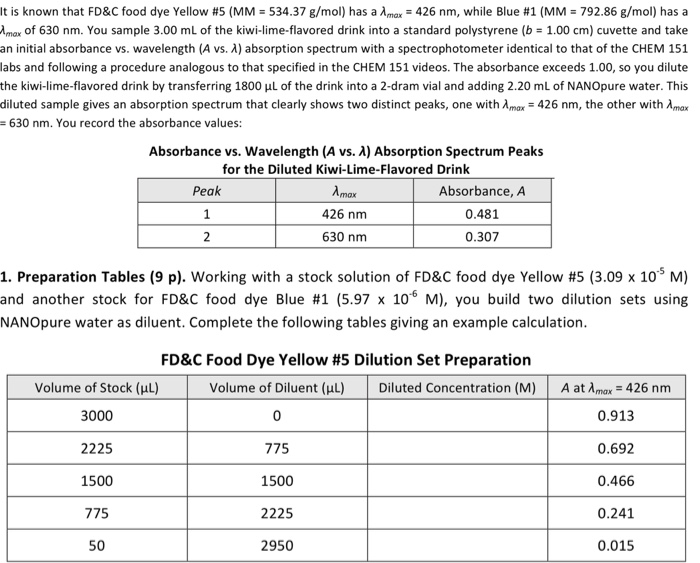
It is known that FD&C food dye Yellow # 5 (MM = 534.37 g/mol) has a...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
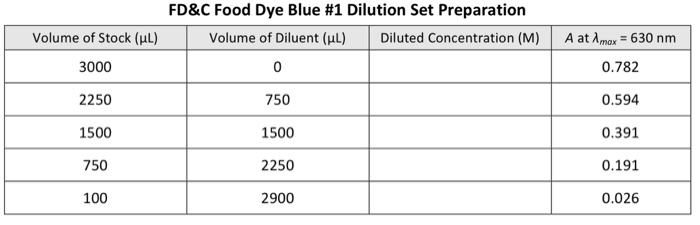
It is known that FD&C food dye Yellow # 5 (MM = 534.37 g/mol) has a Amax = 426 nm, while Blue #1 (MM = 792.86 g/mol) has a Amax of 630 nm. You sample 3.00 mL of the kiwi-lime-flavored drink into a standard polystyrene (b = 1.00 cm) cuvette and take an initial absorbance vs. wavelength (A vs. X) absorption spectrum with a spectrophotometer identical to that of the CHEM 151 labs and following a procedure analogous to that specified in the CHEM 151 videos. The absorbance exceeds 1.00, so you dilute the kiwi-lime-flavored drink by transferring 1800 µL of the drink into a 2-dram vial and adding 2.20 mL of NANOpure water. This diluted sample gives an absorption spectrum that clearly shows two distinct peaks, one with Amax = 426 nm, the other with Amax 630 nm. You record the absorbance values: Absorbance vs. Wavelength (A vs. A) Absorption Spectrum Peaks for the Diluted Kiwi-Lime-Flavored Drink Peak 1 2 Volume of Stock (μL) 3000 2225 1500 775 50 Amax 426 nm 630 nm 1. Preparation Tables (9 p). Working with a stock solution of FD&C food dye Yellow #5 (3.09 x 10¹5 M) and another stock for FD&C food dye Blue #1 (5.97 x 10 M), you build two dilution sets using NANOpure water as diluent. Complete the following tables giving an example calculation. Absorbance, A 0.481 0.307 2950 FD&C Food Dye Yellow # 5 Dilution Set Preparation Volume of Diluent (μl) 0 775 1500 2225 Diluted Concentration (M) A at Amax = 426 nm 0.913 0.692 0.466 0.241 0.015 Volume of Stock (μl) 3000 2250 1500 750 100 FD&C Food Dye Blue # 1 Dilution Set Preparation Volume of Diluent (μl) Diluted Concentration (M) 0 750 1500 2250 2900 A at Amax = 630 nm 0.782 0.594 0.391 0.191 0.026 2. Molar Absorptivity (8 p). Determine the molar absorptivity, , for the two dyes. Paste. Include the linear trendline equation(s) and R2 value(s). Clearly state the determined & for e 2. Molar Absorptivity (8 p). Determine the molar absorptivity, &, for the two dyes. Pastel Include the linear trendline equation(s) and R2 value(s). Clearly state the determined & for e It is known that FD&C food dye Yellow # 5 (MM = 534.37 g/mol) has a Amax = 426 nm, while Blue #1 (MM = 792.86 g/mol) has a Amax of 630 nm. You sample 3.00 mL of the kiwi-lime-flavored drink into a standard polystyrene (b = 1.00 cm) cuvette and take an initial absorbance vs. wavelength (A vs. X) absorption spectrum with a spectrophotometer identical to that of the CHEM 151 labs and following a procedure analogous to that specified in the CHEM 151 videos. The absorbance exceeds 1.00, so you dilute the kiwi-lime-flavored drink by transferring 1800 µL of the drink into a 2-dram vial and adding 2.20 mL of NANOpure water. This diluted sample gives an absorption spectrum that clearly shows two distinct peaks, one with Amax = 426 nm, the other with Amax 630 nm. You record the absorbance values: Absorbance vs. Wavelength (A vs. A) Absorption Spectrum Peaks for the Diluted Kiwi-Lime-Flavored Drink Peak 1 2 Volume of Stock (μL) 3000 2225 1500 775 50 Amax 426 nm 630 nm 1. Preparation Tables (9 p). Working with a stock solution of FD&C food dye Yellow #5 (3.09 x 10¹5 M) and another stock for FD&C food dye Blue #1 (5.97 x 10 M), you build two dilution sets using NANOpure water as diluent. Complete the following tables giving an example calculation. Absorbance, A 0.481 0.307 2950 FD&C Food Dye Yellow # 5 Dilution Set Preparation Volume of Diluent (μl) 0 775 1500 2225 Diluted Concentration (M) A at Amax = 426 nm 0.913 0.692 0.466 0.241 0.015 Volume of Stock (μl) 3000 2250 1500 750 100 FD&C Food Dye Blue # 1 Dilution Set Preparation Volume of Diluent (μl) Diluted Concentration (M) 0 750 1500 2250 2900 A at Amax = 630 nm 0.782 0.594 0.391 0.191 0.026 2. Molar Absorptivity (8 p). Determine the molar absorptivity, , for the two dyes. Paste. Include the linear trendline equation(s) and R2 value(s). Clearly state the determined & for e 2. Molar Absorptivity (8 p). Determine the molar absorptivity, &, for the two dyes. Pastel Include the linear trendline equation(s) and R2 value(s). Clearly state the determined & for e
