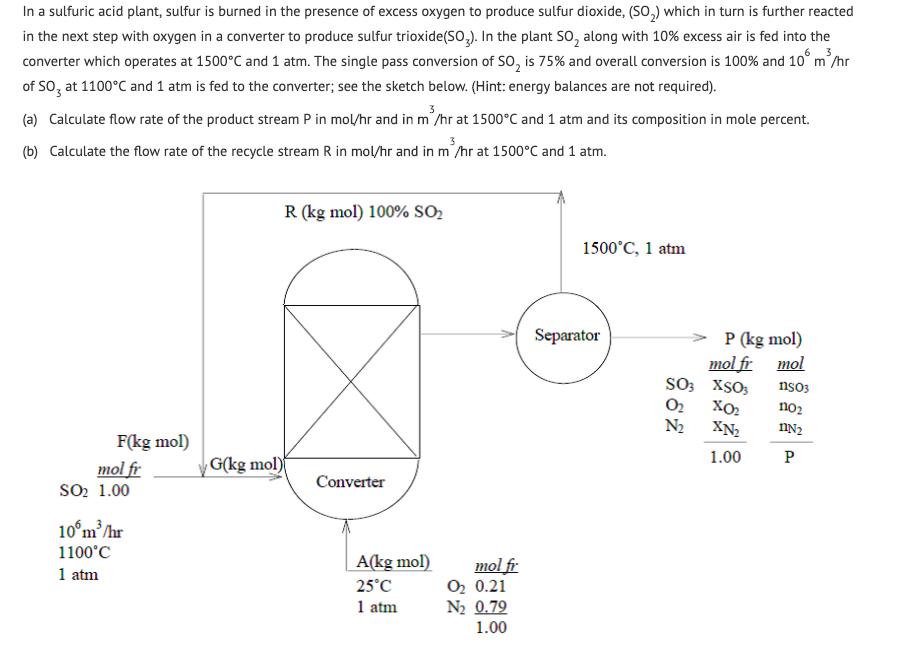
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce sulfur trioxide(SO,). In the plant SO, along with 10% excess air is fed into the converter which operates at 1500°C and 1 atm. The single pass conversion of SO, is 75% and overall conversion is 100% and 10° m'/hr of SO, at 1100°C and 1 atm is fed to the converter; see the sketch below. (Hint: energy balances are not required). (a) Calculate flow rate of the product stream P in mol/hr and in m'/hr at 1500°C and 1 atm and its composition in mole percent. (b) Calculate the flow rate of the recycle stream R in mol/hr and in m'/hr at 1500°C and 1 atm. R (kg mol) 100% SO2 1500°C, 1 atm Separator P (kg mol) mol fr mol SO3 XSO3 O2 N2 nso3 no2 XN2 F(kg mol) 1.00 P G(kg mol) mol fr Soo 1.00 Converter 10°m/hr 1100°C A(kg mol) mol fr O2 0.21 N2 0.79 1.00 1 atm 25°C 1 atm In a sulfuric acid plant, sulfur is burned in the presence of excess oxygen to produce sulfur dioxide, (SO,) which in turn is further reacted in the next step with oxygen in a converter to produce sulfur trioxide(SO,). In the plant SO, along with 10% excess air is fed into the converter which operates at 1500°C and 1 atm. The single pass conversion of SO, is 75% and overall conversion is 100% and 10° m'/hr of SO, at 1100°C and 1 atm is fed to the converter; see the sketch below. (Hint: energy balances are not required). (a) Calculate flow rate of the product stream P in mol/hr and in m'/hr at 1500°C and 1 atm and its composition in mole percent. (b) Calculate the flow rate of the recycle stream R in mol/hr and in m'/hr at 1500°C and 1 atm. R (kg mol) 100% SO2 1500°C, 1 atm Separator P (kg mol) mol fr mol SO3 XSO3 O2 N2 nso3 no2 XN2 F(kg mol) 1.00 P G(kg mol) mol fr Soo 1.00 Converter 10°m/hr 1100°C A(kg mol) mol fr O2 0.21 N2 0.79 1.00 1 atm 25°C 1 atm
Expert Answer:
Answer rating: 100% (QA)
Solution By considering material balance around the SO 2 feeding point F R G Given F 10 6 m 3 hr By ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these finance questions
-
When methanol, CH3OH,CH3OH, is burned in the presence of oxygen gas, O2,O2, a large amount of heat energy is released. For this reason, it is often used as a fuel in high performance racing cars. The...
