Normal rain has a pH of 5.6 due to dissolved atmospheric carbon dioxide at a current...
Fantastic news! We've Found the answer you've been seeking!
Question:

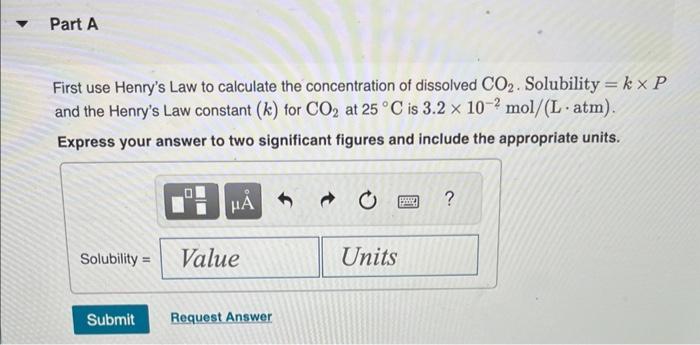
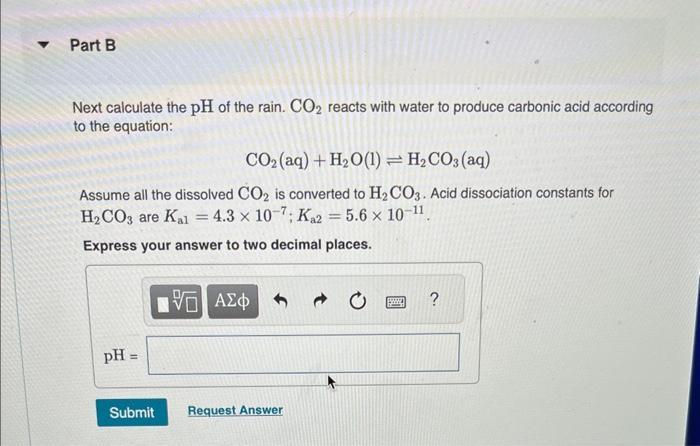
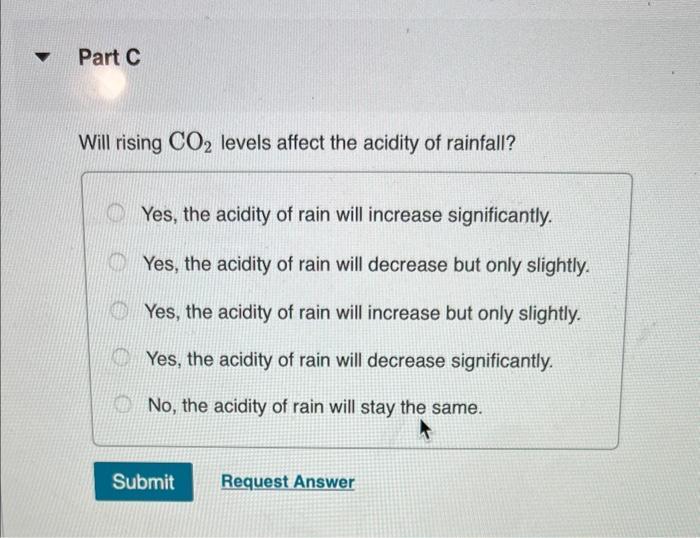
Transcribed Image Text:
Normal rain has a pH of 5.6 due to dissolved atmospheric carbon dioxide at a current level of 400 ppm. Various models predict that burning fossil fuels will increase the atmospheric CO₂ concentration to between 500 and 1000 ppm (by volume) by the year 2100. Calculate the pH of rain in a scenario where the CO₂ concentration is 550 ppm. Part A First use Henry's Law to calculate the concentration of dissolved CO₂. Solubility = kx P and the Henry's Law constant (k) for CO2 at 25 °C is 3.2 x 10-2 mol/(L atm). Express your answer to two significant figures and include the appropriate units. I Submit HA Solubility= Value Request Answer C Units P ? Part B Next calculate the pH of the rain. CO2 reacts with water to produce carbonic acid according to the equation: CO₂ (aq) + H₂O(1) = H₂CO3(aq) Assume all the dissolved CO₂ is converted to H₂CO3. Acid dissociation constants for H₂CO3 are Kal 4.3 x 10-7; K₁2 = 5.6 x 10-11. Express your answer to two decimal places. pH = Submit 15. ΑΣΦΑ Request Answer → www Part C Will rising CO₂ levels affect the acidity of rainfall? Yes, the acidity of rain will increase significantly. Yes, the acidity of rain will decrease but only slightly. Yes, the acidity of rain will increase but only slightly. Yes, the acidity of rain will decrease significantly. No, the acidity of rain will stay the same. Submit Request Answer Normal rain has a pH of 5.6 due to dissolved atmospheric carbon dioxide at a current level of 400 ppm. Various models predict that burning fossil fuels will increase the atmospheric CO₂ concentration to between 500 and 1000 ppm (by volume) by the year 2100. Calculate the pH of rain in a scenario where the CO₂ concentration is 550 ppm. Part A First use Henry's Law to calculate the concentration of dissolved CO₂. Solubility = kx P and the Henry's Law constant (k) for CO2 at 25 °C is 3.2 x 10-2 mol/(L atm). Express your answer to two significant figures and include the appropriate units. I Submit HA Solubility= Value Request Answer C Units P ? Part B Next calculate the pH of the rain. CO2 reacts with water to produce carbonic acid according to the equation: CO₂ (aq) + H₂O(1) = H₂CO3(aq) Assume all the dissolved CO₂ is converted to H₂CO3. Acid dissociation constants for H₂CO3 are Kal 4.3 x 10-7; K₁2 = 5.6 x 10-11. Express your answer to two decimal places. pH = Submit 15. ΑΣΦΑ Request Answer → www Part C Will rising CO₂ levels affect the acidity of rainfall? Yes, the acidity of rain will increase significantly. Yes, the acidity of rain will decrease but only slightly. Yes, the acidity of rain will increase but only slightly. Yes, the acidity of rain will decrease significantly. No, the acidity of rain will stay the same. Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
Part A Henrys Law allows us to figure out how much dissolved CO2 is in the rainfall Per Henrys Law solubility is equal to k P Where The concentration ... View the full answer

