Merck & Company: Evaluating a Drug Licensing Opportunity Rich Kender, Vice President of Financial Evaluation &...
Fantastic news! We've Found the answer you've been seeking!
Question:




Transcribed Image Text:
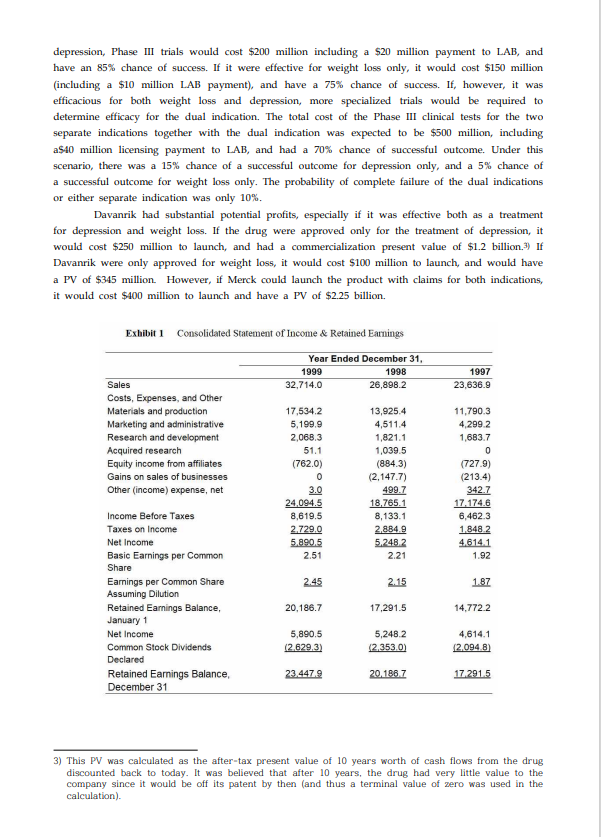
Merck & Company: Evaluating a Drug Licensing Opportunity Rich Kender, Vice President of Financial Evaluation & Analysis at Merck, was working with his team to decide whether his company should license Davanrik, a new drug with the potential to treat both depression and obesity. The small pharmaceutical concern that developed the drug, LAB Pharmaceuticals, lacked the resources to complete the lengthy approval process, manufacture the compound, and market the drug. LAB had approached Merck with an offer to license the compound. Under this agreement, Merck would be responsible for the approval of Davanrik, its manufacture, and its marketing. The company would pay LAB an initial fee, a royalty on all sales, and make additional payments as Davanrik completed each stage of the approval process. Merck In 2000, Merck & Co., Inc. was a global research-driven pharmaceutical company that discovers, develops, manufactures and markets a broad range of human and animal health products, directly and through its joint ventures, and provides pharmaceutical benefit management services (PBM) through Merck-Medco Managed Care. Since 1995, Merck had launched 15 new products including Vioxx for the treatment of osteoarthritis, Fosamax for the treatment of osteoporosis and Singulair for treating asthma. The Company earned $5.9 billion on 1999 sales of $32.7 billion, about a 20% increase from 1998. Exhibits 1 and 2 contain Merck's Income Statement and Balance Sheet. A handful of Merck's most popular drugs, Vasotec, Mevacor, Prinivil, and Pepcid, generated $5.7 billion in worldwide sales. The patents for these drugs, however, would expire by 2002. Once the patents expired, Merck anticipated that the sales of these drugs would decline substantially as generic substitutes became available. The only way to counter the loss of sales from drugs going off patent was to develop new drugs and constantly refresh the company's portfolio. The company develops new compounds primarily through internal research, but complements this through initiatives with biotechnology companies to ensure Merck is on the leading edge of select therapeutic categories. Davanrik LAB Pharmaceuticals originally developed Davanrik to treat depression. Antidepressant drugs work by affecting certain parts of the central nervous system. Various receptors in the human brain, when stimulated or blocked, create or inhibit various moods. The serotonin system controls nervousness, depression, insomnia, hunger, sexual dysfunction, nausea, and headaches. Through a combination of chemical compounds, the receptors in this system of cells can be stimulated or blocked to treat a patient with one or more of the given symptoms. Davanrik seemed not only to stimulate the receptor that promotes antidepression, but also to block the receptor that causes hunger. At the time of LAB's offer, Davanrik was in pre-clinical development, ready to enter the three-phase clinical approval process required for pharmaceuticals in the United States. In Phase I, the drug is given to a small number of healthy volunteers to test for safety. This usually takes about 1 years. In Phase II, a larger number of patients are tested to determine if the drug is effective in treating a certain condition and to measure potential side effects. This usually takes about 2 years. Finally, in Phase III, a large number of patients are tested for safety and efficacy. This phase takes about 3 years to complete. Exhibit 3 summarizes the FDA approval process. LAB Pharmaceuticals specializes in developing compounds for the treatment of neurological disorders. While the company was only 15 years old and though it had a few drugs in Phase II and Phase III testing, none had successfully completed the FDA approval process. In fact, the FDA had recently denied approval of another of LAB's compounds that had completed all three phases of clinical testing; LAB's stock price fell by over 30% in response to this decision. As a result, LAB was hesitant to issue additional equity to finance the testing of Davanrik and was