-
A sulfuric acid plant produces a considerable amount of heat. This heat is used to generate electricity, which helps reduce operating costs. The synthesis of H2SO4 consists of three main chemical...
-
Propane fuel (C3H8) is burned in the presence of air. Assuming that the combustion is theoretical-that is, only nitrogen (N2), water vapor (H2O), and carbon dioxide (CO2) are present in the...
-
If there is a decrease in the demand for Canadian dollars relative to U.S. dollars, a. The price and quantity of Canadian dollars traded will fall. b. The price and quantity of Canadian dollars will...
-
What types of entities issue municipal bonds? What significant feature of municipal bonds attracts investors? Given this feature, who should consider investing in municipal bonds?
-
Graph the polynomials P(x) = 3x5 5x3 + 2x and Q(x) = 3x5 on the same screen, first using the viewing rectangle [ 2, 2] by [ 2, 2] and then changing to [ 10, 10] by [ 10,000, 10,000]. What do you...
-
A long, thin metal plate is hung vertically after a heat treating process. The plate, of width \(W=0.2 \mathrm{~m}\), vertical dimension of \(3 \mathrm{~m}\), and an initial temperature of...
-
Explain how each of the following events or series of events and the related adjusting entry will affect the amount of net income and the amount of cash flow from operating activities reported on the...
-
We will be discussing topics to make sure that you've chosen a manageable one for this assignment, but here are a few sample problems you might want to consider writing about (choose ONLY ONE): not...
-
Your investment bank has an investment of $100 million in the stock of the Swiss Roll Corporation and a short position in the stock of the Frankfurter Sausage Company. Here is the recent price...
-
In a study, subjects are randomly assigned to one of three groups: control, experimental A, or experimental B. After treatment, achievement test scores for the three groups are compared. The...
-
A 56-year-old woman returns from a trip to America in May. During her stay, she became unwell with a high fever, headache and muscle pains. She consulted a local doctor who noticed a rash on the...
-
Aditi is a compliance auditor. She is using a layered audit approach to examine a company's financial system. That system spans multiple domains of a typical infrastructure and third-party providers....
-
What are the key tools for holding public sector entities to account for the use of taxpayer dollars?
-
A manager believes that the shelf life of apple juice is normally distributed. A sample of 30 containers of juice was taken and the shelf life was recorded. You are given the results below. The...
-
Determine the restrictions on the variable in the following function. f(x) = 2 x-4
-
1 0.5 points Question at position 1 In 2023, Bob sold a piece of property (AB = $93971) for $1024512. The purchase price is to be paid as follows: down payment of $213847 (no interest) in 2023 and...
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
Consider the manometer in Fig. P1-78. If the specific weight of fluid A is 100 kN/m3, what is the absolute pressure, in kPa, indicated by the manometer when the local atmospheric pressure is 90 kPa?
-
An air standard cycle with constant specific heats is executed in a closed piston-cylinder system and is composed of the following three processes: 1-2 Constant volume heat addition 2-3 lsentropic...
-
Reconsider Prob. 5 - 74. Using EES (or other) software, investigate the effect of the mass flow rate of the cold stream of R-134a on the temperature and the quality of the exit stream. Let the ratio...
-
A container of an organic liquid a $25^{\circ} \mathrm{C}$ has a mass of $1100 \mathrm{~kg}$ and a volume of $0.94 \mathrm{~m}^{3}$. Find the liquid's weight, its density, and its specific gravity.
-
What is the pressure one mile $(1.6 \mathrm{~km})$ beneath the ocean's surface?
-
At what pressure will water boil at $65^{\circ} \mathrm{C}$ ? The vapor pressure of water can be determined from the Antoine equation: \[\frac{P(P a)}{1 \times...

Study smarter with the SolutionInn App


