The correctly matched indicator(s) for the given titrations is/are (A) Fe?* vs K2Cr20,: diphenylamine (B) l2...
Fantastic news! We've Found the answer you've been seeking!
Question:
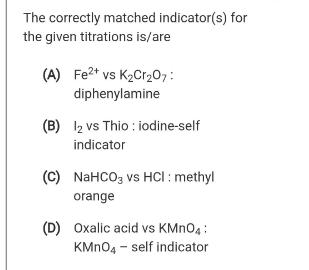
Transcribed Image Text:
The correctly matched indicator(s) for the given titrations is/are (A) Fe?* vs K2Cr20,: diphenylamine (B) l2 vs Thio : iodine-self indicator (C) NAHCO, vs HCI : methyl orange (D) Oxalic acid vs KMNO4 : KMNO4 - self indicator The correctly matched indicator(s) for the given titrations is/are (A) Fe?* vs K2Cr20,: diphenylamine (B) l2 vs Thio : iodine-self indicator (C) NAHCO, vs HCI : methyl orange (D) Oxalic acid vs KMNO4 : KMNO4 - self indicator
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Methyl orange is an azo dye used as an indicator in acid- base titrations. (It is yellow-orange above pH 4.5 and red below pH 3.) Show how it can be synthesized from p aminobenzenesulfonic acid...
-
The indicator methyl orange has been added to both of these solutions. Based on the colors, classify each statement as true or false: (a) The pH of solution A is definitely less than 7.00. (b) The pH...
-
Oxalic acid is a toxic substance used by laundries to remove rust stains. Its composition is 26.7% C, 2.2% H, and 71.1% O (by mass), and its molecular mass is 90 amu. What is its molecular formula?
-
What do you think Dan Barry means when he claims that Ring Dings taste like America?
-
Potassium metal has a body-centered cubic structure with all atoms at the lattice points. The density of the metal is 0.856 g/cm3. Calculate the edge length of a unit cell.
-
Some people believe that chatbots will change how people interact with the Internet and browse online. Prepare a report regarding this.
-
The 2015 annual report of The Procter & Gamble Company (P&G) TA 2, 3, 4 is available at www.pginvestor.com. After reviewing P&Gs annual report, respond to the following questions: a. What percentage...
-
The income statement of Brandon Co. for the month of July shows net income of $1,400 based on Service Revenue $5,500, Salaries and Wages Expense $2,300, Supplies Expense $1,200, and Utilities Expense...
-
How can the relationship between time and value be applied to calculate capital expenditures and progress towards long-term goals? Question 32Answer a. By determining the number of regular deposits...
-
4. (Chapter 3) Skycell, a major European cell phone manufacturer, is making production plans for the coming year. Skycell has worked with its customers (the service providers) to come up with...
-
Ceramic Products, Inc. reports these account balances on January 1, 2015 (in alphabetical order): Accounts payable 23,000 $ Accounts receivable 15,000 $ Buildings 148,000 $ Capital Stock 180,000 $ in...
-
Why hormones reference interval is very low comparing with other molecules in blood? Explain
-
What comes closest to XYZ Corp's beta if the expected return on its stock is 15%, the expected market return is 8%, and the risk-free rate is-2%?
-
Ben has been offered two options for his salary as a trainer at the gym. Option A would pay him $23500 dollars plus $15 for each training session. Under Option A, Ben would be paid P = Option B would...
-
c) A wooden beam (Specific gravity = 0.64) is 140 mm by 5 m and is hinged at A, as shown in Fig 3.2. At what angle, the beam will float in water? 5m +(5-L) 0 Fig 3.2 Water VW beam t (40%)
-
How Intrinsic valuation is more useful than relative valuation , discuss in detail
-
Help Save & Exit Submit When constructing a quick review table in Excel, knowing the formula is essential in populating the cells correctly. Using the Holts exponential smoothing method, complete the...
-
General Electric Capital, a division of General Electric, uses long-term debt extensively. In a recent year, GE Capital issued $11 billion in long-term debt to investors, then within days filed legal...
-
A concentration of 8.00 102 ppm by volume of CO is considered lethal to humans. Calculate the minimum mass of CO in grams that would become a lethal concentration in a closed room 17.6 m long, 8.80...
-
Use ionization energy and electron affinity values (see Table 8.3) to calculate the energychange (in kJ/mol) for the following reactions: (a) Li(g) + I(g) Li+ (g) + I - (g) (b) Na(g) + F(g) Na+ (g)...
-
Draw Lewis structures for chlorine nitrate (ClONO2) and chlorine monoxide (ClO).
-
Show that if there is a non-basic variable all of whose coefficients are non-negative at some stage of execution, then the problem is unbounded, as the message described above claims.
-
Show inductively that at each pass through the loop, the next system represents a basic feasible solution. Show in addition that if there are no degeneracies, the value of the objective increases...
-
Prove that if no degeneracies are encountered at any stage, then the algorithm terminates in finitely many steps with with an unboundedness message or an optimal solution.

Study smarter with the SolutionInn App



