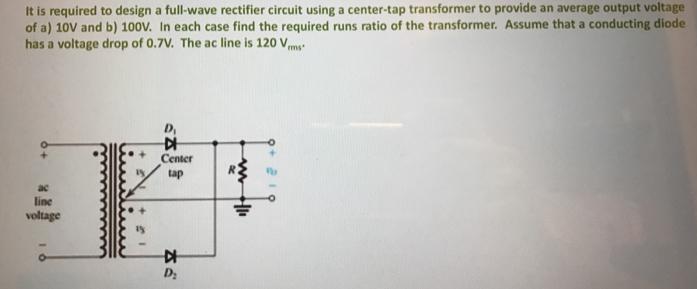
It is required to design a full-wave rectifier circuit using a center-tap transformer to provide an...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
It is required to design a full-wave rectifier circuit using a center-tap transformer to provide an average output voltage of a) 10V and b) 100V. In each case find the required runs ratio of the transformer. Assume that a conducting diode has a voltage drop of 0.7V. The ac line is 120 Vmy D, Center tap ac line voltage 中 D: It is required to design a full-wave rectifier circuit using a center-tap transformer to provide an average output voltage of a) 10V and b) 100V. In each case find the required runs ratio of the transformer. Assume that a conducting diode has a voltage drop of 0.7V. The ac line is 120 Vmy D, Center tap ac line voltage 中 D:
Expert Answer:
Answer rating: 100% (QA)
10V The requined 07 highes dic output wire be the diode hon ... View the full answer

Related Book For 

Posted Date:
Students also viewed these electrical engineering questions
-
In each case find the matrix A. (a) (b) A +2 0 2 2410 -12.1) =44-9 1 0
-
In each case find the QR-factorization of A. (a) (b) 1011 1101
-
In each case find the matrix of T: (R3 R3: (a) 7 is rotation through 6 about the x axis (from the y axis to the z axis). (b) 7 is rotation through 0 about the x axis (from the x axis to the z axis).
-
3 (a) A laser beam of uniform cross-sectional area passes through air into a glass block (refractive index 1.5). Determine the ratios of (i) the velocities (ii) the frequencies and (iii) the...
-
Why are G' values not rigorously applicable to biochemical systems?
-
Three insurance companies have insured a building in the ratio 1/2 to 1/3 to 2/5. How much of a fire loss of $185 000 should each company pay?
-
Consider the methanol oxidation data in Table B.20. Perform a thorough analysis of these data. What conclusions do you draw from this residual analysis? x x2 X3 X4 0 454 8.8 3.90 1.30 1.1 0 474 8.2...
-
Financial statements for Allendale Company follow: ALLENDALE COMPANY Balance Sheets As of December 31 2019 2018 Assets Current assets Cash $ 40,000 $ 36,000 Marketable securities 20,000 6,000...
-
A company owns a building that it rents for $ 3 , 0 0 0 per month. No payment was received for November or December although company officials do expect payment to be collected. If nothing has yet...
-
A month has elapsed since Precision Computer Centres year-end. Tony Freedman will use four specialized journals for recording business transactions in the month of Septemberyou will find two in...
-
John purchased 300 shares of Big Trouble common stock at $26.75 a share in July of 2015. In June of 2016 John buys 3 October 35 puts at 3. If the market of Big Trouble is trading at $33.16 at...
-
Saline solution (a 0.89 % (w/w) sodium chloride solution) is often used to provide intravenous water. Why is distilled water unsuitable? O Cells could shrink due to the difference in osmotic pressure...
-
will Word problem on direct variation 0/5 Arrielle For a moving object, the force acting on the object varies directly with the object's acceleration. When a force of 56 N acts on a certain object,...
-
2. What pressure in psi is equivalent to 2.40 atm?
-
The following diagram represents the risk/return position of four portfolios of shares. Portfolio D is the market portfolio (the portfolio made up of all risky assets in the market in the same...
-
5 (1 point) If I perform this reaction: FeBr + 2 KCl FeCl +2 KBr. What is my theoretical yield of iron (II) chloride if I start with 34 grams of iron (II) bromide? 19.9g 16.9g 17.9g 18.9g
-
A uniformly charged spherical shell has inner and outer radii of 28.0 cm and 32.0 cm, respectively. A point charge of -45.0 nC is located at the center of the spherical shell. A proton orbits around...
-
Which of the followingcarbocations is the least stable? CH3CH2 . CH3CHCH3 CH3 I . CH3C0 T CH3 IV. V. CH3 CH3CCH2 CH3
-
For the VOC stripping task of Exercise 6.26, the expert has suggested that we use a tower diameter of 0.80 m for which we can expect a pressure drop of 500 N/m2-m of packed height (0.612 in. H2O/ft)....
-
For the conditions of Example 15.8 estimate the effective diffusivity of acetone vapor in the pores of activated carbon with the following properties: particle density = 0.85 g/cm 3 , particle...
-
A light-hydrocarbon feed stream contains 45.4 kmol/h of propane, 136.1 kmol/h of isobutane, 226.8 kmol/h of n-butane, 181.4 kmol/h of isopentane, and 317.4 kmol/h of n-pentane. This stream is to be...
-
Selected transactions from the journal of Wong Consultants are presented below (amounts in thousands). Instructions a. Post the transactions to T-accounts. b. Prepare a trial balance at August 31,...
-
The T-accounts below summarize the ledger of Negrete Landscaping at the end of the first month of operations (amounts in ). Instructions a. Prepare the complete general journal (including...
-
Lennon Industries had the following transactions. 1. Borrowed 5,000 from the bank by signing a note. 2. Paid 3,900 cash for a computer. 3. Purchased 650 of supplies on account. Instructions a....

Study smarter with the SolutionInn App


