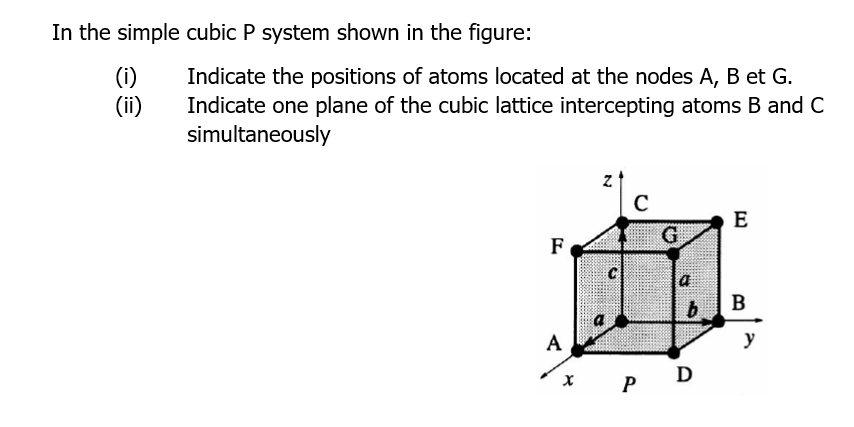
In the simple cubic P system shown in the figure: (i) (ii) Indicate the positions of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In the simple cubic P system shown in the figure: (i) (ii) Indicate the positions of atoms located at the nodes A, B et G. Indicate one plane of the cubic lattice intercepting atoms B and C simultaneously C E G F b. B A P In the simple cubic P system shown in the figure: (i) (ii) Indicate the positions of atoms located at the nodes A, B et G. Indicate one plane of the cubic lattice intercepting atoms B and C simultaneously C E G F b. B A P
Expert Answer:
Answer rating: 100% (QA)
i As we know a simple cubic structure of a unit cell is onein w... View the full answer

Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Posted Date:
Students also viewed these physics questions
-
Dangerfield Pty Ltd would like to determine the variable rate for electricity per machine hour in order to estimate the electricity costs for the months of May and June. Information for the four...
-
In Drosophila, genes a and b are located at positions 22.0 and 42.0 on chromosome 2, and genes c and d are located at positions 10.0 and 25.0 on chromosome 3. A fly homozygous for the wild type...
-
In the model shown here, C atoms are black and H atoms are light blue. a. Write the molecular formula. b. Write the condensed structural formula. c. Write the IUPAC name
-
Ethanol reacts with sodium and forms two products which are Options: 1) Sodium ethanoate and hydrogen 2) Sodium ethanoate and oxygen 3) Sodium ethoxide and hydrogen 4) Sodium ethoxide and oxygen
-
Matts gross pay had been $68 000 per year, when he received an increase of $6000 per year. (a) What was the annual percent increase in Matts pay before federal income taxes? (b) What was the annual...
-
Brite Companys ending inventory values and its cost of goods sold (COGS) amounts for Q1Q4 are listed below. Required a. Calculate Brites inventory turnover ratio for Q2, Q3, and Q4. b. Calculate...
-
Determine whether each experiment is a binomial experiment. If it is, specify the values of n, p, and q, and list the possible values of the random variable x. If it is not, explain why. 1. A certain...
-
On January 10, 2011, Delta Corporation acquired 12,000 shares of the outstanding common stock of Kennedy Company for $600,000. At the time of purchase, Kennedy Company had outstanding 48,000 shares...
-
1. How should a company whose product is highly demanded and highly specialized grow quickly but still source responsibly? How far down the supply chain is the company responsible? How should it...
-
P6. (6 pts) Consider a generator of a CRC scheme 1010011 (i.e., x6+x++x+1). The message is 00110011 10001010. Answer the following questions. a. Compute the CRC bits for this packet (ignoring all...
-
7 -2 1 Let A = -2 10-2 b 1-2 7 =[ 6 12 18 Compute, by hand, the eigenvalues and eigenvectors of A, and use them to solve Ax = b for x (use the "spectral method").
-
Clayton and Margie Gulledge owned a house at 532 Somerset Place, N.W. (the Somerset property) as tenants by the entirety. They had three children: Bernis Gulledge, Johnsie Walker, and Marion Watkins....
-
Mitchell, Nelson, Olsen, and Parker, experts in manufacturing baubles, each owned fifteen of one hundred authorized shares of Baubles, Inc., a corporation of State X that does not permit cumulative...
-
Leonard Wolfe was killed in an automobile accident while driving his Toyota Camry. The car was rendered a total loss, and Wolfes insurance carrier paid his estate \($17,550\) for damage to the...
-
In a football game, a 90 kg receiver leaps straight up in the air to catch the 0.42 kg ball the quarterback threw to him at a vigorous 21 m/s, catching the ball at the highest point in his jump....
-
On March 10, John Carver executed his will, which was witnessed by William Hobson and Sam Witt. By his will, Carver devised his farm, Stonecrest, to his nephew, Roy White. The residue of his estate...
-
Use project costs and benefits to complete the financial feasibility of the SFO SAFE proposal. Background : Read San Francisco International Airport and Quantum Secure's SAFE for Aviation System....
-
Can partitioned join be used for r r.A s? Explain your answer
-
Figure displays a closed cycle for a gas. From c to b, 40 J is transferred from the gas as heat. From b to a, 130 J is transferred from the gas as heat, and the magnitude of the work done by the gas...
-
An alternating emf source with a variable frequency f d is connected in series with an 80.0 resistor and a 40.0 mH inductor. The emf amplitude is 6.00 V. (a) Draw a phasor diagram for phasor V R (the...
-
The potential energy of a diatomic molecule (a two-atom system like H2 or O) is given by where r is the separation of the two atoms of the molecule and A and B are positive constants. This potential...
-
Amherst Metal Works produces two types of metal lamps. Amherst manufactures 20,000 basic lamps and 5,000 designer lamps. Its simple costing system uses a single indirect-cost pool and allocates costs...
-
Amherst Metal Works produces two types of metal lamps. Amherst manufactures 20,000 basic lamps and 5,000 designer lamps. Its activity-based costing system uses two indirect-cost pools. One cost pool...
-
How do managers refine a costing system?

Study smarter with the SolutionInn App


