32 grams of (C,H) is placed in a container with 39 grams of oxygen gas. 28...
Fantastic news! We've Found the answer you've been seeking!
Question:

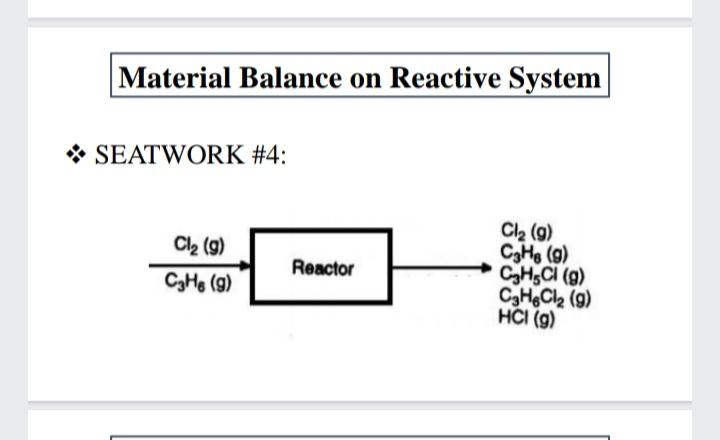

Transcribed Image Text:
32 grams of (C,H) is placed in a container with 39 grams of oxygen gas. 28 grams of CO, are collected at the end of the reaction. Calculate the percent yield. Material Balance on Reactive System * SEATWORK # 4: We have two reactions: Cl2 (g) + CsH6 (g) CHSCI (g) +HCI Cl: (g) + CsH, (g) C,H,Cl; (g) (а) (b) The species recovered after the reaction takes place for some time are listed in table below: Species Cl, CH6. propylene CH,CI, ally chloride C,H,Cl, propylene chloride MW gmol 141.0 651.0 42.08 76.53 112.99 4.6 24.5 HCI 4.6 Material Balance on Reactive System * SEATWORK # 4: Based on the product distribution assuming that no ally chlorides were present in the feed, calculate the following: a. How much Cl2 and C3H6 were fed to the reactor in gmol? b. What was the limiting reactant? c. What was the excess reactant? d. What was the fraction conversion of C,H6 to C3H;C1? e. What was the selectivity of C3H3C1 relative to C3H,C12? f. What was the yield of C3H;CI expressed in g of C;H;CI to the g of C;H, fed to the reactor? g. What was the extent of reaction of the first and second reactions? Material Balance on Reactive System * SEATWORK #4: Cl2 (g) CHe (g) CH,CI (g) CaHeCl2 (9) HCI (g) Cl2 (g) Reactor CHe (g) Estimation of Vapor Pressures SEATWORK #1: Calculate the grams of glucose that must be added to 2.85 kg of H2O at 65 degrees Celsius to lower the vapor pressure by 10.0 torr if the vapor pressure of pure water at this temperature is 188 torr. 32 grams of (C,H) is placed in a container with 39 grams of oxygen gas. 28 grams of CO, are collected at the end of the reaction. Calculate the percent yield. Material Balance on Reactive System * SEATWORK # 4: We have two reactions: Cl2 (g) + CsH6 (g) CHSCI (g) +HCI Cl: (g) + CsH, (g) C,H,Cl; (g) (а) (b) The species recovered after the reaction takes place for some time are listed in table below: Species Cl, CH6. propylene CH,CI, ally chloride C,H,Cl, propylene chloride MW gmol 141.0 651.0 42.08 76.53 112.99 4.6 24.5 HCI 4.6 Material Balance on Reactive System * SEATWORK # 4: Based on the product distribution assuming that no ally chlorides were present in the feed, calculate the following: a. How much Cl2 and C3H6 were fed to the reactor in gmol? b. What was the limiting reactant? c. What was the excess reactant? d. What was the fraction conversion of C,H6 to C3H;C1? e. What was the selectivity of C3H3C1 relative to C3H,C12? f. What was the yield of C3H;CI expressed in g of C;H;CI to the g of C;H, fed to the reactor? g. What was the extent of reaction of the first and second reactions? Material Balance on Reactive System * SEATWORK #4: Cl2 (g) CHe (g) CH,CI (g) CaHeCl2 (9) HCI (g) Cl2 (g) Reactor CHe (g) Estimation of Vapor Pressures SEATWORK #1: Calculate the grams of glucose that must be added to 2.85 kg of H2O at 65 degrees Celsius to lower the vapor pressure by 10.0 torr if the vapor pressure of pure water at this temperature is 188 torr.
Expert Answer:
Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these chemical engineering questions
-
A charge of 28.0 nC is placed in a uniform electric field that is directed vertically upward and has a magnitude of 4.00 X 104 V/m. What work is done by the electric force when the charge moves? (a)...
-
A person is placed in a large hollow metallic sphere that is insulated from ground. If a large charge is placed on the sphere, will the person be harmed upon touching the inside of the sphere?...
