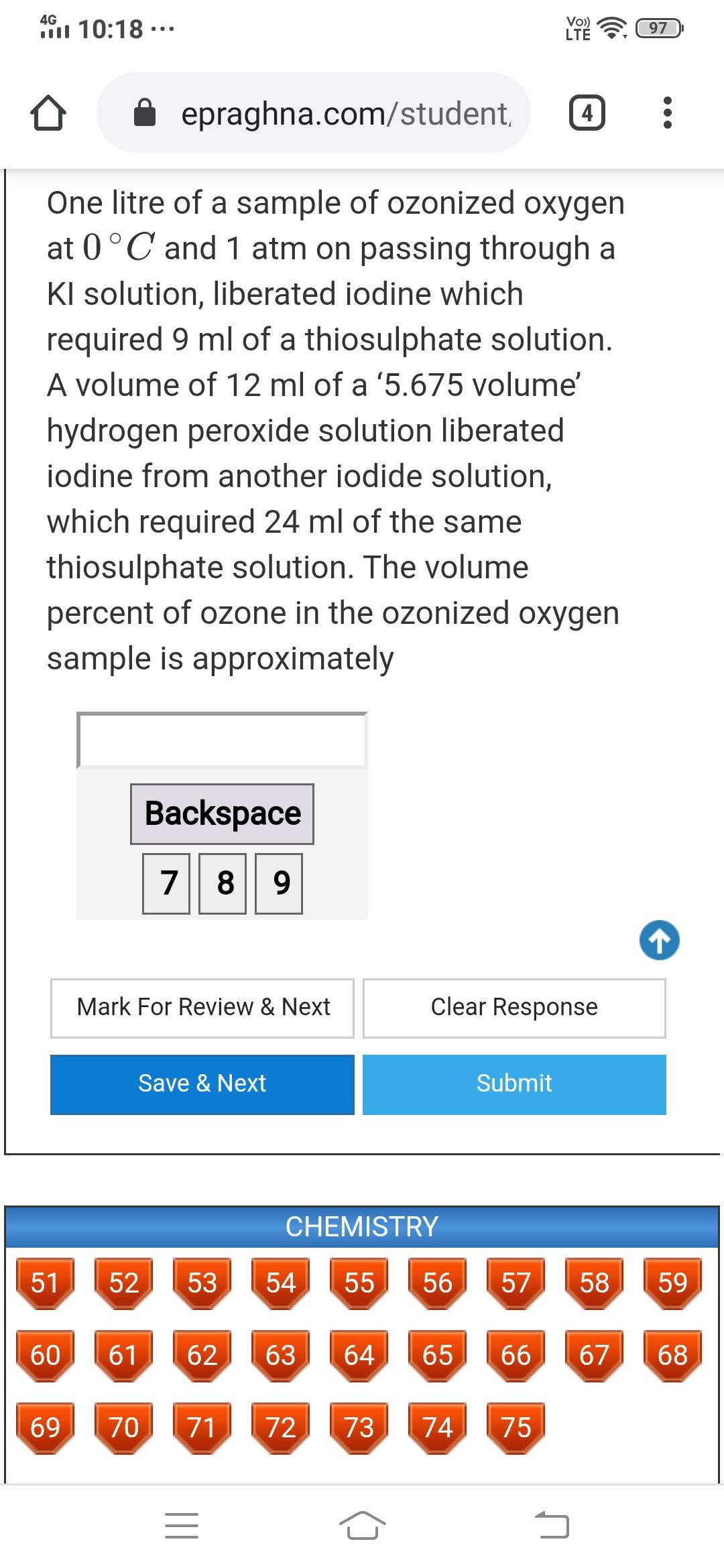
4G 10:18 .. Vo) LT 97 epraghna.com/student, 4 One litre of a sample of ozonized oxygen...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
4G 10:18 ·.. Vo) LTÉ 97 epraghna.com/student, 4 One litre of a sample of ozonized oxygen at 0°C and 1 atm on passing through a KI solution, liberated iodine which required 9 ml of a thiosulphate solution. A volume of 12 ml of a '5.675 volume' hydrogen peroxide solution liberated iodine from another iodide solution, which required 24 ml of the same thiosulphate solution. The volume percent of ozone in the ozonized oxygen sample is approximately Backspace 78 9 Mark For Review & Next Clear Response Save & Next Submit CHEMISTRY 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 LO II 4G 10:18 ·.. Vo) LTÉ 97 epraghna.com/student, 4 One litre of a sample of ozonized oxygen at 0°C and 1 atm on passing through a KI solution, liberated iodine which required 9 ml of a thiosulphate solution. A volume of 12 ml of a '5.675 volume' hydrogen peroxide solution liberated iodine from another iodide solution, which required 24 ml of the same thiosulphate solution. The volume percent of ozone in the ozonized oxygen sample is approximately Backspace 78 9 Mark For Review & Next Clear Response Save & Next Submit CHEMISTRY 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 LO II
Expert Answer:

Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Posted Date:
Students also viewed these chemistry questions
-
One litre of a sample of ozonized oxygen at 0C and 1 atm on passing through a Kl solution, liberated iodine which required 9 ml of a thiosulphate solution. A volume of 12 ml of a '5.675 volume'...