Related Book For 

Fundamentals of Corporate Finance
ISBN: 978-0077861629
8th edition
Authors: Richard Brealey, Stewart Myers, Alan Marcus
Posted Date:
Students also viewed these chemical engineering questions
-
Comprehensive Problem 1 - Part 1: Taxpayer information, Form 1040, Schedules 1, 2, 3 and 4, Schedule A, and Schedule B. Noah and Joan Arc's Tax Return Note: This problem is divided into three parts....
-
To live comfortably in retirement, you decide you will need to save $2 million by the time you are 65 (you are 30 years old today). You will start a new retirement savings account today and...
-
You believe you will need to have saved $500,000 by the time you retire in 40 years in order to live comfortably. You also believe that you will inherit $100,000 in 10 years. If the interest rate is...
-
The 10-year Coupon Bond has a face value of $1,000, the annual coupon rate is 5 percent (out of its face value), the yield to maturity is 10 percent. (2.a) show me the cash flows of this coupon bond,...
-
A total of 1000 adults were surveyed whether they thought getting a mortgage was harder than it was a year ago. Their responses are summarized in the relative frequency bar graph in Fig. 17. Find the...
-
In its first month of operations, Weatherall Company made three purchases of merchandise in the following sequence: (1) 300 units at $6, (2) 400 units at $7, and (3) 200 units at $8. Assuming there...
-
Six principles of internal accounting control are identified in the chapter. Listed below are specific control procedures: 1. Use of cash registers for over-the-counter cash receipts. 2. Different...
-
Cleveland Company has a stock portfolio valued at $4,000. Its cost was $3,300. If the Securities Fair Value Adjustment (Available-for-Sale) account has a debit balance of $200, prepare the journal...
-
Southwest Milling Company purchased a front-end loader to move stacks of lumber. The loader had a list price of $124,960. The seller agreed to allow a 5.00 percent discount because Southwest Milling...
-
For this assignment you will draw a Venn Diagram with three circles, one for each of the creation myths of this section- Out of the Blue, Genesis, and Popul Vu. You should compare and contrast them...
-
(e) Silicon has a Young's modulus, E, of 179 GPa and a density of 2330 kg.m. Q 1(f) (i) What is the mass of a cantilever of length (/) 100 m, width (w) 10 m and thickness (t) 1 m? (ii) Using the...
-
Little Oil Co. is offering a $1000 par bond with a 8% coupon. The bonds will mature in 10 years. If the current market rate for these kinds of bonds is 6% what will the bond sell for? And is it sold...
-
Imagine that in a different neuron, voltage-gated K+ channels open quickly, only 0.3ms after voltage-gated Na+ channels. Predict changes in timing, amplitude, and shape of an AP by drawing one...
-
1.The portfolio uses 75% SPY and 25% bonds.The portfolio market value is $100M.The bond is 8 years semi, yielding 4%, coupon 9%, BBB rating, reinvestment rate of 3%.The available futures are the ES...
-
What is the risk-free rate that should be included in the MRP? There are lots of options, explain your selection. Since you have determined the risk-free rate, how do you determine the market risk...
-
1)How CPP (Canada Pension Plan) becomes a part of the Financial Planning discussion. 2) How OAS/GIS becomes a part of the Financial Planning discussion. 3) List two promises that the Liberal...
-
Li Ping and Joseph are planning to purchase a second home in six years. They plan to invest $1,000 at the end of each quarter to save for this purchase. The investment has a stated annual rate of 9%....
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
Percentage of sales models usually assume that costs, fixed assets, and working capital all increase at the same rate as sales. When do you think that these assumptions do not make sense? Would you...
-
Common Products has just made its first issue of stock. It raised $2 million by selling 200,000 shares of stock to the public. These are the only shares outstanding. The par value of each share was...
-
Modern Artifacts can produce keepsakes that will be sold for $80 each. Nondepreciation fixed costs are $1,000 per year, and variable costs are $60 per unit. The initial investment of $3,000 will be...
-
A construction contract differs from contracts that we generally deal with that focus on an easily defined physical object because the physical object can be examined. How is the object of a...
-
What does the owner contribute to the project and what does the contractor contribute to the project?
-
For what type of project is a line-of-balance schedule particularly suited? Identify specific examples.

Study smarter with the SolutionInn App