-
A flat circle of radius 18cm is placed in a uniform electric field of magnitude 5.8 X 102N/C. What is the electric flux through the circle when its face is? (a) Perpendicular to the field lines, (b)...
-
Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 C/m a) 100 C b) 100.2 C c) 99.67 C d) 101.2 C c) d) O a) b)
-
What is a radioactive tracer? Give an example of the use of such a tracer in chemistry.
-
What are the potential differences V AB and V BC ?
-
Carburettor is used for (a) S.I. engine (b) C.I. engine (c) Gas engine (d) none of the above
-
Gyro Company has the following totals from its operating budgets: Selling and administrative expenses budget . . . . . . . . . . . . . . . . . . . . . . . . $ 244,500 Cost of goods sold budget . . ....
-
Briefly describe the forensic psychology career field Explain what drew you to this field (forensic psychology) Examine the impact of past, current, and developing technology in your field (forensic...
-
Let's say that professor Sanford dies (this is not going to happen for another 20 years), and the estate of decedent- Sanford comes into existence. Also, assume that you are the adopted child of...
-
Discuss whether the below transactions are income to Angelo who is a coffee salesman, citing at least one case or section for each answer: 1. His base wage of $45,000. 2. Commissions paid to him by...
-
Discuss the roles of information technology, management information systems, and big data in a global companys decision-making processes.
-
Describe the various national policies that pertain to exports and imports.
-
Understand the elements of a countrys political environment that can impact global marketing activities.
-
Located along the shoreline of the Persian Gulf and emerging from the sands of the Arabian Desert is one of the worlds fastest-growing citiesDubai, in the United Arab Emirates. The population of...
-
Cotton is an integral part of clothing in societies and cultures across the world. It is crucial to cotton-producing countries as well as to those importing it. According to estimates, cotton...
-
. . Chapter 6 in CMH discusses impact assessments as part the change assessment process. Look at the discussion on high-level impact assessments. What would you add to this? What section could be...
-
Find the market equilibrium point for the following demand and supply functions. Demand: 2p = - q + 56 Supply: 3p - q = 34
-
Air at 16?C and atmospheric pressure enters a 1.25-cm-ID tube at 30 m/s. For an average wall temperature of 100?C, determine the discharge temperature of the air and the pressure drop if the pipe is...
-
A thin-walled horizontal copper tube of 0.5 cm OD is placed in a pool of water at atmospheric pressure and 100?C. Inside the tube, an organic vapor is condensing and the outside surface temperature...
-
A journal bearing has a clearance of 0.5 mm. The journal has a diameter of 100 mm and rotates at 3600 rpm within the bearing. The journal is lubricated by an oil having a density of 800 kg/m3, a...
-
James Halperin, co-chair of Heritage Auctions, was interviewed about his experiences auctioning rare coins. Noting that when the auction house first opened in the 1970s, I used to proofread every...
-
An editorial on Bloomberg. com criticized President Bidens Buy American policy. The editorial argued that the . . . problem is that protectionism sets the US at odds with its international...
-
An article in the Wall Street Journal explained that despite rising national security tensions between the United States and China in 2023, trade between the two nations was rising. China is a...

Study smarter with the SolutionInn App