seeking a larger pharmaceutical company to license the drug and provide LAB with some much-needed cash. The licensee would design, administer, and fund the clinical testing of the compound, its manufacturing and its marketing. The licensor, LAB, would receive an initial payment followed by additional payments as Davanrik completes each clinical testing phase. LAB would also receive a royalty on the eventual sales of Davanrik. Davanrik's Potential Cash Flows Rich Kender assembled a team to evaluate the potential profitability of Davanrik. Senior researchers evaluated scientific aspects of the compound, and marketers evaluated the market size, potential competition, and requirements to successfully launch the drug. Meanwhile, manufacturing managers determined the capital required to produce the drug, and people in Kender's own department built a financial analysis of the licensing decision. The evaluation team determined the costs and likelihood of completing each stage of the FDA approval process along with a forecast of profitability of the drug if it successfully completed the approval process. Overall, the approval process was expected to consume about seven years. LAB obtained a patent on the product which is estimated to have a remaining life, including all possible extensions, of 17 years. Therefore, the product would have a 10 year period of exclusivity, beginning in 7 years. Phase I Davanrik would be administered to 20-80 healthy people to determine if the drug was safe enough to continue into the efficacy stages of clinical testing. Phase I would take two years to complete. It was expected to cost $30 million, including an initial $5 million fee to LAB for licensing the drug. There was a 60% chance that Davanrik would successfully complete Phase I. Phase II In this phase, Davanrik would be given to 100 - 300 patient volunteers to determine its efficacy for treating depression and/or weight loss and to document any side effects. To complete the efficacy tests, Davanrik would have to demonstrate a statistically significant impact on patients suffering from depression, obesity, or both. The Merck team estimated a 10% probability that Phase II would show that Davanrik would be efficacious for depression only, a 15% probability for weight loss only, and a 5% probability that it would be efficacious for both depression and weight loss at the same time.) Like Phase 1, Phase II would require two years of clinical testing to complete. Phase II was expected to cost $40 million, including a $2.5 million licensing milestone payment to LAB.2) Phase III In Phase III, Davanrik would be administered to 1000-5000 volunteers to determine safety and efficacy in long term use. Because of the number of volunteers and nature of testing, this was the most costly of the phases and was expected to take three years to complete. The costs and probabilities of success depended on the outcome from Phase II. If Davanrik was effective for only 1) According to the FDA, a pharmaceutical must prove dual indications in addition to proving each indication separately if it wants to be able to claim therapeutic effects for people suffering from both disorders. 2) All cash flows are expressed as after-tax present values discounted to time zero, including capital expenditures. depression, Phase III trials would cost $200 million including a $20 million payment to LAB, and have an 85% chance of success. If it were effective for weight loss only, it would cost $150 million (including a $10 million LAB payment), and have a 75% chance of success. If, however, it was efficacious for both weight loss and depression, more specialized trials would be required to determine efficacy for the dual indication. The total cost of the Phase III clinical tests for the two separate indications together with the dual indication was expected to be $500 million, including a$40 million licensing payment to LAB, and had a 70 % chance of successful outcome. Under this scenario, there was a 15% chance of a successful outcome for depression only, and a 5% chance of a successful outcome for weight loss only. The probability of complete failure of the dual indications or either separate indication was only 10%. Davanrik had substantial potential profits, especially if it was effective both as a treatment