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In a certain federal prison it is known that 2/3 of the inmates are under 25 years of age. It is also known that 3/5 of the inmates are male and that 5/8 of the inmates are female or 25 years of age...
-
A company services home air conditioners. It is known that times for service calls follow a normal distribution with a mean of 60 minutes and a standard deviation of 10 minutes. a. What is the...
-
Of 600 car parts produced, it is known that 350 are produced in one plant, 150 parts in a second plant, and 100 parts in a third plant. Also it is known that the probabilities are 0.15, 0.2, and 0.25...
-
Damages require one person to pay money as compensation for another persons loss. Damages are therefore a private matter. Since the Charter is part of public law, damages cannot be awarded in...
-
Several years ago The Wall Street Journal reported that the winner of the Massachusetts State Lottery prize had the misfortune to be both bankrupt and in prison for fraud. The prize was $9,420,713,...
-
A consumer behavior researcher is curious as to whether, in terms of the monetary value of Christmas presents received, it makes a difference if a child is naughty or nice. She has parents classify...
-
Algonquin Equipment reported the following items on November 30, 2010 (amounts in thousands, with last years amounts also given as needed): Requirements 1. Compute Algonquin Equipments (a) quick...
-
At the end of 20X5, Singh Inc. has four inventory items, two of which management believes should be written down. The cost and estimated NRVs of the items are as follows: Required: 1. Determine the...
-
you work for a nuclear research laboratory that is contemplating leasing a diagnostic scanner ( leasing is a very common practice with expensive, high - tech equipment ) . the scanner costs $ 4 , 0 0...
-
An end loader for a small garden tractor is shown. All connections are pinned. The only significant weight is W . (a) Draw six FBDs, one each for members BC, DG, and AEF , plate CDE, hydraulic...
-
The Pitts Limited had EBITDA of $900 million in 2019. The depreciation amount for the period $300m. Firms increased investment in fixed assets by $400m from 2018. Firms overall investment in working...
-
What recent human rights violation has been in the media? What happened? Whose rights were violated? Why? What should happen next?
-
briefly explain two of the other 5 reasons our text identifies as a reason to study human relations ?
-
Leadership practices for human capital management, including ethical and legal responsibilities, and describe how these practices might apply to the HRM effectiveness improvement plan
-
in regards to disney world why should an organization be concerned about labor relations? Contrast the style of labor unions in the U.S. to that found in another country? Also, briefly discuss ways...
-
In what ways doyousee adult learningprinciplesandemotional literacy enacted inyourown learning experiences (consider both formal and informal experiences and settings)?
-
a. Suppose the number of pine trees in a certain forest follows the Poisson distribution with parameter per meter. Suppose we randomly select a point in this forest (not a pine tree, just a point)....
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
Show that when the optimal allocation is used, the variance of (2) str for two-phase sampling with stratification is given by (12.18).
-
For the data in golfsrs.dat, estimate the proportion of golf courses that have 18 holes, and give a 95% CI for the population proportion. Discuss.
-
Suppose the true model describing the relation between x and y is Where the εi are independently generated from a N (0, Ï2 i) distribution. Let Æ© be a matrix with...
-
Why will the audit profession move toward more testing of the systems processing as opposed to direct testing of account balances? Explain your rationale.
-
Why are data transmission controls so important? What are the key controls the auditor would expect to be present to ensure proper data transmission?
-
How does an auditor using an ITF keep from contaminating client files with data designed to test the clients processing? Describe the two approaches most often used.

Study smarter with the SolutionInn App


