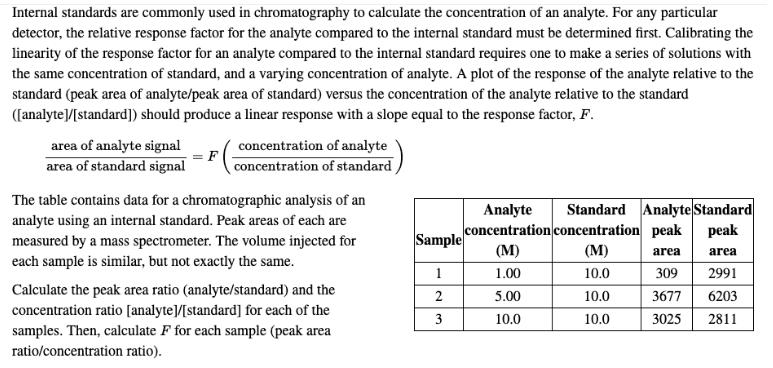
Internal standards are commonly used in chromatography to calculate the concentration of an analyte. For any...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Internal standards are commonly used in chromatography to calculate the concentration of an analyte. For any particular detector, the relative response factor for the analyte compared to the internal standard must be determined first. Calibrating the linearity of the response factor for an analyte compared to the internal standard requires one to make a series of solutions with the same concentration of standard, and a varying concentration of analyte. A plot of the response of the analyte relative to the standard (peak area of analyte/peak area of standard) versus the concentration of the analyte relative to the standard ([analyte]/[standard]) should produce a linear response with a slope equal to the response factor, F. area of analyte signal concentration of analyte area of standard signal concentration of standard The table contains data for a chromatographic analysis of an analyte using an internal standard. Peak areas of each are measured by a mass spectrometer. The volume injected for Standard Analyte Standard Analyte concentration concentration peak (M) pea Sample (M) area area each sample is similar, but not exactly the same. 1.00 10.0 309 2991 Calculate the peak area ratio (analyte/standard) and the concentration ratio [analyte]/[standard] for each of the samples. Then, calculate F for each sample (peak area ratio/concentration ratio). 5.00 10.0 3677 6203 3 10.0 10.0 3025 2811 Internal standards are commonly used in chromatography to calculate the concentration of an analyte. For any particular detector, the relative response factor for the analyte compared to the internal standard must be determined first. Calibrating the linearity of the response factor for an analyte compared to the internal standard requires one to make a series of solutions with the same concentration of standard, and a varying concentration of analyte. A plot of the response of the analyte relative to the standard (peak area of analyte/peak area of standard) versus the concentration of the analyte relative to the standard ([analyte]/[standard]) should produce a linear response with a slope equal to the response factor, F. area of analyte signal concentration of analyte area of standard signal concentration of standard The table contains data for a chromatographic analysis of an analyte using an internal standard. Peak areas of each are measured by a mass spectrometer. The volume injected for Standard Analyte Standard Analyte concentration concentration peak (M) pea Sample (M) area area each sample is similar, but not exactly the same. 1.00 10.0 309 2991 Calculate the peak area ratio (analyte/standard) and the concentration ratio [analyte]/[standard] for each of the samples. Then, calculate F for each sample (peak area ratio/concentration ratio). 5.00 10.0 3677 6203 3 10.0 10.0 3025 2811
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Calculate the concentration of an aqueous solution of NaOH that has a pH of 11.50.
-
Calculate the concentration of an aqueous solution of Ca(OH)2 that has a pH of 10.05.
-
Calculate the concentration of HC 6 H 6 O 6 - in an aqueous solution of 0.0896 M ascorbic acid , H 2 C 6 H 6 O 6 (aq). [ HC 6 H 6 O 6 - ] = M.
-
a. Over a two year time horizon an investor experiences the following net cash flows: Year 0 1 2 Cash flow -$8,000 $5,280 $5,320 What is the internal rate of return for the investor? Demonstrate that...
-
What is the difference between a probability and non-probability sample?
-
John Holland Incorporated provides you with the following information. The company does not report any book tax differences and is subject to a 21% income tax rate. Holland is a US Corporate tax...
-
Forester Company is evaluating the purchase of equipment from two vendors. Differences in the technology and labor requirements to operate the equipment of each vendor affect the projected net cash...
-
Rocky Bayou Golf Clubs, which uses the FIFO method, has the following account balances at July 31, 2012, prior to releasing the financial statements for the year: Rocky Bayou has determined that the...
-
Question 20 5 pts ABC Inc (US). plans to create and finance a subsidiary in Mexico that produces computer components a low cost and exports them other countries. It has no other international...
-
Price Company purchased 90% of the outstanding common stock of Score Company on January 1, 2016, for $450,000. At that time, Score Company had stockholders? equity consisting of common stock,...
-
Suppose a zip-lock bag contains ping-pong balls numbered S = {1, 2, ..., 59, 60}. Define Events: "Odd" = {1, 3, 5, ..., 55, 57, 59} "Even" = {2, 4, 6, ..., 56, 58, 60} "Lowest" = {1, 2, 3, 4, 5, 6,...
-
At the beginning of the current year, an entity purchased two (2) machines for Php1,000,000 each. The machines were put into use immediately. Machine A has useful life of 5 years and can be used only...
-
Amanda sold her Santa Barbara ranch to James. James took the deed Amanda delivered and had it recorded. James then sold the property to Emily and delivered a deed. Emily placed the deed in her safe...
-
Explain the terms "Omitted variable bias (OVB)", "Reverse causality" and "Survivorship bias/selection bias". Please provide me with an example of each term. After you study those three term, explain...
-
Describe how a nursing facility might go about marketing itself and the services it provides to the community. When might management use a public relations firm? provide a details answer with...
-
1. Why can exercising stock options can create cash flow problems for managers at the exercise date? What is an alternative to this problem? 2. Please answer the following questions about defined...
-
A detailed review of the ACA Restaurant Caf' Business plan prior to commencement is recommended. Sections of the business plan can be used throughout this assessment and you may refer to its details...
-
Sue Deliveau opened a software consulting firm that immediately paid $2,000 for a computer. Was this event a transaction for the business?
-
Consider the data presented in Exercise 14.19. (a) By using appropriate graphs, determine whether the reaction is first order or second order. (b) What is the rate constant for the reaction? (c) What...
-
What are the basic SI units for (a) The wavelength of light, (b) The frequency of light, (c) the speed of light?
-
How many nonbonding electron pairs are there in each of the following molecules: (a) (CH3)2S, (b) HCN, (c) H2C2, (d) CH3F?
-
Match the concept listed in items 1 through 6 with the most appropriate description listed in items a through \(f\). Description Concept 1. Enterprise Resource Planning (ERP) a. system 2. Brands of...
-
Indicate which type of data analytics, 1 through 4, is described in each of the following statements, a through \(h\). Data Analytics 1. Descriptive analytics 2. Diagnostic analytics 3. Predictive...
-
Match each item in the value chain, 1 through 7 , with an impact of an artificial intelligence initiative, choosing from items a through g. Value Chain 1. Research and development 2. Design 3. Supply...

Study smarter with the SolutionInn App


