Indicate whether the compounds in each set are constitutional isomers. Indicate whether the compounds in each...
Fantastic news! We've Found the answer you've been seeking!
Question:
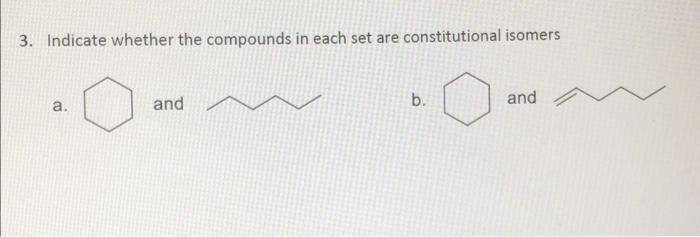
Transcribed Image Text:
Indicate whether the compounds in each set are constitutional isomers. Indicate whether the compounds in each set are constitutional isomers.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Indicate which of the compounds in each pair will give a higher substitution/elimination ratio when it reacts with isopropyl bromide: a. Ethoxide ion or tert-butoxide ion b. -OCN or -SCN c. CI- or...
-
Rank the compounds in each set in order of increasing acid strength. (a) CH3CH2COOH CH3CHBrCOOH CH3CBr2COOH (b) CH3CH2CH2CHBrCOOH CH3CH2CHBrCH2COOH CH3CHBrCH2CH2COOH (c) CH CHCOOH CH,CHCOOH CH,CH...
-
The compounds in each part below have the same (or similar) molecular weights. Which compound in each part would you expect to have the higher boiling point? Explain your answers. (a) (b) (c) OH or...
-
Jake, a single taxpayer, has $100,000 of ordinary income, a $10,000 net short-term capital loss, and $7,000 of qualified dividends. What is the result?
-
What is the difference between a centralized and decentralized debt denomination for an MNC?
-
B Vaughn Corporation began operations in 2025 and reported pretax financial income of $236,000 for the year. Vaughn's tax depreciation exceeded its book depreciation by $32,000. Vaughn's tax rate for...
-
An HR consultant gives an applicant a 75% chance of getting a job after registering with his agency. If the applicant gets a job, then there is a 60% possibility that he would be satisfied and would...
-
At Cambridge Company, prepayments are debited to expense when paid and unearned revenues are credited to revenue when cash is received. During January of the current year, the following transactions...
-
1. Frank was director of research-development (R&D) for a large pharmaceutical company. The following memorable incident is related to his name: During a research project the mebers of the team...
-
The following data relate to the operations of Shilow Company, a wholesale distributor of consumer goods: Current assets as of March 31: Cash $ 8,000 Accounts receivable 20,000 Inventory 36,000...
-
Hybe Company distributes rubber tires, automotive oils and lubricants to many transport companies and retailers all over the region. The company offers a contract term of 2/10, n/30. Currently, sixty...
-
A car suffers from 12% annual depreciation. If the initial value is $40,000, find the value after 4 years. Hint: Depreciation means inflation (losing money). Evaluate (2-)-. Simplify (2x). Show that...
-
Let G = (V, E) be an undirected graph. For any subset A, BCV, let C(A, B) denote the set of edges between A and B, i.e., C(A, B) = {e EE e connects some vertex in A and some vertex in B}. Note that,...
-
iversification at Ratcliffe Investments What's the current situation? Sports investors around the world have reaped unprecedented returns in recent years. In the National Basketball Association...
-
What percentage of your gross salary does the Consumer Financial Protection Bureau suggest your student loan payment be in order to be affordable and limit your risk of delinquency and default?
-
What marketing strategy and plan would you recommend for the new fitness tracking device startup company to generate awareness and drive sales with limited resources?
-
Donna Motors is a Japanese-owned company that produces automobiles; all of its automobiles are produced in Korean factories. In 2017, Donna Motors produced $40 million worth of automobiles in the...
-
Provide examples of a situations in which environmental disruptions affected consumer attitudes and buying behaviors.
-
Answer the following questions: a. What six cofactors act as oxidizing agents? b. What are the cofactors that donate one-carbon groups? c. What three one-carbon groups are various tetrahydrofolates...
-
a. Draw the two chair conformers for each of the stereoisomers of trans-1-tert-butyl-3 methylcyclohexane. b. For each pair, indicate which conformer is more stable.
-
In order to solve this problem, you must read the description of the Hammett Ï, Ï treatment given in Chapter 17, Problem 76. When the rate constants for the hydrolysis of several morpholine...
-
At the instant shown in Figure 32. 40 , the potential difference across the capacitor is half its maximum value and the charge on the plates is increasing. Draw the direction of the current and...
-
Construct a phasor diagram representing the current and potential difference in Figure 32. 10 at \(t=T / 4, T / 2\), and \(3 T / 4\). Data from Figure 32.10 Ve maximum, current zero Ve minimum,...
-
The research laboratory you work in is less than a block away from the transmitting antenna of a radio station that broadcasts at \(1100 \mathrm{kHz}\). You notice that every piece of equipment in...

Study smarter with the SolutionInn App