for depression and weight loss. If the drug were approved only for the treatment of depression, it would cost $250 million to launch, and had a commercialization present value of $1.2 billion.3) If Davanrik were only approved for weight loss, it would cost $100 million to launch, and would have a PV of $345 million. However, if Merck could launch the product with claims for both indications, it would cost $400 million to launch and have a PV of $2.25 billion. Exhibit 1 Consolidated Statement of Income & Retained Earnings Year Ended December 31, 1999 32,714.0 Sales Costs, Expenses, and Other Materials and production Marketing and administrative Research and development Acquired research Equity income from affiliates Gains on sales of businesses Other (income) expense, net Income Before Taxes Taxes on Income Net Income Basic Earnings per Common Share Earnings per Common Share Assuming Dilution Retained Earnings Balance, January 1 Net Income Common Stock Dividends Declared Retained Earnings Balance, December 31 17,534.2 5,199.9 2,068.3 51.1 (762.0) 0 3.0 24,094.5 8,619.5 2.729.0 5.890.5 2.51 2.45 20,186.7 5,890.5 (2,629,3) 23.447.9 1998 26,898.2 13,925.4 4,511.4 1,821.1 1,039.5 (884.3) (2,147.7) 499.7 18,765.1 8,133.1 2,884.9 5.248.2 2.21 2.15 17,291.5 5,248.2 (2,353.0) 20.186.7 1997 23,636.9 11,790.3 4,299.2 1,68 0 (727.9) (213.4) 342.7 17.174.6 6,462.3 1,848.2 4.614.1 1.92 1.87 14,772.2 4,614.1 (2,094.8) 17.291.5 3) This PV was calculated as the after-tax present value of 10 years worth of cash flows from the drug discounted back to today. It was believed that after 10 years, the drug had very little value to the company since it would be off its patent by then (and thus a terminal value of zero was used in the calculation). 0 Exhibit 2 Consolidated Balance Sheet Assets Current Assets Cash and cash equivalents Short-term investments Accounts receivable Inventories Years Prepaid expenses and taxes Total current assets Investments Property. Plant and Equipment (at cost) Land & Buildings Machinery, equipment and office furnishings Construction in progress Less allowance for depreciation Goodwill and Other intangibles Other Assets Liabilities and Stockholders Equity Current Liabilities Accounts payable and accrued liabilities Loans payable and current portion of long-term Income tax payable Dividends payable Total current liabilities Long-Term Debt Deferred Income Taxes and Noncurrent Liabilities Minority interests Stockholder's Equity Common Stock Other Paid-in capital Retained earnings Accumulated Other Comprehensive Income (loss) 2 Exhibit 3 Compound Success Rates by Stage COMPOUND SUCCESS RATES BY STAGES Less treasury stock, at cost Total stockholders' equity Discovery (2-10 Years) Preclinical Testing Laboratory and animal testing Phase III 1,000-5,000 pasient volunteers used to monitor adverse reactions to long-term use 10 Year Ended December 31, 1998 1999 2.021.9 1.180.5 4.000 0 2,846.9 1.120.9 11.259.2 4.761.5 4,725.0 7.385.7 2.236.3 14,347.0 4.670.3 9.676.7 7,584.2 2.353.3 35.634.9 T 12 4,158.7 2.859.0 1,064.1 677.0 3,143.9 7,030.1 3.460.5 Phase I 20-80 healthy volunteers used to determine safety and dosage Phase II 100-300 patient volunteers used to look for efficacy and side effects 29.7 5,920.5 23,447.9 8.1 29.406 2 16.164.6 13.241.6 30 634.9 FDA Review! Approval Additional Post-marketing Testing T 14 16 2,606.2 749.5 3,374.1 2,623.9 874.8 10.228.5 3,607.7 3,892.8 6,211.7 1.782 1 11,886.6 4.042.8 7.843.8 8.287.2 1.886.2 31.853.4 3,682.1 624.2 1,125.1 637.4 6.068.8 3,220.8 6,057.0 3.705.0 29.7 5,614.5 20,186.7 (21.3) 25.809.6 13.007.8 12.801.8 31.843.4 Compound Success Bates by Stage 5,000-10,000 screened 250 enter predinical testing enter dinical testing Approved by the FDA Study Questions Case analysis might be guided by the following questions: 1. How has Merck been able to achieve substantial returns to capital given the large costs and lengthy time to develop drug? 2. Build a decision tree that shows the cash flows and probabilities at all stages of the FDA approval process. 3. Should Merck bid to license Davanrik? How much should they pay? 4. What is the expected value of the licensing arrangement to LAB? Assume a 5% royalty fee on any cash flows that Merck receives from Davanrik after a successful launch. 