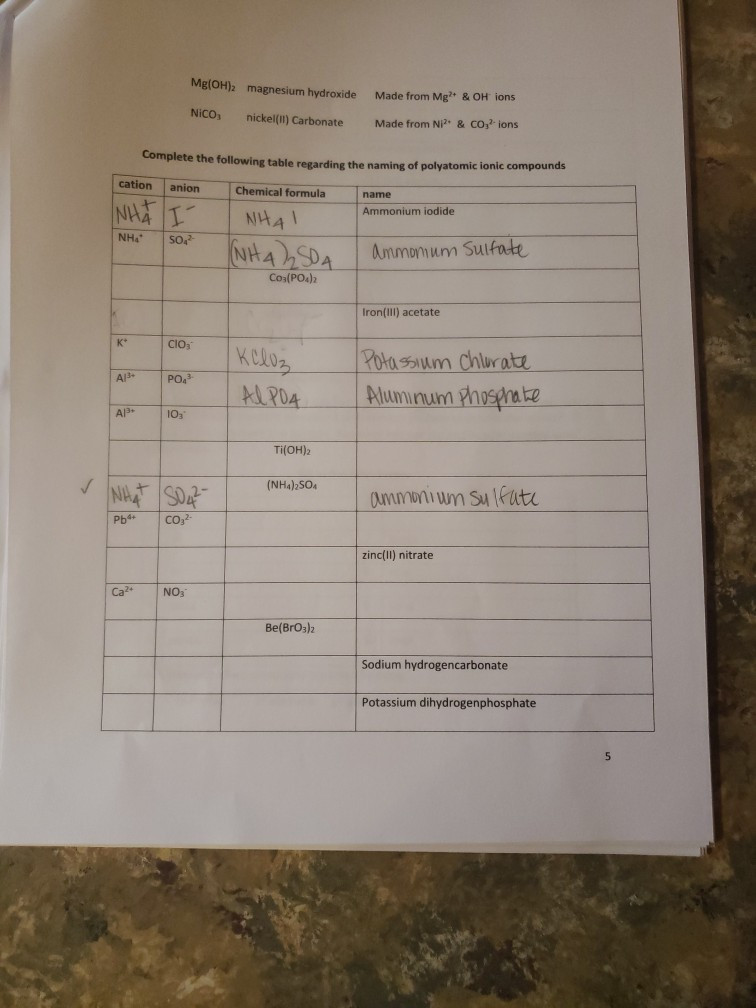
K* Complete the following table regarding the naming of polyatomic ionic compounds cation anion Chemical formula...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
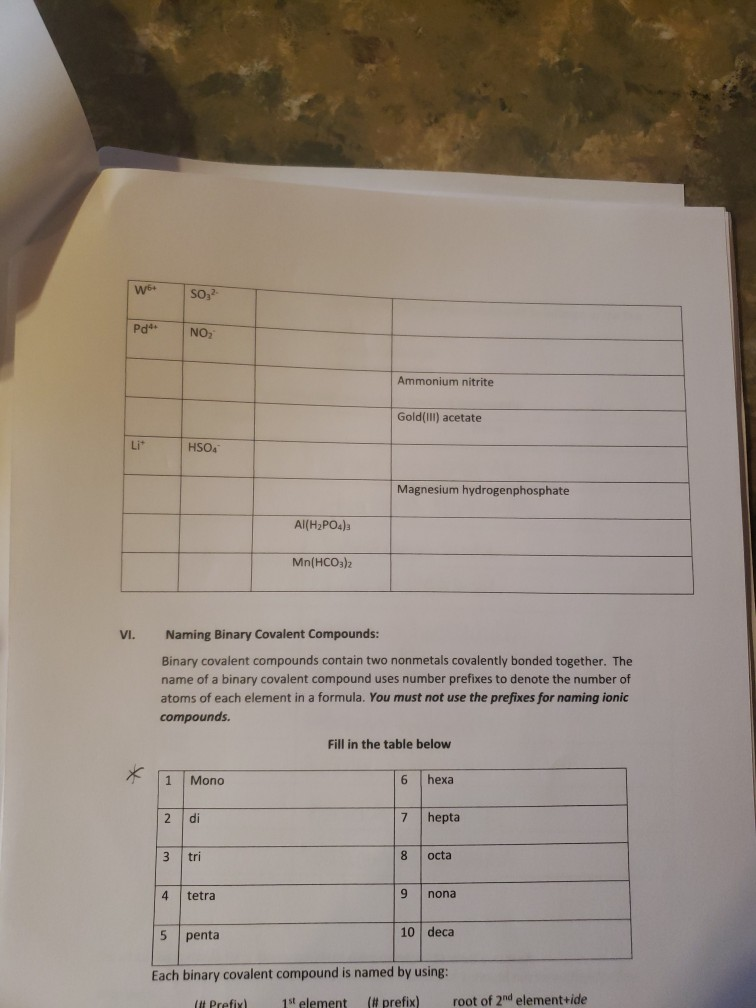
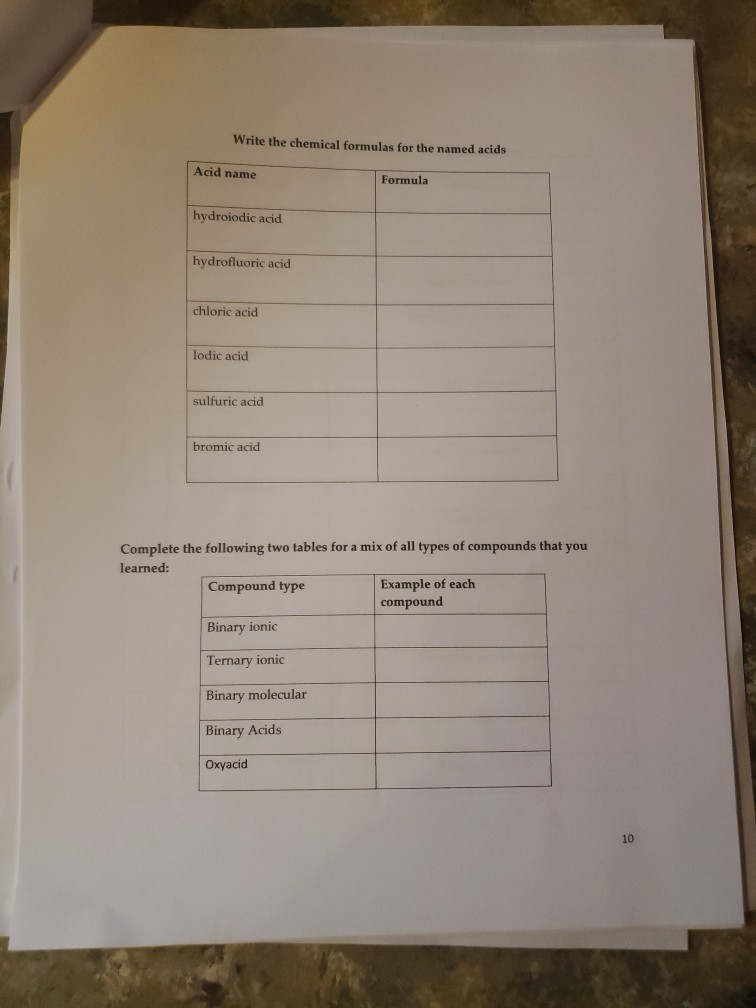
✓ K* Complete the following table regarding the naming of polyatomic ionic compounds cation anion Chemical formula NHÀ Í NH41 NH₂ NHASDA CO₂(PO4)2 Al3+ Al³+ Ca²+ Mg(OH)2 magnesium hydroxide NICO₁ nickel(II) Carbonate SO4²- CIOs PO4³ 10₂₁ NHÀ SO Pb4+ CO32- NO₁™ кслоз AlPO4 Ti(OH)2 (NH4)2SO4 Made from Mg2+ & OH ions Made from Ni²+ & CO3²- ions Be(BrO3)2 name Ammonium iodide Ammomum Sulfate Iron(III) acetate Potassium Chlorate Aluminum Phosphate ammonium sulfate zinc(II) nitrate Sodium hydrogencarbonate Potassium dihydrogenphosphate W5+ Pd4 Li VI. SO₂²- 1 NO₂ HSOA Mono 2 di 3 tri Naming Binary Covalent Compounds: Binary covalent compounds contain two nonmetals covalently bonded together. The name of a binary covalent compound uses number prefixes to denote the number of atoms of each element in a formula. You must not use the prefixes for naming ionic compounds. 4 tetra Al(H₂PO4)3 5 penta Mn(HCO3)2 Ammonium nitrite Gold(III) acetate Magnesium hydrogenphosphate Fill in the table below 6 7 8 9 hexa hepta octa nona 10 deca Each binary covalent compound is named by using: ( Prefix) 1st element (# prefix) root of 2nd element+ide Write a formula for the following binary covalent compounds: carbon disulfide sulfur trioxide dinitrogen pentoxide trisilicon tetrahydride sulfur hexafluoride Trisulfur heptanitride VII. Naming molecular acids: Acids are uncharged molecules. They contain the familiar anions with added H* ions. Naming Binary Acids: Binary acids (two atoms types only, hydrogen + a nonmetal) are named by adding hydro at the beginning of the respective anion name and the end of the anion name is changed to ic acid from ide. Ex. HCl, a binary acid is named: hydrochloric acid (respective anion is chloride [Cl]). Naming ternary oxyacids: Oxyacids are ternary (3 elements). They contain oxygen, hydrogen and another element. If the anion is an oxyanion then change the ate ending of the polyatomic anion that they contain to ic acid. A prefix is not needed. Most molecular ions that you have learnt about have an oxygen in them and they