Compound D, C4H8O, shows a downfield singlet in its 'H-NMR spectrum. Is it compound a or...
Fantastic news! We've Found the answer you've been seeking!
Question:


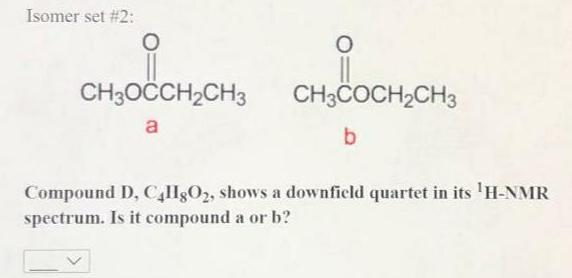
Transcribed Image Text:
Compound D, C4H8O₂, shows a downfield singlet in its 'H-NMR spectrum. Is it compound a or b? Isomer set #1: O SOCCHICH₂ CHICOCH.CH a b Isomer set #2: O CH₂OCCH₂CH₂ CH₂COCH,CHS a b Compound D, C4llgO2, shows a downfield quartet in its ¹H-NMR spectrum. Is it compound a or b? Compound D, C4H8O₂, shows a downfield singlet in its 'H-NMR spectrum. Is it compound a or b? Isomer set #1: O SOCCHICH₂ CHICOCH.CH a b Isomer set #2: O CH₂OCCH₂CH₂ CH₂COCH,CHS a b Compound D, C4llgO2, shows a downfield quartet in its ¹H-NMR spectrum. Is it compound a or b?
Expert Answer:
Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Isomer set #1: Compound D, C 4 H 8 O 2 , shows a downfield singlet in its 1 H-NMR spectrum. Is it compound a or b? ___ab Isomer set #2: Compound D, C 4 H 8 O 2 , shows a downfield quartet in its 1...
-
A compound X (C10H14O) dissolves in aqueous sodium hydroxide but is insoluble in aqueous sodium bicarbonate. Compound X reacts with bromine in water to yield a dibromo derivative, C10H12Br2O. The...
-
When dissolved in CDCl3, a compound (K) with the molecular formula C4H8O2 gives a 1H NMR spectrum that consists of a doublet at d 1.35, a singlet at d 2.15, a broad singlet at d 3.75 (1H), and a...
-
help me pls P acquired \( 80 \% \) of the capital of S on 1 January 2021. At the year end 31 December 2021 the two companies have the following statements of financial position: Notes 1....
-
A hypothetical element A has the following properties: First ionization energy: 850 kJ/mol Second ionization energy: 1700 kJ/mol Third ionization energy: 13,999 kJ/mol a. If you were to react element...
-
Explain what a guardian ad litem (GAL) is. What role do GALs play in custody cases?
-
Describe the impact of a payroll tax on wages and employment in a competitive industry. Why is part of the tax shifted to workers? What is the deadweight loss of the payroll tax?
-
Carson Corporation produces and sells three products, Alpha, Beta, and Gamma, in a local market and in a regional market. At the end of the first quarter of the current year, the following income...
-
Describe the situation from either your professional experience or your research in which unethical or fraudulent behavior occurred. If you are describing an example from experience, please do not...
-
In problem 8.16, a college chemistry instructor thinks the use of embedded tutors will improve the success rate in introductory chemistry courses. The instructor carried out a hypothesis test and...
-
You have been tasked with incorporating interest rates that are linked to the SARB rates. This means that any changes to the repo rate will affect the interest rate used for a loan. Your client would...
-
A McDonalds Happy Meal consists of an entre, a small order of French fries, a small drink, and a toy. In the early 1990s, McDonalds Corp. began to aim its Happy Meal marketing at children aged one to...
-
How would the following transactions be categorized in the U.S. balance of payments accounts? Would they be entered in the current account (as a payment to or from a foreigner) or the financial...
-
In setting monetary policy, which central bankone that operates according to a Taylor rule or one that operates by inflation targetingis likely to respond more directly to a financial crisis? Explain.
-
The Internet Corporation for Assigned Names and Numbers (ICANN) is a nonprofit entity that organizes Internet domain names. It is governed by a board of directors elected by various groups with...
-
Suppose the United States and Japan are the only two trading countries in the world. What will happen to the value of the U.S. dollar if the following occur, other things equal? a. Japan relaxes some...
-
hi im wondering what i can do to fix this and please explain it to me thank you i appreciate it 1 input values input("Please enter a series of intergers seperated by spaces: ") 2 user_integers =...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
As a method for the synthesis of cinnamaldehyde (3-phenyl-2-propenal), a chemist treated 3-phenyl-2-propen-1-ol with K2Cr2O7 in sulfuric acid. The product obtained from the reaction gave a signal at...
-
The initial steps of a laboratory synthesis of several prostaglandins reported by E. J. Corey (Section 7.15B) and co-workers in 1968 are outlined here. Supply each of the missing reagents: (a) (b)...
-
If (2Z,4Z)-hexa-2,4-diene were able to undergo a Diels-Alder reaction with methyl propenoate, what would be the products? (Hint: There are four products comprised of two pairs of enantiomers. One...
-
You are the product manager of Lexcon Fitness and are considering the production of new elliptical trainers. You feel you can sell 5,500 of these per year for 6 years (after which time this project...
-
At present, Global Skateboards Limited is considering expanding its product line to include solar powered skateboards; however, it is questionable how well they will be received by skateboarders....
-
Assume that Fiskars Finland plans to invest in a new project that will annually generate revenues of \($2.3\) million. Cash expenses including both fixed and variable costs will be \($950,000,\) and...

Study smarter with the SolutionInn App



