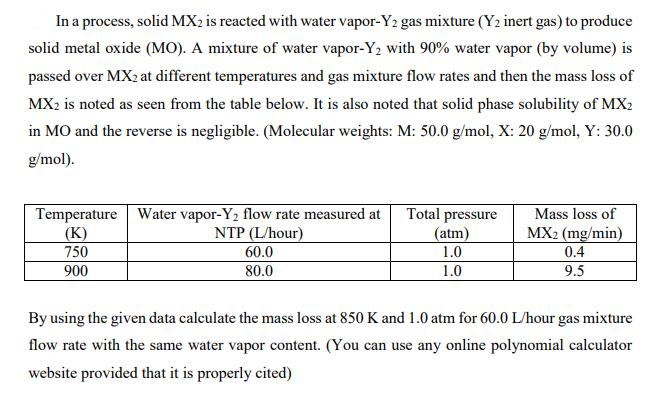
In a process, solid MX2 is reacted with water vapor-Y2 gas mixture (Y2 inert gas) to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In a process, solid MX2 is reacted with water vapor-Y2 gas mixture (Y2 inert gas) to produce solid metal oxide (MO). A mixture of water vapor-Y2 with 90% water vapor (by volume) is passed over MX2 at different temperatures and gas mixture flow rates and then the mass loss of MX2 is noted as seen from the table below. It is also noted that solid phase solubility of MX2 in MO and the reverse is negligible. (Molecular weights: M: 50.0 g/mol, X: 20 g/mol, Y: 30.0 g/mol). Temperature Water vapor-Y2 flow rate measured at (K) 750 900 Total pressure (atm) Mass loss of MX2 (mg/min) 0.4 9.5 NTP (L/hour) 60.0 1.0 80.0 1.0 By using the given data calculate the mass loss at 850 K and 1.0 atm for 60.0 L/hour gas mixture flow rate with the same water vapor content. (You can use any online polynomial calculator website provided that it is properly cited) In a process, solid MX2 is reacted with water vapor-Y2 gas mixture (Y2 inert gas) to produce solid metal oxide (MO). A mixture of water vapor-Y2 with 90% water vapor (by volume) is passed over MX2 at different temperatures and gas mixture flow rates and then the mass loss of MX2 is noted as seen from the table below. It is also noted that solid phase solubility of MX2 in MO and the reverse is negligible. (Molecular weights: M: 50.0 g/mol, X: 20 g/mol, Y: 30.0 g/mol). Temperature Water vapor-Y2 flow rate measured at (K) 750 900 Total pressure (atm) Mass loss of MX2 (mg/min) 0.4 9.5 NTP (L/hour) 60.0 1.0 80.0 1.0 By using the given data calculate the mass loss at 850 K and 1.0 atm for 60.0 L/hour gas mixture flow rate with the same water vapor content. (You can use any online polynomial calculator website provided that it is properly cited)
Expert Answer:
Related Book For 

Discovering Advanced Algebra An Investigative Approach
ISBN: 978-1559539845
1st edition
Authors: Jerald Murdock, Ellen Kamischke, Eric Kamischke
Posted Date:
Students also viewed these chemistry questions
-
You can use any angular measureradians, degrees, or revolutionsin some of the equations in Chapter 9, but you can use only radian measure in others. Identify those for which using radians is...
-
You will create a financial portfolio that will track and analyze its performance throughout the course. You need to analyze the market you choose over the length of the course. What economic factors...
-
You can use any remote access method or combination of remote access schemes. You must demonstrate understanding of the principles of remote access connections and technologies and show that they...
-
You measure 30 textbooks' weights, and find they have a mean weight of 72 ounces. Assume the population standard deviation is 4 ounces. Based on this, construct a 90% confidence interval for the true...
-
Order the following oxidizing agents by increasing strength under standard-state conditions: O2(g); MnO4 (aq); NO3 (aq) (in acidic solution).
-
True and False. All current liabilities should be excluded while computing capital employed.
-
Refer to the information in Problem P11-43. a. Assume the net realizable values of the joint products are as follows: Allocate the joint cost incurred in March on the basis of net realizable value....
-
The following transactions occurred during February 2012, for Soul Art Gift Shop, Inc.: Feb 3 Purchased $2,700 of inventory on account under terms of 4/10, n/eom (end of month) and FOB shipping...
-
You are an analyst with an investment firm. A California developer is offering an investment that may return the following amounts at the end of the next eight years: Year 1 : $ 1 0 0 , 0 0 0 Year 2...
-
Grants Graphics has a December 31 year end. Grants Graphics records adjusting entries on an annual basis. The following information is available. 1. At the end of the year, the unadjusted balance in...
-
Determine the maturity date and compute Interest for each note. Contract Date Interest Note 1. 2. Principal $17,000 22,000 15,000 Period of Nete (Tern) 60 days 90 days 45 days Rate March B 6% May 22...
-
Contrast audit objectives and audit steps. Give an example of each in terms of a purchasing function.
-
How much of a "normal" population will fall within one standard deviation? two standard deviations? three standard deviations?
-
In using statistical sampling for testing audit populations, rejecting a satisfactory population as unsatisfactory is an silphia error, whereas accepting an unsatisfactory population as satisfactory...
-
As they prepare their audit programs, auditors should keep in mind: a. The budgeted audit days of the prior job. b. Their vacation schedule. c. Their current budget. d. The final audit report. e. The...
-
What two factors does the term measurement, as used in the text, imply?
-
Amounts are in thousands of dollars ( except number of shares and price per share ) : Kiwi Fruit Company Balance Sheet Cash and equivalents $ 5 7 0 Operating assets 6 5 0 Property, plant, and...
-
Why is the national security argument for tariffs questionable?
-
The graph at right is the image of y = 1/x after a transformation. a. Write an equation for each asymptote. b. What translations are involved in transforming y = 1/x to its image? c. The point (4, -...
-
Solve. a. log 35 + log 7 = log x b. log 500 - log 25 = log x c. log (1/8 = x log8 d. 15(9.4)x = 37000 e. f. log6 342 = 2x
-
Evaluate each expression without using your calculator. a. 10( / 3(7( b. 7( / 4(3( c. 15( / 13(2( d. 7( / 7(0(
-
On May 1 of the current year, Marsha Huerta and John Ward form a partnership. The partnership assumes the assets and liabilities of the two partners existing businesses. Partners share equally in all...
-
Partnerships have operating expenses each month. Advertising, depreciation, and rent expense are examples of those operating expenses. Controlling these expenses affects the net income of the...
-
Partner A contracted with a vendor to buy a computer for the partnership. Partner A did not discuss the transac- tion with Partner B or get Partner B's approval. Partner B refused to approve payment...

Study smarter with the SolutionInn App