5. How would your analysis change if the costs of launching Davanrik for weight loss were $225 million instead of $100 million as given in the case? Merck & Company: Evaluating a Drug Licensing Opportunity Rich Kender, Vice President of Financial Evaluation & Analysis at Merck, was working with his team to decide whether his company should license Davanrik, a new drug with the potential to treat both depression and obesity. The small pharmaceutical concern that developed the drug, LAB Pharmaceuticals, lacked the resources to complete the lengthy approval process, manufacture the compound, and market the drug. LAB had approached Merck with an offer to license the compound. Under this agreement, Merck would be responsible for the approval of Davanrik, its manufacture, and its marketing. The company would pay LAB an initial fee, a royalty on all sales, and make additional payments as Davanrik completed each stage of the approval process. Merck In 2000, Merck & Co., Inc. was a global research-driven pharmaceutical company that discovers, develops, manufactures and markets a broad range of human and animal health products, directly and through its joint ventures, and provides pharmaceutical benefit management services (PBM) through Merck-Medco Managed Care. Since 1995, Merck had launched 15 new products including Vioxx for the treatment of osteoarthritis, Fosamax for the treatment of osteoporosis and Singulair for treating asthma. The Company earned $5.9 billion on 1999 sales of $32.7 billion, about a 20% increase from 1998. Exhibits 1 and 2 contain Merck's Income Statement and Balance Sheet. A handful of Merck's most popular drugs, Vasotec, Mevacor, Prinivil, and Pepcid, generated $5.7 billion in worldwide sales. The patents for these drugs, however, would expire by 2002. Once the patents expired, Merck anticipated that the sales of these drugs would decline substantially as generic substitutes became available. The only way to counter the loss of sales from drugs going off patent was to develop new drugs and constantly refresh the company's portfolio. The company develops new compounds primarily through internal research, but complements this through initiatives with biotechnology companies to ensure Merck is on the leading edge of select therapeutic categories. Davanrik LAB Pharmaceuticals originally developed Davanrik to treat depression. Antidepressant drugs work by affecting certain parts of the central nervous system. Various receptors in the human brain, when stimulated or blocked, create or inhibit various moods. The serotonin system controls nervousness, depression, insomnia, hunger, sexual dysfunction, nausea, and headaches. Through a combination of chemical compounds, the receptors in this system of cells can be stimulated or blocked to treat a patient with one or more of the given symptoms. Davanrik seemed not only to stimulate the receptor that promotes antidepression, but also to block the receptor that causes hunger. At the time of LAB's offer, Davanrik was in pre-clinical development, ready to enter the three-phase clinical approval process required for pharmaceuticals in the United States. In Phase I, the drug is given to a small number of healthy volunteers to test for safety. This usually takes about 1 years. In Phase II, a larger number of patients are tested to determine if the drug is effective in treating a certain condition and to measure potential side effects. This usually takes about 2 years. Finally, in Phase III, a large number of patients are tested for safety and efficacy. This phase takes about 3 years to complete. Exhibit 3 summarizes the FDA approval process. LAB Pharmaceuticals specializes in developing compounds for the treatment of neurological disorders. While the company was only 15 years old and though it had a few drugs in Phase II and Phase III testing, none had successfully completed the FDA approval process. In fact, the FDA had recently denied approval of another of LAB's compounds that had completed all three phases of clinical testing; LAB's stock price fell by over 30% in response to this decision. As a result, LAB was hesitant to issue additional equity to finance the testing of Davanrik and was seeking a larger pharmaceutical company to license the drug and provide LAB with some much-needed cash. The licensee would