are therefore oxyanions. Ex. HNO3 is named: Nitric acid (respective anion is nitrate [NO3]). Continue to next page Write the chemical formulas for the named acids Acid name hydroiodic acid hydrofluoric acid chloric acid Iodic acid sulfuric acid bromic acid Complete the following two tables for a mix of all types of compounds that you learned: Compound type Formula Binary ionic Ternary ionic Binary molecular Binary Acids Oxyacid Example of each compound 10 Name S/N 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Chemical Formula SinCh H₂O₂ COS SnO₂ Cds(PO4)2 Zn3P₂ Pb(OH)2 H₂SO4 SnP₂ Section Time_ Name Beryllium iodide Triphosphorus hexanitride Gold(III) carbonate Copper (1) hydroxide Silver nitride Nitric acid Tetrasulfur pentachloride Aluminum nitride 11 18 19 20 21 22 23 24 25 26 27 28 29 30 31 33 34 35 36 37 38 Fe (SO4)3 NISO, Cu(NO3)2 НЕ Cd(CH3COO)2 Pb(BrO3)2 KNO InP MnSe₂ V₂05 Zinc(11) hydrogencarbonate Copper (1) sulfide Chromium(VI) dihydrogenphosphate Phosphoric acid Iron(III) carbonate Acetic acid Cobalt(II) chlorite Barium Nitrite Strontium phosphate Titanium(II) bromide 12 ✓ K* Complete the following table regarding the naming of polyatomic ionic compounds cation anion Chemical formula NHÀ Í NH41 NH₂ NHASDA CO₂(PO4)2 Al3+ Al³+ Ca²+ Mg(OH)2 magnesium hydroxide NICO₁ nickel(II) Carbonate SO4²- CIOs PO4³ 10₂₁ NHÀ SO Pb4+ CO32- NO₁™ кслоз AlPO4 Ti(OH)2 (NH4)2SO4 Made from Mg2+ & OH ions Made from Ni²+ & CO3²- ions Be(BrO3)2 name Ammonium iodide Ammomum Sulfate Iron(III) acetate Potassium Chlorate Aluminum Phosphate ammonium sulfate zinc(II) nitrate Sodium hydrogencarbonate Potassium dihydrogenphosphate W5+ Pd4 Li VI. SO₂²- 1 NO₂ HSOA Mono 2 di 3 tri Naming Binary Covalent Compounds: Binary covalent compounds contain two nonmetals covalently bonded together. The name of a binary covalent compound uses number prefixes to denote the number of atoms of each element in a formula. You must not use the prefixes for naming ionic compounds. 4 tetra Al(H₂PO4)3 5 penta Mn(HCO3)2 Ammonium nitrite Gold(III) acetate Magnesium hydrogenphosphate Fill in the table below 6 7 8 9 hexa hepta octa nona 10 deca Each binary covalent compound is named by using: ( Prefix) 1st element (# prefix) root of 2nd element+ide Write a formula for the following binary covalent compounds: carbon disulfide sulfur trioxide dinitrogen