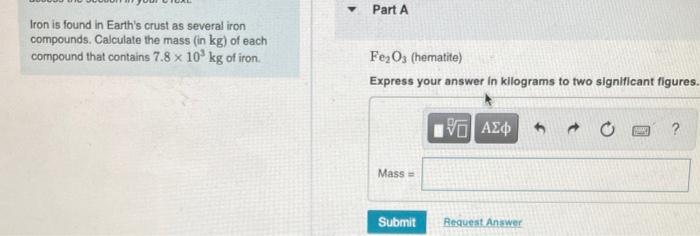
Iron is found in Earth's crust as several iron compounds. Calculate the mass (in kg) of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
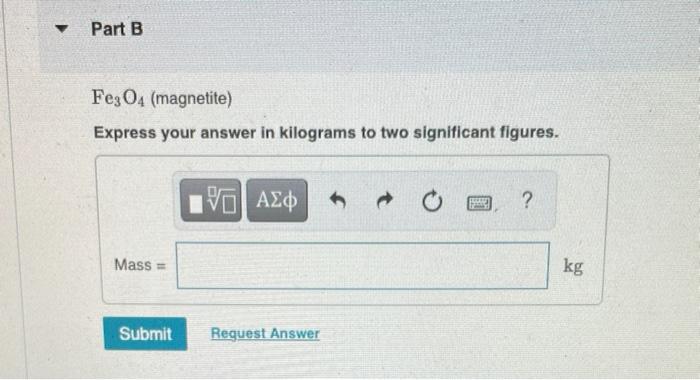
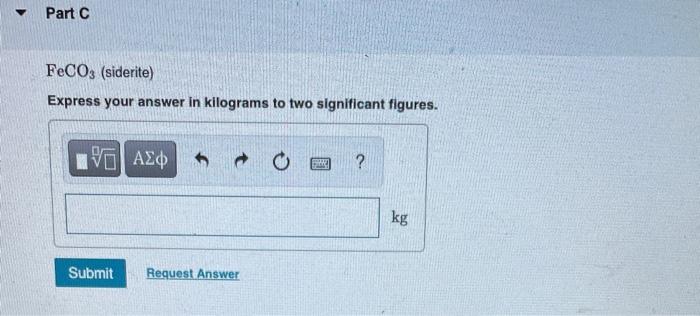
Iron is found in Earth's crust as several iron compounds. Calculate the mass (in kg) of each compound that contains 7.8 x 10³ kg of iron. Y Part A Fe₂O3 (hematite) Express your answer in kilograms to two significant figures. Mass= Submit [5] ΑΣΦ Request Answer B Part B Fe3O4 (magnetite) Express your answer in kilograms to two significant figures. Mass= Submit 15. ΑΣΦ Request Answer 1988 ? kg Part C FeCO3 (siderite) Express your answer in kilograms to two significant figures. ΕΙ ΑΣΦ Submit Request Answer www ? kg Iron is found in Earth's crust as several iron compounds. Calculate the mass (in kg) of each compound that contains 7.8 x 10³ kg of iron. Y Part A Fe₂O3 (hematite) Express your answer in kilograms to two significant figures. Mass= Submit [5] ΑΣΦ Request Answer B Part B Fe3O4 (magnetite) Express your answer in kilograms to two significant figures. Mass= Submit 15. ΑΣΦ Request Answer 1988 ? kg Part C FeCO3 (siderite) Express your answer in kilograms to two significant figures. ΕΙ ΑΣΦ Submit Request Answer www ? kg
Expert Answer:
Answer rating: 100% (QA)
i Calculate the force in the horizontal and inclined members of the gantr... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Compound A has two stereoisomers, but compounds B and C exist as single compounds. Explain. CH CH CH CH2 HCH-CH2 CH2CH3 CH3
-
The two blocks A and B each have a mass of 5 kg and are suspended from parallel cords. A spring having a stiffness of K = 60 N/n, is attached to B and is compressed 0.3 m against A and B as shown....
-
The small cylinder C has as mass of 10 kg and is attached to the end of a rod whose mass may be neglected. If the frame is subjected to a couple M = (8t2 + 5) N, m where t is in seconds, and the...
-
What types of costs should be considered in deriving the economic order quantity?
-
In what ways can a corporations structure and culture be internal strengths or weaknesses?
-
Atomic electrons interact with the nucleus with not only the electromagnetic interaction (photon exchange) but also with neutral current weak interactions (Z exchange). The latter contribution is...
-
Ann Duxbury started her practice as a design consultant on March 1, 2010. During the first month of operations, the business completed the following transactions: Requirements 1. Open the following...
-
Calculating Returns and Standard Deviations Based on the following information, calculate the expected return and standard deviation for the two stocks: State of Rate of Return if State Occurs...
-
The board of directors and chief marketing officer (CMO) are sitting in on the weekly meeting you have with your team. You have decided to make a five-page informational brochure for the team to...
-
Consider a coal plant with the following characteristics: Capital cost of $3300 per kW CCF of 0.13 FOM costs of $103 per kW-year Fuel costs of $1.07 per GJ Utilization of 72% (about 6312 hours per...
-
1. What R command(s) would you use to remove null values from a dataset? 2. What R command can be used to install an additional R package? 3. What R function is used to encode a vector as a category?...
-
Sunland Health is considering two alternatives for the financing of some high technology medical equipment. These two alternatives are: 1. Issue 60,000 shares of $10 par value common stock at $50 per...
-
In June 2023, Sue exchanges a sport-utility vehicle (adjusted basis of $118,880; fair market value of $148,600) for cash of $22,290 ar pickup truck (fair market value of $126,310). Both vehicles are...
-
Find the derivative of the function. y = ln(x = 7) -
-
Stock A has an expected return of 1 1 % and a standard deviation of 3 5 % . Stock B has an expected return of 1 9 % and a standard deviation of 6 5 % . The correlation coefficient between Stocks A...
-
2. A law enforcement officer cannot accept a reward on a fugitive when the law enforcement office apprehends the individual. This is due to multiple reasons. Explain, though, how consideration plays...
-
What can you learn about each of the following topics? Public Agencies and Cooperative Purchasing Broader Public Sector Directive and Trade Agreements Risk Management and Case Analysis Review Public...
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
Helium contains the same number of electrons in its outer shell as do the alkaline earth metals. Explain why helium is inert whereas the Group 2A metals are not.
-
A quantity of 0.225 g of a metal M (molar mass = 27.0 g/mol) liberated 0.303 L of molecular hydrogen (measured at 17C and 741 mmHg) from an excess of hydrochloric acid. Deduce from these data the...
-
You are given a soluble compound of unknown molecular formula. (a) Describe three tests that would show that the compound is an acid.
-
Analytical procedures: a. Are intended to detect material misstatements in financial statement accounts. b. Are concerned with how internal control policies or procedures are applied. c. Are...
-
Substantive tests of details: a. Are intended to detect material misstatements in financial statement accounts. b. Are concerned with how internal control policies or procedures are applied. c. Are...
-
Tests of controls: a. Are intended to detect material misstatements in financial statement accounts. b. Are concerned with how internal control policies or procedures are applied. c. Are evaluations...

Study smarter with the SolutionInn App


