Above is a graph of the pressure as a function of the volume that the gas occupies.
Fantastic news! We've Found the answer you've been seeking!
Question:

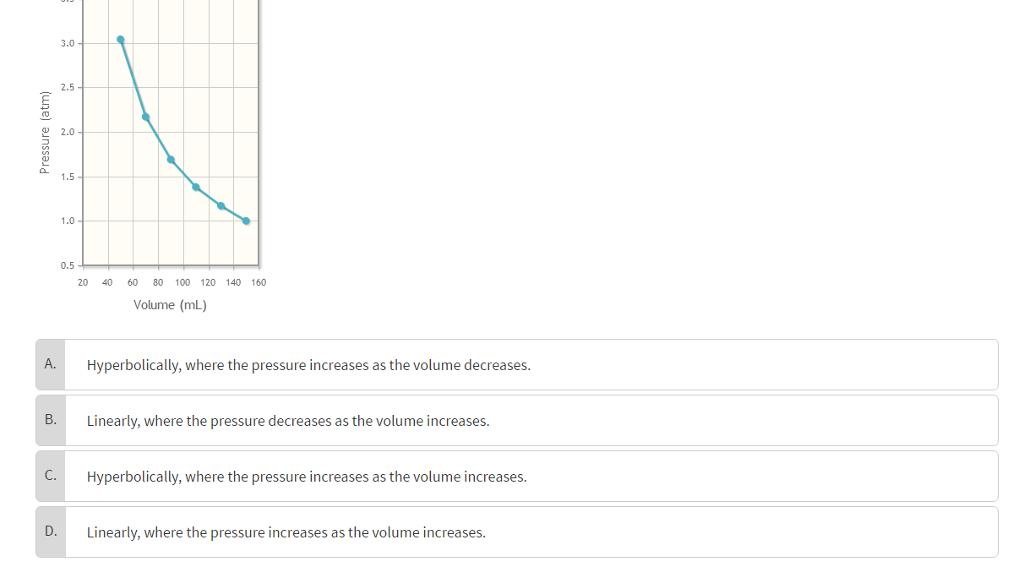
Above is a graph of the pressure as a function of the volume that the gas occupies. How does the data appear to be related?
Transcribed Image Text:
In a real gas, the molecules take up finite space and can interact with each other. How would you expect a real gas to behave differently than an ideal gas? A. In a real gas, the mass and volume can deviate from the ideal gas behavior but the pressure is unaffected. B. In a real gas, the pressure and mass can deviate from the ideal gas behavior but the volume is unaffected. C. In a real gas, the pressure and volume can deviate from the ideal gas behavior but the mass is unaffected. D. An ideal gas is identical to a real gas. In a real gas, the molecules take up finite space and can interact with each other. How would you expect a real gas to behave differently than an ideal gas? A. In a real gas, the mass and volume can deviate from the ideal gas behavior but the pressure is unaffected. B. In a real gas, the pressure and mass can deviate from the ideal gas behavior but the volume is unaffected. C. In a real gas, the pressure and volume can deviate from the ideal gas behavior but the mass is unaffected. D. An ideal gas is identical to a real gas.
Expert Answer:
Answer rating: 100% (QA)
The answer to the first question is Option C in a real gas the pressure and volume ... View the full answer

Related Book For 

International Economics Theory and Policy
ISBN: 978-0273754206
9th Edition
Authors: Paul R. Krugman, Maurice Obstfeld, Marc J. Melitz
Posted Date:
Students also viewed these accounting questions
-
Other things being equal, how would you expect each of the following events to affect the size distribution of after-tax income? a. An increase in unemployment b. Rapid population growth in an...
-
Other things equal, how would you expect the following shifts to affect a currencys real exchange rate against foreign currencies? a. The overall level of spending doesnt change, but domestic...
-
How would you expect antitrust authorities to react to? a. A proposed merger of Ford and General Motors. b. Evidence of secret meetings by contractors to rig bids for highway construction projects....
-
What is the payback period? Name and discuss three possible reasons that the payback period is used to help make capital investment decisions.
-
In a cable TV program concerning the risk of travel accidents, it was stated that the chance of a fatal airplane crash was 1 in 11 million. An explanation of this risk was that you could fly daily...
-
Turner Corporation uses the calendar year as its tax year. It purchases and places into service \($2.27\) million of property during 2023 to use in its business: What is Turners total depreciation...
-
The following contingency table presents observed frequencies. Compute the expected frequencies. 1 2 3 A 13 8 27 B 18 21 35 19 13 15 D 20 17 27
-
Answer the questions below for assets A to D shown in the table. Asset Beta A ...... 0.50 B ...... 1.60 C ...... 0.20 D ...... 0.90 a. What impact would a 10% increase in the market return be...
-
Discuss income replacement versus needs analysis approaches to estimating family insurance needs. 2. Discuss the issues surrounding key employee insurance. 3. Discuss the adverse tax consequences of...
-
I need help on this read from bottom to top. Image transcription text 7"} The second ?nancial statement to prepare is the statement of retained earnings. To determine the ending balance of...
-
Which type of decision making approach believes that those who are affected by a decision should be invited to participate in the decision making?
-
Which is one of the lines output by this code? A. [] B. [8] C. [9] D. [10] E. The code does not compile. 10: var list = new ArrayList (); 11: list.add (10); 12: List.add(9); 13: list.add(8); 14: 15...
-
Which are considered part of a service? A. Classes referenced by the implementation, but not the interface B. Classes referenced by the interface, but not the implementation C. Classes referenced by...
-
Which of the following is true of the following module declaration? A. The first line that fails to compile is line 1. B. The first line that fails to compile is line 2. C. The first line that fails...
-
Which of the following produces different output than the others? A. Stream.of ("eeny", "meeny", "miny", "moe") .collect (partitioningBy (x -> x.charAt(0) .get(false) B. C. .stream ( ) .collect...
-
How many lines does this code output? A. One. B. Two. C. Three. D. None. The code does not compile. E. None. The code throws an exception at runtime. 1: import java.util.*; 2: 3: 4: 5: 6: 7: 8: 9:...
-
Discuss the First Amendment defense to off-label promotion claims. Do you agree that companies should be permitted to share truthful, non-misleading information with customers?Why? How, if at all,...
-
Find the center of mass of a thin triangular plate bounded by the y-axis and the lines y = x and y = 2 - x if (x, y) = 6x + 3y + 3.
-
What were some of the reasons for the decline in the import-substituting industrialization strategy in favor of a strategy that promotes open trade?
-
Give two examples of products that are traded on international markets for which there are dynamic increasing returns. In each of your examples, show how innovation and learning-by-doing are...
-
If governments make trade policies based on national economic welfare, is the problem of trade warfare still represented by a Prisoners dilemma game as in Table? What is the equilibrium solution to...
-
What is thermodynamic diagram? How can it be categorized? What is its importance? How is the thermodynamic diagram constructed?
-
Show that (a) \(\left(\frac{\partial H}{\partial P} ight)_{T}=\left[\frac{\partial(V / T)}{\partial(1 / T)} ight]_{P}\) (b) \(\frac{\partial}{\partial T}\left(\frac{\Delta G}{T} ight)=-\frac{\Delta...
-
Estimate the fugacity of iso-butane at 15 atm and 87C using the compressibility factor correlation. BP Z=1+- > given that the second virial coefficient, B=-4.28 10 4 m/mol. RT

Study smarter with the SolutionInn App