design, administer, and fund the clinical testing of the compound, its manufacturing and its marketing. The licensor, LAB, would receive an initial payment followed by additional payments as Davanrik completes each clinical testing phase. LAB would also receive a royalty on the eventual sales of Davanrik. Davanrik's Potential Cash Flows Rich Kender assembled a team to evaluate the potential profitability of Davanrik. Senior researchers evaluated scientific aspects of the compound, and marketers evaluated the market size, potential competition, and requirements to successfully launch the drug. Meanwhile, manufacturing managers determined the capital required to produce the drug, and people in Kender's own department built a financial analysis of the licensing decision. The evaluation team determined the costs and likelihood of completing each stage of the FDA approval process along with a forecast of profitability of the drug if it successfully completed the approval process. Overall, the approval process was expected to consume about seven years. LAB obtained a patent on the product which is estimated to have a remaining life, including all possible extensions, of 17 years. Therefore, the product would have a 10 year period of exclusivity, beginning in 7 years. Phase I Davanrik would be administered to 20-80 healthy people to determine if the drug was safe enough to continue into the efficacy stages of clinical testing. Phase I would take two years to complete. It was expected to cost $30 million, including an initial $5 million fee to LAB for licensing the drug. There was a 60% chance that Davanrik would successfully complete Phase I. Phase II In this phase, Davanrik would be given to 100 - 300 patient volunteers to determine its efficacy for treating depression and/or weight loss and to document any side effects. To complete the efficacy tests, Davanrik would have to demonstrate a statistically significant impact on patients suffering from depression, obesity, or both. The Merck team estimated a 10% probability that Phase II would show that Davanrik would be efficacious for depression only, a 15% probability for weight loss only, and a 5% probability that it would be efficacious for both depression and weight loss at the same time.) Like Phase 1, Phase II would require two years of clinical testing to complete. Phase II was expected to cost $40 million, including a $2.5 million licensing milestone payment to LAB.2) Phase III In Phase III, Davanrik would be administered to 1000-5000 volunteers to determine safety and efficacy in long term use. Because of the number of volunteers and nature of testing, this was the most costly of the phases and was expected to take three years to complete. The costs and probabilities of success depended on the outcome from Phase II. If Davanrik was effective for only 1) According to the FDA, a pharmaceutical must prove dual indications in addition to proving each indication separately if it wants to be able to claim therapeutic effects for people suffering from both disorders. 2) All cash flows are expressed as after-tax present values discounted to time zero, including capital expenditures. depression, Phase III trials would cost $200 million including a $20 million payment to LAB, and have an 85% chance of success. If it were effective for weight loss only, it would cost $150 million (including a $10 million LAB payment), and have a 75% chance of success. If, however, it was efficacious for both weight loss and depression, more specialized trials would be required to determine efficacy for the dual indication. The total cost of the Phase III clinical tests for the two separate indications together with the dual indication was expected to be $500 million, including a$40 million licensing payment to LAB, and had a 70 % chance of successful outcome. Under this scenario, there was a 15% chance of a successful outcome for depression only, and a 5% chance of a successful outcome for weight loss only. The probability of complete failure of the dual indications or either separate indication was only 10%. Davanrik had substantial potential profits, especially if it was effective both as a treatment for depression and weight loss. If the drug were approved only for the treatment of depression, it would cost $250 million