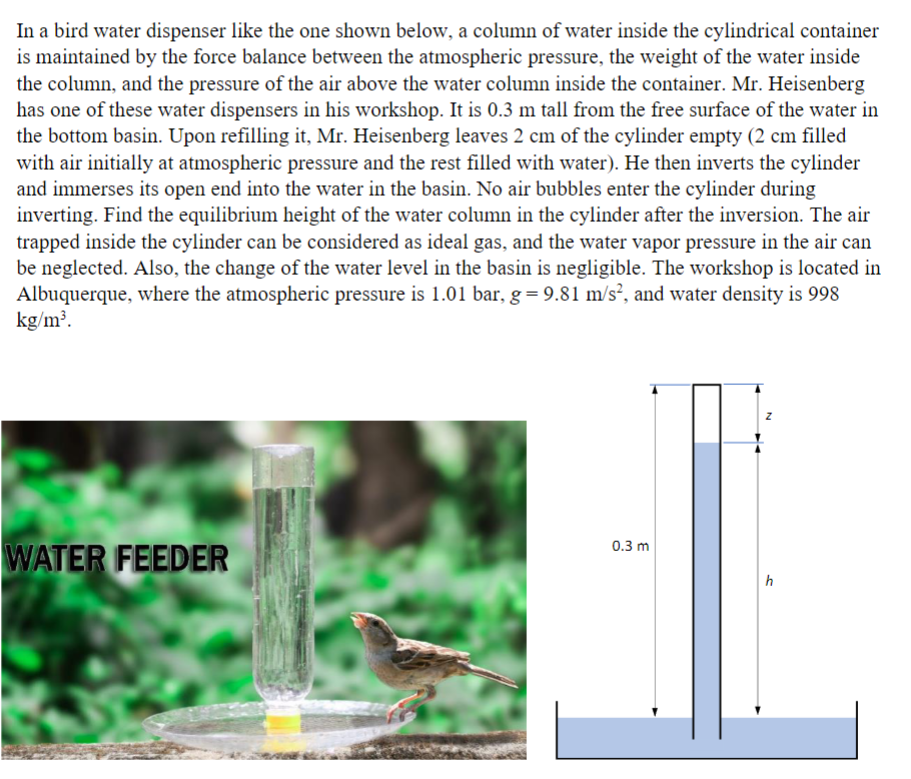
In a bird water dispenser like the one shown below, a column of water inside the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In a bird water dispenser like the one shown below, a column of water inside the cylindrical container is maintained by the force balance between the atmospheric pressure, the weight of the water inside the column, and the pressure of the air above the water column inside the container. Mr. Heisenberg has one of these water dispensers in his workshop. It is 0.3 m tall from the free surface of the water in the bottom basin. Upon refilling it, Mr. Heisenberg leaves 2 cm of the cylinder empty (2 cm filled with air initially at atmospheric pressure and the rest filled with water). He then inverts the cylinder and immerses its open end into the water in the basin. No air bubbles enter the cylinder during inverting. Find the equilibrium height of the water column in the cylinder after the inversion. The air trapped inside the cylinder can be considered as ideal gas, and the water vapor pressure in the air can be neglected. Also, the change of the water level in the basin is negligible. The workshop is located in Albuquerque, where the atmospheric pressure is 1.01 bar, g = 9.81 m/s², and water density is 998 kg/m³. 0.3 m WATER FEEDER h In a bird water dispenser like the one shown below, a column of water inside the cylindrical container is maintained by the force balance between the atmospheric pressure, the weight of the water inside the column, and the pressure of the air above the water column inside the container. Mr. Heisenberg has one of these water dispensers in his workshop. It is 0.3 m tall from the free surface of the water in the bottom basin. Upon refilling it, Mr. Heisenberg leaves 2 cm of the cylinder empty (2 cm filled with air initially at atmospheric pressure and the rest filled with water). He then inverts the cylinder and immerses its open end into the water in the basin. No air bubbles enter the cylinder during inverting. Find the equilibrium height of the water column in the cylinder after the inversion. The air trapped inside the cylinder can be considered as ideal gas, and the water vapor pressure in the air can be neglected. Also, the change of the water level in the basin is negligible. The workshop is located in Albuquerque, where the atmospheric pressure is 1.01 bar, g = 9.81 m/s², and water density is 998 kg/m³. 0.3 m WATER FEEDER h
Expert Answer:
Answer rating: 100% (QA)
To find the equilibrium height of the water column in the cylinder after the inversion we need to co... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
A closed tank contains oil with a specific gravity 0.85. If the gauge pressure at a point 10 feet below the oil surface is 23. 7 psi (lb/in.2), determine the absolute pressure and gauge pressure (in...
-
Explain why there are several choices (in StatTools and other packages) for the type of confidence intervals in one-way ANOVA. Specifically, whatp roblem do these variations attempt to solve?
-
A position vector has components x-34.6 m and y = -53.5 m. Find the vectors length and angle with the x-axis.
-
What is the mirror-image rule?
-
On January 1, 2014, Richard Corporation had retained earnings of $550,000. During the year, Richard had the following selected transactions. 1. Declared cash dividends $96,000. 2. Corrected...
-
DataBase managment design. Do the dowsings: 1. Run the sql commands written in ddl.sql file attached 2. Create_a dummy data. You can use "fake data generators" 3. Write followings - Write the query...
-
Under Armour, Inc. is an American supplier of sportswear and casual apparel. Following are selected financial data for the company for the period 20092013. a. Calculate Under Armours annual...
-
2. The number of doctors of osteopathic medicine (in thousands) in the U.S. from 2000 through 2008 is given below. Year 2000 2001 2002 2003 2004 2005 2006 2007 2008 Number (in thousands) 44.9 47.0...
-
Why do taxpayers try to increase the debt- equity ratio of a corporation?
-
Jose and Roberta were divorced in 2019. Jose is required to pay alimony of $35,000 per year to Roberta. Are any of the payments by Jose tax deductible as alimony?
-
What rules do traditional IRAs and Roth IRAs have in common?
-
Is salvage value ever considered when using MACRS depreciation? When using MACRS, is depreciation ever allowed on an asset in the year of disposal?
-
How is basis computed when a sale of shares of stock occurs?
-
An Electrical Company has three workshops in Koforidua, Accra and Kumasi. These workshops produce the following quantities of electric fan. Workshops Monthly production (units) Koforidua 130 Accra 70...
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
`The standard enthalpies of formation of ions in aqueous solutions are obtained by arbitrarily assigning a value of zero to H+ ions; that is, Hf [H+(aq)] = 0. (a) For the following reaction ...
-
A gaseous hydrocarbon (containing C and H atoms) in a container of volume 20.2 L at 350 K and 6.63 atm reacts with an excess of oxygen to form 205.1 g of CO2 and 168.0 g of H2 O. What is the...
-
The SO2 present in air is mainly responsible for the acid rain phenomenon. Its concentration can be determined by titrating against a standard permanganate solution as follows: Calculate the number...
-
You can think of a financial plan as a "financial road map to guide you through life." Develop a visual display that illustrates this concept and the five steps of the financial planning process. Try...
-
Summarize the five steps that make up the financial planning process.
-
Visit your campus career counseling office to learn about the services available to assist you with your career search and your job search. What career management services, if any, are available...

Study smarter with the SolutionInn App


