A solid mixture consists of 32.3 g of KNO3 (potassium nitrate) and 5.7 g of K2SO4 (potassium
Fantastic news! We've Found the answer you've been seeking!
Question:
A solid mixture consists of 32.3 g of KNO3 (potassium nitrate) and 5.7 g of K2SO4 (potassium sulfate). The mixture is added to 130. g of water.
Use this solubility curve (Figure 1) to answer the questions.
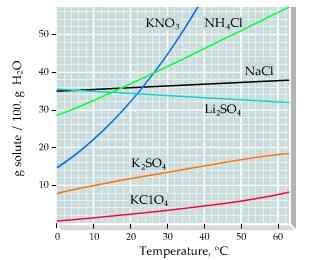
If the solution described in the introduction is cooled to 0 ?C what mass of K2SO4 will crystallize?
Related Book For 

Posted Date:




