In 9 Days 0 General Scien... Fuad Hassan - CA General Science 2 A Tier Practice:...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
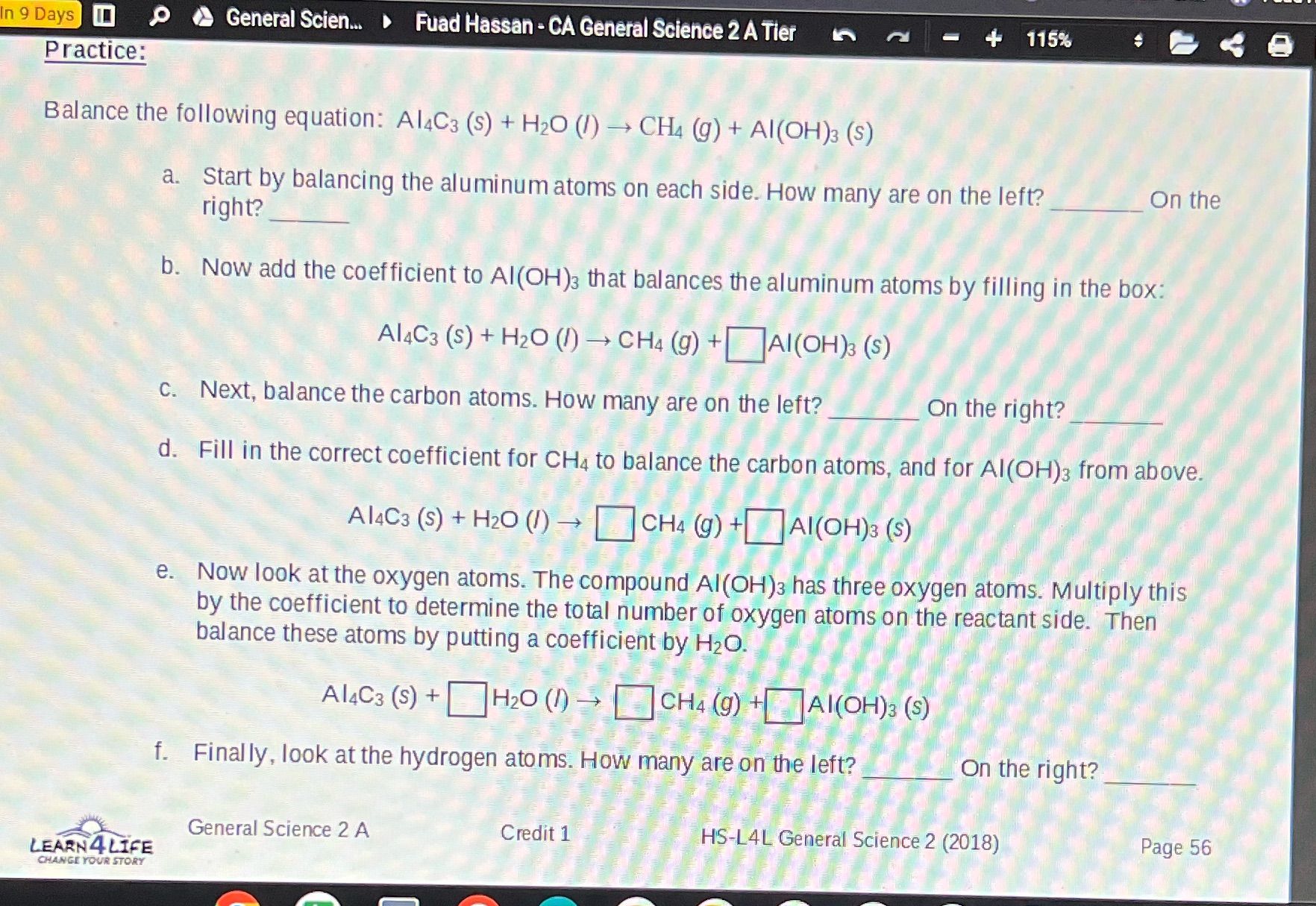
In 9 Days 0 General Scien... Fuad Hassan - CA General Science 2 A Tier Practice: 115% Balance the following equation: Al4C3 (s) + H2O (l) CH4 (g) + Al(OH)3 (S) a. Start by balancing the aluminum atoms on each side. How many are on the left? right? On the b. Now add the coefficient to Al(OH)3 that balances the aluminum atoms by filling in the box: Al4C3 (s) + H2O (1) CH4 (g) + Al(OH)3 (S) c. Next, balance the carbon atoms. How many are on the left? On the right? d. Fill in the correct coefficient for CH4 to balance the carbon atoms, and for Al(OH)3 from above. A14C3 (s) + H2O (1) CH4 (g) +Al(OH)3 (S) e. Now look at the oxygen atoms. The compound Al(OH)3 has three oxygen atoms. Multiply this by the coefficient to determine the total number of oxygen atoms on the reactant side. Then balance these atoms by putting a coefficient by HO. Al4C3 (s) + H2O (1) CH4 (g) +Al(OH)3 (s) f. Finally, look at the hydrogen atoms. How many are on the left? General Science 2 A LEARN 4 LIFE CHANGE YOUR STORY Credit 1 On the right? HS-L4L General Science 2 (2018) Page 56 In 9 Days 0 General Scien... Fuad Hassan - CA General Science 2 A Tier Practice: 115% Balance the following equation: Al4C3 (s) + H2O (l) CH4 (g) + Al(OH)3 (S) a. Start by balancing the aluminum atoms on each side. How many are on the left? right? On the b. Now add the coefficient to Al(OH)3 that balances the aluminum atoms by filling in the box: Al4C3 (s) + H2O (1) CH4 (g) + Al(OH)3 (S) c. Next, balance the carbon atoms. How many are on the left? On the right? d. Fill in the correct coefficient for CH4 to balance the carbon atoms, and for Al(OH)3 from above. A14C3 (s) + H2O (1) CH4 (g) +Al(OH)3 (S) e. Now look at the oxygen atoms. The compound Al(OH)3 has three oxygen atoms. Multiply this by the coefficient to determine the total number of oxygen atoms on the reactant side. Then balance these atoms by putting a coefficient by HO. Al4C3 (s) + H2O (1) CH4 (g) +Al(OH)3 (s) f. Finally, look at the hydrogen atoms. How many are on the left? General Science 2 A LEARN 4 LIFE CHANGE YOUR STORY Credit 1 On the right? HS-L4L General Science 2 (2018) Page 56
Expert Answer:

Posted Date:
Students also viewed these physics questions
-
An inductor has a linear change in current from 50 mA to 100 mA in 2 ms and induces a voltage of 160 mV. Calculate the value of the inductor.
-
Regina Repair Shop has a monthly target operating income of $32,000. Variable expenses are 75% of sales, and monthly fixed expenses are $8,000. Requirements 1. Compute the monthly margin of safety in...
-