pentoxide trisilicon tetrahydride sulfur hexafluoride Trisulfur heptanitride VII. Naming molecular acids: Acids are uncharged molecules. They contain the familiar anions with added H* ions. Naming Binary Acids: Binary acids (two atoms types only, hydrogen + a nonmetal) are named by adding hydro at the beginning of the respective anion name and the end of the anion name is changed to ic acid from ide. Ex. HCl, a binary acid is named: hydrochloric acid (respective anion is chloride [Cl]). Naming ternary oxyacids: Oxyacids are ternary (3 elements). They contain oxygen, hydrogen and another element. If the anion is an oxyanion then change the ate ending of the polyatomic anion that they contain to ic acid. A prefix is not needed. Most molecular ions that you have learnt about have an oxygen in them and they are therefore oxyanions. Ex. HNO3 is named: Nitric acid (respective anion is nitrate [NO3]). Continue to next page Write the chemical formulas for the named acids Acid name hydroiodic acid hydrofluoric acid chloric acid Iodic acid sulfuric acid bromic acid Complete the following two tables for a mix of all types of compounds that you learned: Compound type Formula Binary ionic Ternary ionic Binary molecular Binary Acids Oxyacid Example of each compound 10 Name S/N 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Chemical Formula SinCh H₂O₂ COS SnO₂ Cds(PO4)2 Zn3P₂ Pb(OH)2 H₂SO4 SnP₂ Section Time_ Name Beryllium iodide Triphosphorus hexanitride Gold(III) carbonate Copper (1) hydroxide Silver nitride Nitric acid Tetrasulfur pentachloride Aluminum nitride 11 18 19 20 21 22 23 24 25 26 27 28 29 30 31 33 34 35 36 37 38 Fe (SO4)3 NISO, Cu(NO3)2 НЕ Cd(CH3COO)2 Pb(BrO3)2 KNO InP MnSe₂ V₂05 Zinc(11) hydrogencarbonate Copper (1) sulfide Chromium(VI) dihydrogenphosphate Phosphoric acid Iron(III) carbonate Acetic acid Cobalt(II) chlorite Barium Nitrite Strontium phosphate Titanium(II) bromide 12
Expert Answer:
Answer rating: 100% (QA)
Cation Anion Chemical Formulae Name NH4 I NH4I Ammoniu... View the full answer

Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Posted Date:
Students also viewed these chemistry questions
-
If the nominal rate of return is 11.09% and the inflation rate is 4.62%, by what percent is actual purchasing power increasing? Enter your answer as a percentage with four decimal places but without...
