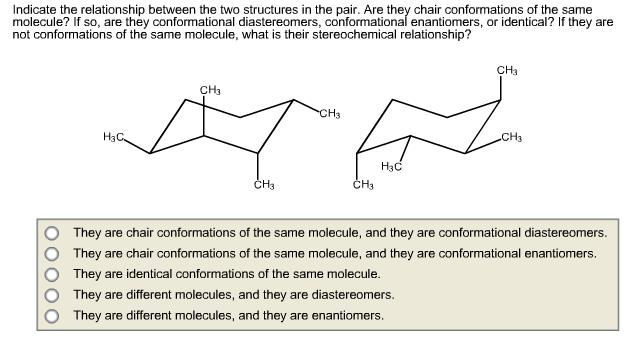
Indicate the relationship between the two structures in the pair. Are they chair conformations of the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Indicate the relationship between the two structures in the pair. Are they chair conformations of the same molecule? If so, are they conformational diastereomers, conformational enantiomers, or identical? If they are not conformations of the same molecule, what is their stereochemical relationship? CHa CH3 CH3 H3C CH3 H3C CH3 They are chair conformations of the same molecule, and they are conformational diastereomers. They are chair conformations of the same molecule, and they are conformational enantiomers, They are identical conformations of the same molecule. They are different molecules, and they are diastereomers. They are different molecules, and they are enantiomers. O000 Indicate the relationship between the two structures in the pair. Are they chair conformations of the same molecule? If so, are they conformational diastereomers, conformational enantiomers, or identical? If they are not conformations of the same molecule, what is their stereochemical relationship? CHa CH3 CH3 H3C CH3 H3C CH3 They are chair conformations of the same molecule, and they are conformational diastereomers. They are chair conformations of the same molecule, and they are conformational enantiomers, They are identical conformations of the same molecule. They are different molecules, and they are diastereomers. They are different molecules, and they are enantiomers. O000
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is meant when the relationship between the two variables is called positive? Negative?
-
Determine the relationship between the two structures below. Are they resonance structures or are they constitutional isomers?
-
For each pair, give the relationship between the two compounds. Making models will be helpful. (a) (2R, 3S)-2, 3-dibromohexane and (2S, 3R)-2, 3-dibromohexane (b) (2R, 3S)-2, 3-dibromohexane and (2R,...
-
What is the output produced from the following statements? System.out.println("name\tage\theight"); System.out.println("Archie\t17\t5'9\""); System.out.println("Betty\t17\t5'6\"");...
-
Three dice are rolled. Find the probability of getting a. Triples b. A sum of
-
Not everything that is sensed is perceived. Do you think there could ever be a case where something could be perceived without being sensed?
-
Swift Company was organized on March 1 of the current year. After five months of start-up losses, management had expected to earn a profit during August. Management was disappointed, however, when...
-
Marie Hardys will has the following provisions: I leave the cash balance deposited in the First National Bank (up to a total of $50,000) to Jack Abrams. I leave $18,000 cash to Suzanne Benton. I...
-
What values of y and z make ACDE AFHG? F Z+6 H y+37 C E 15 y = D G 41
-
Ramada Company produces one golf cart model. A partially complete table of company costs follows: Required: 1. Complete the table. 2. Ramada sells its carts for $1,200 each. Prepare a contribution...
-
McK Nursery, Incorporated, reports the following account balances on December 31, 2024: cash, $31,000; accounts receivable. $26,000; Inventory, $40,000; equipment (net), $82,500; operating leases,...
-
You log into a closed group on a social networking site and read a posting from a fellow nursing student. The posting describes in detail the complex care the student gave to a 9 y/o patient in a...
-
How do advanced computational fluid dynamics (CFD) simulations aid in optimizing the design of hydraulic structures such as dams and spillways to ensure efficient water management and mitigate...
-
Case Study 2b - Starting Design Thinking for team collaboration In preparation for your leadership role with a work team to innovate, Carol wants to review your understanding of the Design Thinking...
-
Review the material from the lesson, text book, and video on how the unemployment rate is calculated by the Bureau of Labor Statistics (BLS). Question To Answer: Did I tell l you that I am going to...
-
Consider the following power generation cycle. The working steam is heated inside a boiler by nuclear fission, which provides 27.8 MW of heat transfer at a steady 1900 K. The turbine generates 22 MW...
-
*25.7 A company (which is not a member of a group) has the following results for the year to 31 March 2022: Adjusted trading profit, after deduction of capital allowances Bank deposit interest (a/c...
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
The synthesis of the alkylbenzenesulfonate detergent shown in Figure 25-6 begins with the partial polymerization of propylene to give a pentamer. Show how aromatic substitution reactions can convert...
-
What characteristics of the methyl benzoate spectrum rule out an aldehyde or carboxylic acid functional group giving the absorption at 1723 cm-1?
-
Account for the peaks at m/z 87, 111, and 126 in the mass spectrum of 2,6-dimethylheptan-4-ol. 100 OH 80 2,6-dimethylheptan-4-o1 E 40 83 111 126 20 10 20- 30 40 50 60mm 80 m90ml00 110T20 130T 140...
-
In the previous question, the Australian interest rates increased from 6 to 11 percent. According to the IFE, what is the underlying factor that would cause such a change? Give an explanation based...
-
Forecasting the Future Spot Rate Based on IFE. Assume that the spot exchange rate of the Singapore dollar is $.70. The 1-year interest rate is 11 percent in the United States and 7 percent in...
-
Assume that the nominal interest rate in Mexico is 48 percent and the interest rate in the United States is 8 percent for 1-year securities that are free from default risk. What does the IFE suggest...

Study smarter with the SolutionInn App