to launch, and had a commercialization present value of $1.2 billion.3) If Davanrik were only approved for weight loss, it would cost $100 million to launch, and would have a PV of $345 million. However, if Merck could launch the product with claims for both indications, it would cost $400 million to launch and have a PV of $2.25 billion. Exhibit 1 Consolidated Statement of Income & Retained Earnings Year Ended December 31, 1999 32,714.0 Sales Costs, Expenses, and Other Materials and production Marketing and administrative Research and development Acquired research Equity income from affiliates Gains on sales of businesses Other (income) expense, net Income Before Taxes Taxes on Income Net Income Basic Earnings per Common Share Earnings per Common Share Assuming Dilution Retained Earnings Balance, January 1 Net Income Common Stock Dividends Declared Retained Earnings Balance, December 31 17,534.2 5,199.9 2,068.3 51.1 (762.0) 0 3.0 24,094.5 8,619.5 2.729.0 5.890.5 2.51 2.45 20,186.7 5,890.5 (2,629,3) 23.447.9 1998 26,898.2 13,925.4 4,511.4 1,821.1 1,039.5 (884.3) (2,147.7) 499.7 18,765.1 8,133.1 2,884.9 5.248.2 2.21 2.15 17,291.5 5,248.2 (2,353.0) 20.186.7 1997 23,636.9 11,790.3 4,299.2 1,68 0 (727.9) (213.4) 342.7 17.174.6 6,462.3 1,848.2 4.614.1 1.92 1.87 14,772.2 4,614.1 (2,094.8) 17.291.5 3) This PV was calculated as the after-tax present value of 10 years worth of cash flows from the drug discounted back to today. It was believed that after 10 years, the drug had very little value to the company since it would be off its patent by then (and thus a terminal value of zero was used in the calculation). 0 Exhibit 2 Consolidated Balance Sheet Assets Current Assets Cash and cash equivalents Short-term investments Accounts receivable Inventories Years Prepaid expenses and taxes Total current assets Investments Property. Plant and Equipment (at cost) Land & Buildings Machinery, equipment and office furnishings Construction in progress Less allowance for depreciation Goodwill and Other intangibles Other Assets Liabilities and Stockholders Equity Current Liabilities Accounts payable and accrued liabilities Loans payable and current portion of long-term Income tax payable Dividends payable Total current liabilities Long-Term Debt Deferred Income Taxes and Noncurrent Liabilities Minority interests Stockholder's Equity Common Stock Other Paid-in capital Retained earnings Accumulated Other Comprehensive Income (loss) 2 Exhibit 3 Compound Success Rates by Stage COMPOUND SUCCESS RATES BY STAGES Less treasury stock, at cost Total stockholders' equity Discovery (2-10 Years) Preclinical Testing Laboratory and animal testing Phase III 1,000-5,000 pasient volunteers used to monitor adverse reactions to long-term use 10 Year Ended December 31, 1998 1999 2.021.9 1.180.5 4.000 0 2,846.9 1.120.9 11.259.2 4.761.5 4,725.0 7.385.7 2.236.3 14,347.0 4.670.3 9.676.7 7,584.2 2.353.3 35.634.9 T 12 4,158.7 2.859.0 1,064.1 677.0 3,143.9 7,030.1 3.460.5 Phase I 20-80 healthy volunteers used to determine safety and dosage Phase II 100-300 patient volunteers used to look for efficacy and side effects 29.7 5,920.5 23,447.9 8.1 29.406 2 16.164.6 13.241.6 30 634.9 FDA Review! Approval Additional Post-marketing Testing T 14 16 2,606.2 749.5 3,374.1 2,623.9 874.8 10.228.5 3,607.7 3,892.8 6,211.7 1.782 1 11,886.6 4.042.8 7.843.8 8.287.2 1.886.2 31.853.4 3,682.1 624.2 1,125.1 637.4 6.068.8 3,220.8 6,057.0 3.705.0 29.7 5,614.5 20,186.7 (21.3) 25.809.6 13.007.8 12.801.8 31.843.4 Compound Success Bates by Stage 5,000-10,000 screened 250 enter predinical testing enter dinical testing Approved by the FDA Study Questions Case analysis might be guided by the following questions: 1. How has Merck been able to achieve substantial returns to capital given the large costs and lengthy time to develop drug? 2. Build a decision tree that shows the cash flows and probabilities at all stages of the FDA approval process. 3. Should Merck bid to license Davanrik? How much should they pay? 4. What is the expected value of the licensing arrangement to LAB? Assume a 5% royalty fee on any cash flows that Merck receives from Davanrik after a successful launch. 5. How would your analysis change if the costs of launching Davanrik for weight loss were $225 million instead of $100 million as given in the case?
