Label each carbon atom with the appropriate geometry. trigonal planar CH2 trigonal planar CH trigonal planar...
Fantastic news! We've Found the answer you've been seeking!
Question:
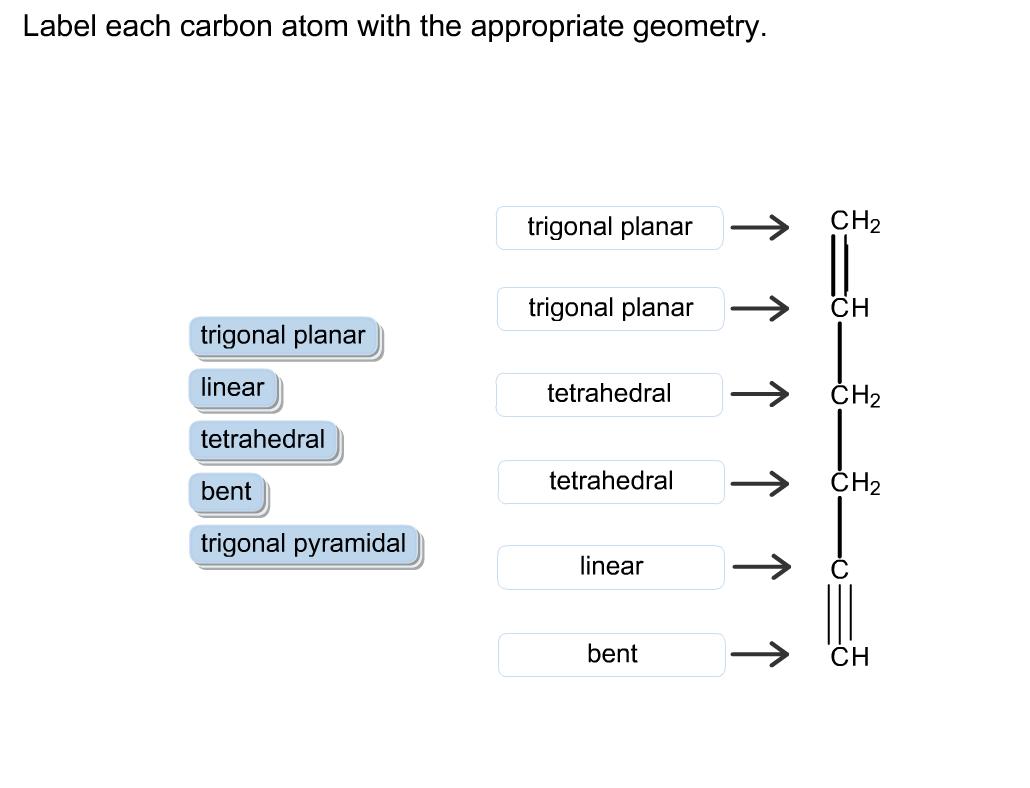
Transcribed Image Text:
Label each carbon atom with the appropriate geometry. trigonal planar CH2 trigonal planar CH trigonal planar linear tetrahedral ČH2 tetrahedral bent tetrahedral ČH2 trigonal pyramidal linear bent CH ↑ ↑ ↑ ↑ Label each carbon atom with the appropriate geometry. trigonal planar CH2 trigonal planar CH trigonal planar linear tetrahedral ČH2 tetrahedral bent tetrahedral ČH2 trigonal pyramidal linear bent CH ↑ ↑ ↑ ↑
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Label each hydrogen atom in the following compounds as primary (1°), secondary (2°), or tertiary (3°). (a) CH3 CH2 CH (CH3)2 (b) (CH3)3 CCH2CH3 (c) (CH3)2 CHCH (CH3) CH2 CH3 (d) (e) (f) ...
-
For each Fischer projection, label each asymmetric carbon atom as (R) or (S). (a)-(f) the structures in Problem 5-18 In problem 5-18 (a to f) (a) (b) (c) (d) (e) (f) (g) (h) (i) CH,OH CH,OH Br CH,OH...
-
Label each of the following statements about linear programming problems as true or false, and then justify your answer. (a) If a feasible solution is optimal but not a CPF solution, then infinitely...
-
Researchers at the Hospital for Sick Children in Toronto compared babies born to mothers with diabetes to babies born to mothers without diabetes (Conditioning and Hyperanalgesia in Newborns Exposed...
-
1. What does the phrase sustainable earnings mean? What types of earnings are not sustainable? 2. What are abnormal earnings? 3. Briefly describe the key features of the abnormal earnings approach to...
-
Dan Jacobs, production manager for GreenLife, invested in computer- controlled production machinery last year. He purchased the machinery from Superior Design at a cost of $ 3,000,000. A...
-
Consider a publicly held company whose products you are familiar with. Some examples might include: Access the companys public website and search for its most recent annual report. (Some companies...
-
David's Robotics Company manufactures three products. Profit computations for these products for the most recent year are as follows: David's Robotics traditionally allocates manufacturing overhead...
-
Problem 18-23 Price Discrimination, Customer Costs Jorell, Inc., manufactures and distributes a variety of labelers. Annual production of labelers averages 340,000 units. A large chain store...
-
1.Identify the three countries with the most favorable collection experience as measured by their receivables turnover ratios. RTR US = RTR Saudi Arabia = RTR UK = 2. Which country appears to have...
-
def wordlens (words): passprint('Starting wordlens test:')print('Test 1:')wordlens(['cat','mouse','bear','snake'])print('Test 2:')wordlens([])print('Test...
-
Find the equation of the circle which passes through the points (2,-2) and (3,4) and whose Centre lies on the line x+y=2. [10]
-
Rebecca decides to participate in an ordinary annuity investment vehicle by depositing $25,000 per year for 3 years. Assuming an annual compounding interest rate of 3%, what is the future value of...
-
Q5. Express the matrix B= -1 symmetric matrix. 1 -2 3 4 as the sum of a symmetric and skew -2-3]
-
What is the total rate of return of a bond if your investment horizon is 2 years, you are expected to reinvest coupons at 6% and your expected Yield To Maturity on the sale of the bond is 7% at the...
-
i) Find the equation of tangent and normal to the curve x/3 + y/3 = 2 at (1,1). [5] ii) The volume of the cube is increasing at a rate of 9 cubic cm/sec. How fast is the surface area increasing when...
-
I want answer with explanation... I'll upvote you... hello, I need a single question to be answered in operations management case. can you please help
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
Propose a mechanism involving a hydride shift or an alkyl shift for each solvolysis reaction. Explain how each rearrangement forms a more stable intermediate. (a) (b) (c) (d) CH I CH OCH CH,O CH CH...
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
Show the resonance forms for the enolate ions that result when the following compounds are treated with a strong base. (a) Ethyl acetoacetate (b) Pentane-2,4-dione (c) Ethyl a-cyanoacetate (d)...
-
Match the concept listed in items 1 through 8 with the most appropriate description listed in items a through h. Concept 1. Capital investment analysis 2. Type of environmental control 3. End-of-pipe...
-
Which one of the following statements regarding workforce diversity is inaccurate? a. Interpretation of accounting information is generally not dependent on the culture of the reporting entity. b....
-
For each of the following items, a through f, indicate whether the item represents (1) an advantage or (2) a disadvantage of downsizing. a. Decease in employee morale b. Incurrence of costs due to...

Study smarter with the SolutionInn App



