Why is it impossible to perform a liquid-liquid extraction using HO and CHOH as the two...
Fantastic news! We've Found the answer you've been seeking!
Question:
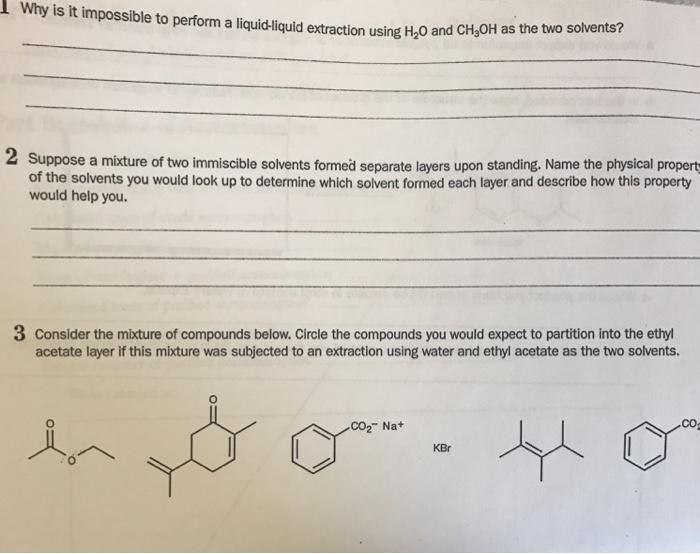
Transcribed Image Text:
Why is it impossible to perform a liquid-liquid extraction using H₂O and CH₂OH as the two solvents? 2 Suppose a mixture of two immiscible solvents formed separate layers upon standing. Name the physical propert of the solvents you would look up to determine which solvent formed each layer and describe how this property would help you. 3 Consider the mixture of compounds below. Circle the compounds you would expect to partition into the ethyl acetate layer if this mixture was subjected to an extraction using water and ethyl acetate as the two solvents. lon CO₂ Na+ KBr Why is it impossible to perform a liquid-liquid extraction using H₂O and CH₂OH as the two solvents? 2 Suppose a mixture of two immiscible solvents formed separate layers upon standing. Name the physical propert of the solvents you would look up to determine which solvent formed each layer and describe how this property would help you. 3 Consider the mixture of compounds below. Circle the compounds you would expect to partition into the ethyl acetate layer if this mixture was subjected to an extraction using water and ethyl acetate as the two solvents. lon CO₂ Na+ KBr
Expert Answer:
Answer rating: 100% (QA)
1 LiquidLiquid extraction refers to or separating drawing compound o... View the full answer

Related Book For 

Introduction to Chemical Engineering Thermodynamics
ISBN: 978-0071247085
7th edition
Authors: J. M. Smith, H. C. Van Ness, M. M. Abbott
Posted Date:
Students also viewed these chemistry questions
-
A saturated liquid feed at 125 psia contains 200 lbmol/h of 5 mol% iC4, 20 mol% nC4, 35 mol% iC5, and 40 mol% nC5. This feed is to be distilled at 125 psia with a column equipped with a total...
-
Calculate the mass fraction stochiometric ratio for the combustion of Ethane (CHs) in air (20% v/v Oz 80% v/v N2) Given the following Atomic Weights for the elements and their valencies." Symbol...
-
A binary liquid system exhibits LLE at 25C. Determine from each of the following sets of miscibility data estimates for parameters A12 and A21 in the Margules equation at 25CC: (a) x1a = 0.10, x1 =...
-
The following are some examples of typical business transactions: 1. Purchased office equipment on credit. 2. The owner contributed capital. 3. Purchased office furniture for cash. 4. Received...
-
In a speech to financial analysts, William Poole, former Chairman of the St. Louis Federal Reserve said: Over most of the post-World War II period, the personal saving rate averaged about 6 percent,...
-
As HR director at Crangle Fixtures, your bonus this year is based on your ability to cut employee benefit costs. Your boss has said that its okay to shift some of the costs over to employees (right...
-
True or False. The derivation of system matrices involves the assembly of element matrices.
-
Pet Bottle Company (PBC) manufactures plastic two-liter bottles for the beverage industry. The cost standard per 100 two-liter bottles are as follows: Cost Category Standard Cost Per 100 Two ???...
-
Complete the following challenge and upload your screenshot proof. 1. You won't be given many tips on this but you should be able to figure it out. 2. Show evidence of a successful three way...
-
The following balances come from the trial balance of Wilson State College as of the end of the 2020 fiscal year. Required a. Prepare a statement of revenues, expenses, and changes in net position...
-
This is a coding class, use python programming to answer questions (a) Image above--Calculate the correlation between the LIFEEXPECTANCY and INFANTMORTALITY features. (b) The image below shows a...
-
Sharaf is an Iranian national. He is Baha'i, a minority religion in a country where Islam is dominant. Sharaf could not apply for any government jobs nor obtain a business license, all because of his...
-
Some parts of this question ask you to apply course concepts to the song I Can, by Nas. This is a rap song that contains elements of classical music, specifically Beethoven's F Elise. The artist raps...
-
How can accountability be maintained in collaborative or decentralized organizational structures, where traditional hierarchical methods may be less effective, without stifling innovation and...
-
what ways can technology and data analytics be leveraged to enhance accountability in organizations, and what are the potential risks and ethical considerations associated with such technological...
-
Identify the general legal principle relating to finding, including reference to its primary source(s) relating to property law in Australia with respect to the scenario below. SCENARIO Emma is...
-
Use the decomposition ALU to solve the system Az = b, where 1 0 4 1 L= 3 1 0U=0 -2 2 b= 24 1 31 0 0 3 First solve Lyb, and then solve Uz = y.
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
One mole of an ideal gas with CP = (7/2) R and CV = (5/2) R expands from P1 = 8 bar and T1 = 600 K to P2 = 1 bar by each of the following paths: (a) Constant volume: (b) Constant temperature; (c)...
-
A mass of 140(lbm) of 15-wt-% solution of H2SO4 in water at 160(F) is mixed atmospheric pressure with 230(lbm) of 80-wt-% H2SO4 at 100(F). During the process heat in the amount of 20,000(Btu) is...
-
If the heat of mixing at temperature to is A Ho and if the heat of mixing of the same solution at temperature t is AH, show that the two heats of mixing are related by: where Cp is the heat-capacity...
-
What is a computer service center? Are the auditors of a client that uses a service center concerned about the controls applied at the organization? Explain.
-
What is a dual-purpose test?
-
If you were preparing a credit sales system flowchart, what document would you show as: a. The source for posting debits to a customers account in the accounts receivable ledger? b. Authorization to...

Study smarter with the SolutionInn App


