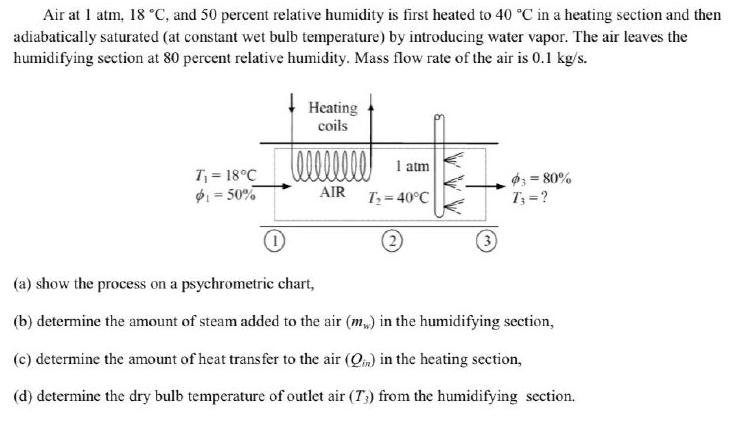
Air at I atm, 18 C, and 50 percent relative humidity is first heated to 40...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Air at I atm, 18 °C, and 50 percent relative humidity is first heated to 40 °C in a heating section and then adiabatically saturated (at constant wet bulb temperature) by introducing water vapor. The air leaves the humidifying section at 80 percent relative humidity. Mass flow rate of the air is 0.1 kg/s. Heating coils 1 atm T = 18°C 1 = 50% 3 = 80% T;=? AIR T; = 40°C (a) show the process on a psychrometric chart, (b) determine the amount of steam added to the air (m) in the humidifying section, (c) determine the amount of heat transfer to the air (Qm) in the heating section, (d) determine the dry bulb temperature of outlet air (T;) from the humidifying section. Air at I atm, 18 °C, and 50 percent relative humidity is first heated to 40 °C in a heating section and then adiabatically saturated (at constant wet bulb temperature) by introducing water vapor. The air leaves the humidifying section at 80 percent relative humidity. Mass flow rate of the air is 0.1 kg/s. Heating coils 1 atm T = 18°C 1 = 50% 3 = 80% T;=? AIR T; = 40°C (a) show the process on a psychrometric chart, (b) determine the amount of steam added to the air (m) in the humidifying section, (c) determine the amount of heat transfer to the air (Qm) in the heating section, (d) determine the dry bulb temperature of outlet air (T;) from the humidifying section.
Expert Answer:

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these electrical engineering questions
-
I need the answer as soon as possible. Halsey Corporation first issued stock on January 1, 2016. Halsey has the following stock outstanding on December 31, 2019: Preferred Stock (5 percent...
-
Assume the risk-free rate is 3% and the market return is 10%. Stock X Stock Y Stock Z Beta 0.65 0.90 -- Current price $13.50 $26.50 Correlation (X/Y) = 0.35 (X/Z) = 0 (Y/Z) = 0.55 a) Most equity...
-
It is required to design a BPF with the following specifications -0.02 < |H(ejw)l < 0.02, 0<= |w| <=2n 0.95 < |H(ejw) < 1.05, ,3n <= Iw| <= 0.7n, -0.001 < |H(ejw)| <...
-
The lifespans of gorillas in a particular zoo are normally distributed. The average gorilla lives 20.8 years; the standard deviation is 3.1 years. Use the empirical rule (68 - 95 - 99.7%) to estimate...
-
Brandons Bentwood Furniture Company designs, makes, and sells custom furniture. The cost of the special resin used to finish the furniture has been increasing over the past 12 to 18 months. For...
-
Farr Industries Inc. manufactures only one product. For the year ended December 31, the contribution margin increased by $560,000 from the planned level of $5,200,000. The president of Farr...
-
Which statement best describes which theoretical perspective nurse managers should use to motivate their employees? A. Each employee has individual needs, so no one theory applies to all employees....
-
Illustrate the effects on the accounts and financial statements of recording the following selected transactions of Bronson Leather Co.: Apr. 15. Paid the first installment of the estimated income...
-
Anne wrote a Novel. She receives a payment of 5% of the $100 retail price for every book sold. This financial year she has sold 1,000 textbooks. What amount should Anne include in her assessable...
-
John Parsons (123-45-6781) and George Smith (123-45-6782) are 70% and 30% owners, respectively, of Premium, Inc. (11-1111111), a candy company located at 1005 16th Street, Cut and Shoot, TX 77303....
-
(b) Consider the covariance matrix 9-1 =(,1) for random variables -1 4 X and X 2 (i) Give a table showing eigenvalue, percent of variance and cumulative percent for both and p (ii) Compute the...
-
A compact car with a mass of 525 kg is traveling at 23 m/sec. a. What is the momentum of the car? (12,075 kg m/sec) b. If the brakes are applied so that it slows down to 10 m/sec, what is the change...
-
Mazie Supply Company uses the percent of accounts receivable method. On December 31, it has outstanding accounts receivable of $85,000, and it estimates that 5% will be uncollectible. Prepare the...
-
Give the domain of t(x) = 11 -x in inequality notation.
-
Delta Company produces a single product. The cost of producing and selling a single unit of this product at the company s normal activity level of 9 4 , 8 0 0 units per year is: Direct materials $ 1...
-
Do rising egg prices affect demand? With the Avian flu destroying flocks all over the world as well as countless disasters wiping out chicken houses like the Muskegon chicken coop fire. However, as...
-
1. If there are eight candidates for a scholarship and we let a, b, c, d, e, f, g, and h denote that it is awarded to Ms. Adam, Mr. Bean, Miss Clark, Mrs. Daly, Mr. Earl, Ms. Fuentes, Ms. Gardener,...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Reconsider Prob. 12-12. Using EES (or other) software, verify the validity of the last Maxwell relation for refrigerant-134a at the specified state.
-
Determine the change in the enthalpy of helium, in kJ/kg, as it undergoes a change of state from 150 kPa and 20oC to 750 kPa and 380oC using the equation of state P(v - a) = RT where a = 0.01 m3/kg,...
-
Steam at 75 kPa and 8 percent quality is contained in a spring-loaded piston - cylinder device, as shown in Fig. P4 - 39, with an initial volume of 2 m3. Steam is now heated until its volume is 5 m3...
-
Solve Exercise 1 using the tableau method.
-
Redo the winery problem (3)-(4) using the tableau implementation of the simplex algorithm. Note the connection between the tableau you obtain at each step, and the corresponding system of equations...
-
(a) What would you look for in a simplex tableau in order to conclude that a problem is unbounded? (b) What would you look for in a simplex tableau in order to detect a degeneracy? (c) What...

Study smarter with the SolutionInn App