-
Complete the following table showing which ledger account is to be debited and which is to be credited: Debit Credit al Sratorery purchased on credit from Gomley bl Rates paid by direct debit...
-
Zannel plc is considering a new project to produce a revolutionary surveillance device. The initial capital costs of 300,000 will be paid immediately. The project is expected to last five years. The...
-
Compute the determinant below. 14 8 8 8 51 1000 8 7 3 0 0 0 8 8 8 8 8 2
-
In its report prepared for audit committees and other stakeholders in December 2014, "Our Commitment to Audit Quality," EY acknowledges that recent PCAOB findings indicate the need for improving...
-
Determine the optimum bit life for the bit run described below using cost per foot analysis. (13 Marks) Bit cost: 600 $ Rig operating cost: 1000 $ Trip time: 6 hours Drilling time (hours) Footage...
-
Microsoft, Inc., recognizes revenue from its licensed software products at the time of sale or time of installation. Despite receiving such revenue in cash, Microsoft defers the recognition of a...
-
On March 20, 10,000 shares of Thorlite Company are acquired at a price of $30 per share plus a $250 brokerage fee. On May 30, a $0.25-per-share dividend was received on the Thorlite Company stock. On...
-
Find the y-intercept of the line. 18 10 y=-- 17x+ 3
-
The plant can make two products, P and Q. Max demand for P= 100 units/week; Max demand for Q = 50 units/week. Four workers, A, B, C, and D, with non-interchangeable skills. Fixed operating expense =...
-
Get a live quote on insurance, car or health insurance. Explain the levels of coverage that you picked. For example, what is your deductible? State the annual cost for both the insurance and the...
-
A callable bond pays annual interest of $40, has a par value of $1000, matures in 4 years but is callable in 3 years at a price of $1100, and has a value today of $1020. What is the yield to call on...
-
After watching the Ted talk by Nadine Burke Harris, identify twoadverse childhood experiences, how they negatively impact mentalhealth and wellness, and two ways to cope with thetype of stress/trauma...
-
Camille Sikorski was divorced in 2018. She currently provides a home for her 15-year-old daughter Kaly. Kaly lived in Camille's home for the entire year, and Camille paid for all the costs of...
-
How has the rapid advancement of technology influenced social behavior and interpersonal relationships? Discuss the potential social consequences of technological integration in everyday life .
-
1 Each year Molly Madison makes and sells 30,000 statues of "The Thinker" for $60/unit. She only has capacity to make 30,000 statues. Unit costs, based on this quantity, is as follows: Direct...
-
The manager of Dixie Furniture Company estimates that the daily output of her factory (in thousands of dollars) Q is given by Q = 4x1/4y3/4 where x is the amount spent on labor and y is the amount...
-
If someone's Z-score for a variable was 0.67. Their score is a significant extreme score. Their score is not significant. O Their score is slightly above average. O Their score is an outlier.
-
Ammonia, NH 3 , reacts with chlorine gas, Cl 2 , to form ammonium chloride, NH 4 Cl, and nitrogen trichloride, NCl 3 . Write a balanced equation for this reaction.
-
What gave Schrdinger justification to think of an electron in an atom as a nebulous cloud?
-
Mendeleev arranged the elements by atomic mass and then separated them into various groups. What was his criterion for which elements were grouped together?
-
Which of the following are important roles of the government? a. Protecting property rights b. Providing a legal system c. Intervening when insufficient competition occurs in the marketplace d....
-
What is a value-added tax?
-
Social Security and Medicare are financed by a. personal income taxes. b. payroll taxes. c. excise taxes. d. corporation income taxes. e. none of the above taxes.

Study smarter with the SolutionInn App


