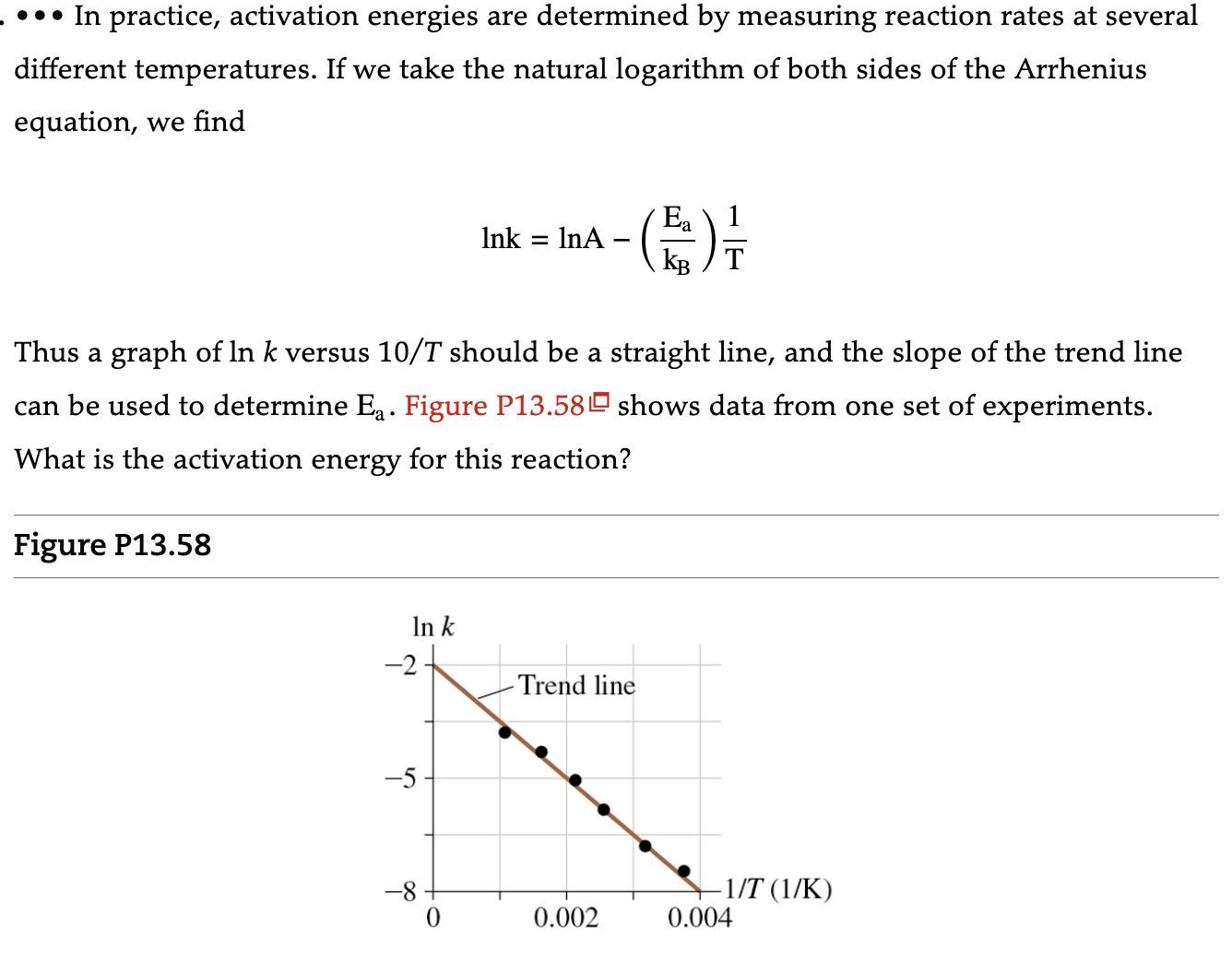
In practice, activation energies are determined by measuring reaction rates at several different temperatures. If we...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
••• In practice, activation energies are determined by measuring reaction rates at several different temperatures. If we take the natural logarithm of both sides of the Arrhenius equation, we find Figure P13.58 Thus a graph of ln k versus 10/T should be a straight line, and the slope of the trend line can be used to determine E₁. Figure P13.58 shows data from one set of experiments. What is the activation energy for this reaction? In k -2. -5 Ink InA -8 -Trend line Ea KB T 0.002 −1/T (1/K) 0.004 ••• In practice, activation energies are determined by measuring reaction rates at several different temperatures. If we take the natural logarithm of both sides of the Arrhenius equation, we find Figure P13.58 Thus a graph of ln k versus 10/T should be a straight line, and the slope of the trend line can be used to determine E₁. Figure P13.58 shows data from one set of experiments. What is the activation energy for this reaction? In k -2. -5 Ink InA -8 -Trend line Ea KB T 0.002 −1/T (1/K) 0.004
Expert Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Posted Date:
Students also viewed these physics questions
-
At June 1 , 2 0 2 0 , Bramble company had an accounts receivable balance of $ 1 1 , 2 0 0 . During the month, the company performed credit services of $ 2 8 , 0 0 0 and collected accounts receivable...