Assume the same information as E19-23B, except that based on the weight of available evidence in 2014, it is more likely than not that 40 percent of the benefits of any loss carryforward will not be...
-
Management of Bicent Company uses the following unit costs for the one product it manufactures: Required: Prepare projected income statements for June 20X1 for management purposes under each of the...
-
Elm Corporation and Maple Company have announced terms of an exchange agreement under which Elm will issue 8,000 shares of its $10 par value common stock to acquire all of Maple Companys assets. Elm...
-
Prepare the Statement of Cash flows for the year ended June 30, 2021 using the Direct Method. Hint Use the Cash T-account on the General Ledger tab to identify the sources and uses of cash. List cash...
-
Discuss the financial aspects of El Wey Del Norte Business. What are related start-up costs? What is the earning potential? What is the potential return on investment?
-
Revu's Company's costs for the year were: Factory overhead applied: $400,000 Cost of goods manufactured: $880,000 Factory overhead incurred: $392,600 Finished goods inventory was $218,000 on January...
-
How is differential analysis used in deciding whether to keep or drop product lines?
-
Define Managed Care Organizations (MCOs) and Accountable Care Organizations (ACOs) and what are the differences and similarities between them ?
-
Betram Chemicals Company processes a number of chemical compounds used in producing industrial cleaning products. One compound is decomposed into two chemicals: anderine and dofinol. The cost of...
-
1.We have 4 ways to recruit operators for out factory: on line internet, head hunters, ads in trade journals, and on-line company intranet.HR has discovered on-line internet ads reach 300 people per...
-
You have been asked by management to explain the variances in costs under your inpatient capitated contract. The following data is provided. Problem 1: What dollar amount of the total variance is...
-
A genetically engineered strain of Escherichia coli (E. coli) is used to synthesize human insulin for people suffering from type I diabetes mellitus. In the following simplified reaction scheme,...
-
The following information pertains to Ping Company for 2011. Ending inventory consisted of 30 units. Ping sold 210 units at \(\$ 50\) each. All purchases and sales were made with cash. Required a....
-
University Sales had the following transactions for T-shirts for 2011, its first year of operations. During the year, University Sales sold 725 T-shirts for \(\$ 20\) each. Required a. Compute the...
-
The Keys Company had cash sales of \(\$ 250,000\) for 2011, its first year of operation. On April 2, the company purchased 200 units of inventory at \(\$ 350\) per unit. On September 1 , an...

Study smarter with the SolutionInn App

