mL of the leaching solution was pipetted into a separate 50.0 mL volumetric flask. The same known, constant amount of the yttrium stock solution was added to the flask containing the leaching solution, again resulting in a final concentration of 0.80 ppm Y. The flask was then diluted to the mark. The intensities of the cadmium and yttrium emissions are shown in the table for each of the standard solutions and the unknown solution. Volume Cd solution added (mL) 0.10 0.20 0.30 0.40 0.50 0.60 unknown [Cd] = Intensity of Cd emission 11.71 16.87 41.31 70.76 86.39 150.9 46.25 Calculate the cadmium concentration in the original leaching solution. Intensity of Y emission 21.49 17.27 26.91 35.08 34.57 48.03 41.15 ppm
Expert Answer:

Related Book For 

An Introduction To Statistical Methods And Data Analysis
ISBN: 9781305465527
7th Edition
Authors: R. Lyman Ott, Micheal T. Longnecker
Posted Date:
Students also viewed these economics questions
-
The EPA set emission standards for vinyl chloride, a toxic substance that is carcinogenic to humans. The Clean Air Act says such standards must be "at the level which.provides an ample margin of...
-
AES was formed in 1996, at which time it hired eight employees. At a meeting of these employees in 1997, they expressed concern that the company might not survive, as it was using outdated equipment....
-
Acetic acid can be manufactured by combining methanol with carbon monoxide, an example of a carbonylation reaction: (a) Calculate the equilibrium constant for the reaction at 25°C. (b)...
-
____, one-way ANOVA compares the means of three or more independent samples.
-
Prove that E[b' b] = ' + 2Kk=1(1/k) where b is the ordinary least squares estimator and k is a characteristic root of X' X.
-
A steel tube having an outer diameter of 2.5 in. is used to transmit 9 hp when turning at 27 rev/min. Determine the inner diameter d of the tube to the nearest 18 in. if the allowable shear stress is...
-
Lorraine Scott asks your help in understanding the essentials of effective budgeting. Identify the essentials for Lorraine.
-
Refer to the data in Exercise 5.7. In Exercise 5.7 Number of direct labor hours ...... 6,720 Direct labor cost .........$67,200 Overhead cost ............$50,400 During the year, the following actual...
-
Find an equation of the tangent line to the curve y 7 at the point (1,7) Tangent line: y = Check your result by plotting the curve and the tangent line together.
-
Savita Kapur, CEO, founded Absent Clothing Company (ACC) in 2005. ACC sells practical athletic wear to service the yoga and Pilates market. Savita originally created ACC with the intention of...
-
A department of Delta Company incurred the following costs for the month of June. Variable costs, and the variable portion of mixed costs, are a function of the number of units of activity: Activity...
-
The temperature of the dry ice (solid carbon dioxide) in ice cream vending carts is - 78.5 degree C. What is temperature on the Fahrenheit and on the Kelvin scales?
-
Why is tap water a homogenous mixture, whereas distilled wateris a pure substance? Explain.
-
What is meant by Written representations? What are the matters to be included in the written representations?
-
a) Using a molecular orbital diagram of NO, calculate the bond order (show calculation) b) What effect would the addition of one electron to form NO- have on the bond length and why? c)Is NO-...
-
Describe the best practices for structuring various types of business documents, such as reports, proposals, and business plans. How does the structure impact readability and comprehension?
-
7-7. (Bond relationship) Crawford Inc. has two bond issues outstanding, both paying the same annual interest of $55, called Series A and Series B. Series A has a maturity of 12 years, whereas Series...
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
In the January 2004 issue of Consumer Reports, an article titled "Cut the Fat" described some of the possible problems in the diets of the U. S. public. The following table gives data on the increase...
-
Defining the Problem (1). Lead is an environmental pollutant especially worthy of attention because of its damaging effects on the neurological and intellectual development of children. Morton et al....
-
Refer to Exercise 14.8. a. Draw a profile plot for the two factors, age and product type. b. Perform appropriate F tests and draw conclusions from these tests concerning the effects of age and...
-
Nutrition Products, Inc., uses a traditional product costing system to assign over- head costs uniformly to all products. To meet Food and Drug Administration require- ments and to assure its...
-
The New York Times reported that Neil Simon planned to open his play London Suite off Broadway. Why? For financial reasons. Producer Emanuel Azenberg predicted the following costs before the play...
-
Agribiz Food Products, Inc. produces a wide variety of food and related products. The companys tomato canning operation relies partly on tomatoes grown on Agribizs own farms and partly on tomatoes...

Study smarter with the SolutionInn App