-
The Arrhenius equation can be reformulated in a way that permits the experimental determination of activation energies. For this purpose, we take the natural logarithm of both sides and convert into...
-
If P dollars are invested at a yearly interest rate r per year, compounded m times per year for t years, then the compound amount A is When m , then the interest is compounded continuously. Use the...
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
Give some everyday examples of situations in which friction is beneficial.
-
JetBlue Airways Corporation (JBLU) is a passenger airline with flights to destinations throughout the United States, the Caribbean, and Latin America. Costco (COST) operates membership retail...
-
Robert Ryan practiced accounting with a partnership for five years. Recently he opened his own accounting firm, which he operates as a professional corporation. The name of the new entity is Robert...
-
A foundation promises to donate $1 million to a local public broad casting station (a not-for-prot organization) in one year. When, and in what amount, should the station recognize revenue? The...
-
Question 3 (a) Ann Lee is the portfolio manager of a Singapore based equity fund. She is analysing the value of Union Chemical Bhd which manufactures fertilizers and food additives. The company's...
-
The management of Zigby Manufacturing prepared the following balance sheet for March 31. Cash Accounts receivable Raw materials inventory Assets ZIGBY MANUFACTURING Balance Sheet March 31 Liabilities...
-
4. (4 points total) A 27-year-old woman has been consuming a low-fat, high- carbohydrate diet for the past week. Assume that this time of chronic high carbohydrate consumption promotes upregulation...
-
Firm-specific knowledge -including the capacity to exploit it -is an essential feature of competitive success. True False
-
7. Determine Vo1 and Vo2 for the networks given below. +12 Vo Si Si +10 V 8. Determine I and V, for the networks given below. 1k2 4.7 k Ge Ge Si 9. Determine Vo1, Vo2, and I. +16 V Si 4,7 k 12 V Si...
-
XYZ Pty Ltd is a new business and provides you with their first-year financial statements for tax calculation. XYZ Pty Ltd Profit & Loss Statement For the year ended 30 June 2022 Sales Less: Expenses...
-
2. The reaction is given below at constant temperature in a continuous stirred tank reactor: k A+B+C+D -1A = (6h-)CA [kmol/mh] The concentrations of A and B in the feed are 0.5 and 1 kmol/m,...
-
What did you think about the two contrasting patterns in this chapter? Do you or someone you know fit one or the other of these patterns? Which pattern do you think is more likely to emphasize...
-
Cost-Utility Analysis of Best Supportive Care Versus Oncoplatin And Oncotaxel in the Treatment of Recurrent Metastatic Breast Cancer BACKGROUND : For patients diagnosed with recurrent metastatic...
-
What are technical skills At what level are they most important and why?
-
Formulate a plausible mechanism for the hydration of ethyne in the presence of mercuric chloride. MR 3 H IH 1H 1 H (CH3)4Si 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 300-MHz 'H NMR...
-
Based on the energy differences for the various conformations of ethane, propane, and butane in Figures 2-10, 2-11, and 2-13, determine the following: (a) The energy associated with a single...
-
The benzene rings of many compounds in nature are prepared by a biosynthetic pathway similar to that operating in fatty acid synthesis. Acetyl units are coupled, but the ketone functions are not...
-
State whether the data are symmetrical, skewed to the left, or skewed to the right. 1; 1; 1; 2; 2; 2; 2; 3; 3; 3; 3; 3; 3; 3; 3; 4; 4; 4; 5; 5
-
What is the average rate of return for the values that follow? 4.2, 4.3, 4.5
-
When the data are skewed left, what is the typical relationship between the mean and median?

Study smarter with the SolutionInn App