Expert Answer:
Answer rating: 100% (QA)
Here is my analysis of Mercks decision to license Davanrik from LAB Pharmaceuticals 1 How has Merck been able to achieve substantial returns to capita... View the full answer

Related Book For 

Canadian Business & the Law
ISBN: 978-0176501624
4th edition
Authors: Dorothy DuPlessis, Shannnon o'Byrne, Steven Enman, Sally Gunz
Posted Date:
Students also viewed these finance questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Dickens, Kristen, is enrolled as a doctoral student in the Counselor Education at the University of New Orleans. She is a registered counselor intern in the state of Louisiana and works at a...
-
Hanover Inc. expects to have a cash balance of 546.000 on January 1, 2017. Relevant monthly budget Prepare cash budget, data for the first two months of 2017 are as follows: (10 4) Al' 1. Collections...
-
Refer to exhibit 2-5 and prepare a monthly budget to cover the next six months. Pay particular attention to variable but non-repetitive expenses, such as tuition fees, textbooks, and...
-
(a) If R is a ring. then so is R op . where R o p is defined as follows. The underlying set of R op is precisely Rand addition in R o p coincides with addition in R. Multiplication in R op , denoted...
-
Consider two alternatives, each of which will accomplish the same EPA-mandated pollution control. Using an incremental IRR analysis, determine the preferred alternative assuming MARR is 20...
-
Sandi Scott obtained a patent on a small electronic device and organized Scott Products, Inc., to produce and sell the device. During the first month of operations, the device was very well received...
-
A professor designing a class demonstration connects a parallel-plate capacitor to a battery, so that the potential difference between the plates is 255 V. Assume a plate separation of d = 1.72 cm...
-
Coverall Inc. produces and sells a unique type of case for a standard size tablet computer that is guaranteed waterproof but still allows for regular functionality of the tablet. The company has just...
-
Consider the Region R bounded by the curves Y=I, X=1, x^2+y^2 =1 i. Find the Centroid R (ii) Find the Centroid of 3 dimensional fold made by rotating R about the y-axis
-
Two friends, Zach and Cody, are setting up a business that involves buying property, renovating the properties and selling them for a profit. The business will also offer design solutions to existing...
-
You may have heard that whale songs travel nearly half way around the earth, so that whales have lon-distance communication. Given that the speed of sound in seawater is 1500 m/s (that's "meters per...
-
Your company is scheduled to conduct a crucial meeting regarding its future business plans. This meeting will include series of intense negotiation sessions with a few European and Middle Eastern...
-
Java Question QUESTION 1 Saved 20 points The ParkCity company plans to build an application to let parking lot owners manage their parking llots for daily commuting customers. A parking lot owner can...
-
An essential part of being a special educator is writing and implementing IEPs and lesson plans associated with goals on IEPs. Due to legal requirements, special educators should understand the steps...
-
Write a research paper discussing the concept of risk modeling.
-
You are a Loan Officer with an Investment Bank. Today you need to set your lending parameters. They are: LTV: 55% 10 Year T-Bill: TBD Rate Markup: 300 Basis Points Term: 30 Years Amortization: 30...
-
Is alcohol and drug testing in the workplace permissible? Explain.
-
Does the sharing of profits result in the creation of a partnership? Explain.
-
What is passing off and what practices was this tort created to prevent?
-
Internet Inhand Ltd began producing netbooks on 1 July 2019. A unit of production passes through two processes manufacturing and finishing. Production data for the month of July are presented below....
-
Tsoulos, Tsoulakis and Associates is a small firm of architectural consultants. At 1 July 2018, three architects other than the principals, Tony Tsoulos and Maria Tsoulakis, are employed. The...
-
Melaleuca Manufacturing Ltd produces timber felling machines for the forestry industry around the world. Its two machines are the Tree Toppler, which cuts down trees and clears undergrowth, and the...

Study smarter with the SolutionInn App