-
Carbon monoxide is a gas at 0 C and a pressure of 1.01 10 5 Pa. It is a diatomic gas, each of its molecules consisting of one carbon atom (atomic mass = 12.0 u) and one oxygen atom (atomic mass =...
-
Hydrogen gas and iodine vapor react to produce hydrogen iodide gas: Calculate the free-energy change G for the following two conditions, at 25oC. Which one is closer to equilibrium? Explain. a. The...
-
To complete the pipings in the newly constructed power plant, 60 men must work for 72 days. If all 60 men are employed at the start but the number working is decreased by 5 men at the end of each...
-
Propose mechanisms for the following reactions. Additional products may be formed, but your mechanism only needs to explain the products shown. (a) (b) (c) OEt EtOH heat Br OEt CH2OH H SO4 heat...
-
A manufacturer is preparing to set the price on a new action game. Demand is thought to depend on the price and is represented by the model: D = 2,000 3.5P The accounting department estimates that...
-
A stock has volatility \(\sigma=.30\) and a current value of \(\$ 36\). An American put option on this stock has a strike price of \(\$ 40\), and expiration is in 5 months. The interest rate is \(8...
-
The Green Company produces chemicals in a perfectly competitive market. The current market price is $40; the firms total cost is C = 100 + 4Q + Q2. a. Determine the firms profit-maximizing output....
-
You pay $1,000 to flip a two-sided, fair coin at the local fair. If you flip 'heads', you walk away with $3,000, a return of 200%. However, if you flip 'tails', you walk away with $250, a return of...
-
All Mopped Up Company has journalized the adjusting entries for the period ending December 31, 2018, and posted the adjustments to the following T-accounts. (Click the icon to view the T-accounts.)...
-
Assume a world with a tax rate of 34% and no bankruptcy. If WSB Inc. was an all-equity firm its return would be 9.2% per year. However, it does have bonds that currently have a YTM of 6.3%. By how...
-
A pinhole of diameter \(0.20 \mathrm{~mm}\) is illuminated with \(550 \mathrm{~nm}\) light. What is the width of the Airy disk on a screen \(1.5 \mathrm{~m}\) away?
-
A layer of oil \(\left(n_{\text {oil }}=1.48 ight) 0. 0100 \mathrm{~mm}\) thick is resting on a puddle of water \(\left(n_{\text {water }}=1.33 ight)\). If white light is incident on the oil, what is...
-
When a monochromatic beam of light passes through a thin slit, the \(n=9\) dark fringe is \(10^{\circ}\) beyond the adjacent \(n=8\) dark fringe. What is the ratio of the wavelength of the light to...
-
With same-day and one-hour delivery services from Amazon and Google commonplace, discuss whether this fulfillment model is long lasting or just a passing trend. Discuss the types of products that...
-
Electrons are accelerated from rest through a \(2.0-\mathrm{kV}\) potential difference. What are \((a)\) their speed after this acceleration and \((b)\) their wavelength? (c) If the accelerated...
-
Josh is a 7th grader who does not appear to be having major reading difficulties. From standardized testing over a period of three years (4th, 5th, and 6th grades), Josh's scores placed him with...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
A 5.0-cm-thick layer of oil is sandwiched between a 1.0-cm-thick sheet of glass and a 2.0-cm-thick sheet of polystyrene plastic. How long (in ns) does it take light incident perpendicular to the...
-
A camera takes a properly exposed photo with a 3.0-mm-diameter aperture and a shutter speed of 1/125 s. What is the appropriate aperture diameter for a 1/500 s shutter speed?
-
A 2.0 cm 3.0 cm rectangle lies in the xz-plane. What is the magnitude of the electric flux through the rectangle if a. E = (100i - 200k) N/C? b. E = (100i - 200j) N/C?
-
Attach LEDs to your system bus so that you can monitor its activity. For example, use an LED to monitor the read/write line on the bus.
-
Describe the role of these signals in a bus: a. R/W b. data ready c. clock
-
Draw a UML sequence diagram that shows a four-cycle handshake between a bus master and a device.

Study smarter with the SolutionInn App


