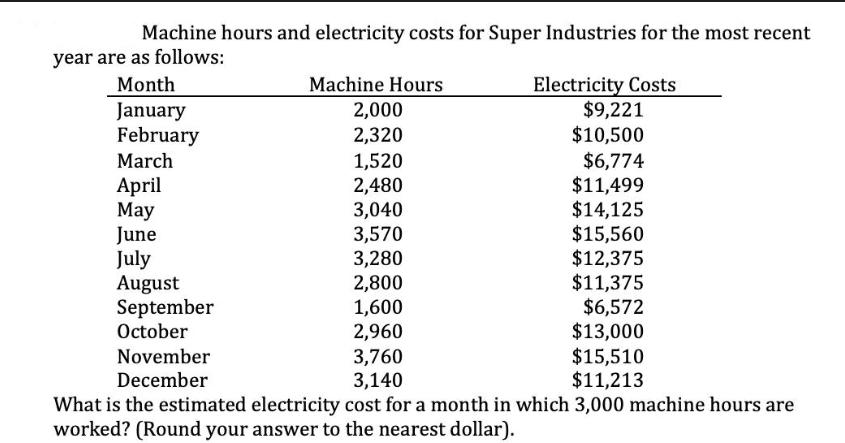
Machine hours and electricity costs for Super Industries for the most recent year are as follows:...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Machine hours and electricity costs for Super Industries for the most recent year are as follows: Month January February March April May June July August September October November December Machine Hours 2,000 2,320 1,520 2,480 3,040 3,570 3,280 2,800 1,600 2,960 3,760 3,140 Electricity Costs $9,221 $10,500 $6,774 $11,499 $14,125 $15,560 $12,375 $11,375 $6,572 $13,000 $15,510 $11,213 What is the estimated electricity cost for a month in which 3,000 machine hours are worked? (Round your answer to the nearest dollar). Machine hours and electricity costs for Super Industries for the most recent year are as follows: Month January February March April May June July August September October November December Machine Hours 2,000 2,320 1,520 2,480 3,040 3,570 3,280 2,800 1,600 2,960 3,760 3,140 Electricity Costs $9,221 $10,500 $6,774 $11,499 $14,125 $15,560 $12,375 $11,375 $6,572 $13,000 $15,510 $11,213 What is the estimated electricity cost for a month in which 3,000 machine hours are worked? (Round your answer to the nearest dollar).
Expert Answer:
Answer rating: 100% (QA)
I apologize for any confusion earlier It appears there was a typo in the electricity cost for ... View the full answer

Related Book For 

Managerial Accounting Decision Making and Motivating Performance
ISBN: 978-0137024872
1st edition
Authors: Srikant M. Datar, Madhav V. Rajan
Posted Date:
Students also viewed these accounting questions
-
Complete the following statements: The differential equation u' (t) + u(t) sint=1 is of Click for List The differential equation u' (t) + u(t) u(t)= cost is of Click for List The differential...
-
Two aquariums contain 10 liters of water each. Initially, the first aquarium contains no oxygen, but has a plant that produces oxygen at a rate of 0.1 mg/hour. The second aquarium initially has 0.3...
-
The following arrangement consists of an ideal transformer and an attenuator which attenuates by a factor of 0.8. An ac voltage Vwx = 100 V is applied across WX to get an open circuit voltage Vyz...
-
In Exercises 8486, use a graphing utility to graph f and g in the same [-8, 8, 1] by [-5, 5, 1] viewing rectangle. In addition, graph the line y = x and visually determine if f and g are inverses....
-
An air-filled toroidal solenoid has a mean radius of 15.0 cm and a cross-sectional area of 5.00 cm2. When the current is 120 A, the energy stored is 0.390 I. How many turns does the winding have?
-
Is this a right-, left-, or two-tailed test? A researcher is testing the effects of plant food on plant growth. Nine plants have been given the plant food. Another nine plants have not been given the...
-
In 2006, Danaher, Inc. announced its intent to acquire Sybron Dental Specialties, Inc., a manufacturer of dental products. Danaher indicated that it had agreed to pay \($47\) per share for all of...
-
The wireless phone manufacturing division of a consumer electronics company uses activity-based costing. For simplicity, assume that its accountants have identified only the following three...
-
All techniques, conflicting rankings Nicholson Roofing Materials, Inc., is considering two mutually exclusive projects, that both cost $ 1 5 0 , 0 0 0 . The company's board of directors has set a 4 -...
-
Buhler Industries is a farm implement manufacturer. Management is currently evaluating a proposal to build a plant that will manufacture lightweight tractors. Buhler plans to use a cost of capital of...
-
There are six households in a rural community. Each household earns $40,000 per year. Suppose that a new resident builds a mansion in the community and that the income in the new household is $4...
-
In project management, it is vital to have a management plan and effectively communicate that plan. The Project Quality Management Plan and the Project Communications Plan are two tools that you can...
-
If f(x)=(2x25) (7x+6), find: f'(x) = f'(5) = Submit Question
-
What are the different types of payment methods available to consumers and merchants? 2. What is the difference between a debit card and a credit card? 3. How do online payment systems like PayPal...
-
Use the table to provide examples of how educators can support children during these times to foster behaviours that respect diversity Educator strategies to support positive, inclusive and...
-
The CEO for MediBank is very happy to have you manage both the departments, namely customer service and human resources. However, to ensure a consistent strategic approach inline with the...
-
What are two timestamps required to measure lead time when using a Team Kanban board? Explain.
-
Given the table below, about how much force does the rocket engine exert on the 4.0 kg payload? Distance traveled with rocket engine firing (m) Payload final velocity (m/s) 500 320 490 310 1020 450...
-
Jackson Company is a manufacturer of rocking chairs. Each chair passes through the assembly department and finishing department. This problem focuses on the assembly department. Direct materials are...
-
Jiffy Mart has a kaizen (continuous improvement) approach to budgeting monthly activity costs for each month of 2013. Each successive month, the budgeted cost- driver rate decreases by 0.4% relative...
-
Green Thumb, Inc., provides lawn care and landscaping services to commercial clients. Green Thumb uses activity-based costing to bid on jobs and to evaluate their profitability. Green Thumb reports...
-
A \(2 \mathrm{~cm}\)-diameter, \(19 \mathrm{~cm}\)-long tube is placed touching a pool of liquid. The end away from the liquid pool \((\mathrm{z}=0.19 \mathrm{~m})\) is in an air stream (component C)...
-
\(\mathrm{NaCl}\) is crystallizing from an aqueous (water) liquid solution onto a crystal particle of pure \(\mathrm{NaCl}\) at \(18^{\circ} \mathrm{C}\). Assume particle growth is controlled by mass...
-
Solve Example 15-7 using the difference equation form of the Maxwell-Stefan equations. Example 15-7 Because naphthalene C10Hg melts at 80.2C, it is solid at room temperature. Naphthalene also has a...

Study smarter with the SolutionInn App


