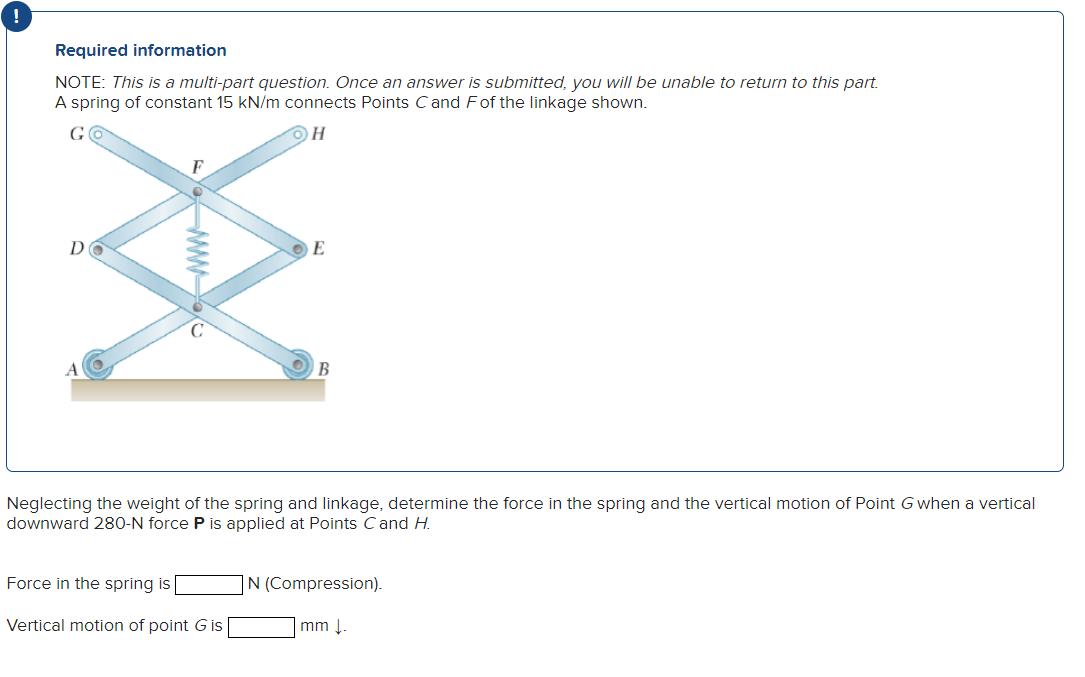
Required information NOTE: This is a multi-part question. Once an answer is submitted, you will be...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Required information NOTE: This is a multi-part question. Once an answer is submitted, you will be unable to return to this part. A spring of constant 15 kN/m connects Points C and Fof the linkage shown. G H D F Force in the spring is E Neglecting the weight of the spring and linkage, determine the force in the spring and the vertical motion of Point G when a vertical downward 280-N force P is applied at Points C and H. Vertical motion of point G is B N (Compression). mm 1. Required information NOTE: This is a multi-part question. Once an answer is submitted, you will be unable to return to this part. A spring of constant 15 kN/m connects Points C and Fof the linkage shown. G H D F Force in the spring is E Neglecting the weight of the spring and linkage, determine the force in the spring and the vertical motion of Point G when a vertical downward 280-N force P is applied at Points C and H. Vertical motion of point G is B N (Compression). mm 1.
Expert Answer:
Answer rating: 100% (QA)
501 given data or now from fig deflection relation and or and app... View the full answer

Related Book For 

Managerial Accounting Creating Value in a Dynamic Business Environment
ISBN: 978-0078025662
10th edition
Authors: Ronald Hilton, David Platt
Posted Date:
Students also viewed these mechanical engineering questions
-
RTL is a family owned and operated business that prints flyers and banners. It has been in operation for over 20 years and is being passed on to the next generation. Profits from the last two years...
-
Bels Bakery (BB) is a family owned business. In 2010 it recorded a $3 million operating loss. Apparently, 50% of the losses stemmed from a failed acquisition. With short term interest rates at 5%,...
-
Seattle Telecom, Inc. manufactures telecommunications equipment. The company has always been production oriented and sells its products through agents. Agents are paid a commission of 15 percent of...
-
We listed management of expectations as one of the Feds new unconventional monetary policy tools. Explain what this new policy tool does. List the potential problem of this policy tool.
-
A 55 year old woman applied online for a financial analyst position and other jobs with an employer. The woman had a Masters degree in accounting and a M.B.A. in finance, in addition to 15 years of...
-
A wind turbine has \(12,000 \mathrm{~kg}\) blades that are \(38 \mathrm{~m}\) long. The blades spin at \(22 \mathrm{rpm}\). If we model a blade as a point mass at the midpoint of the blade, what is...
-
Suzanne Luken opened a medical practice titled Suzanne Luken M.D., Inc. During March, the first month of operations, the business experienced the following events: Requirement 1.Analyze the effects...
-
1. Why has there been a corporate governance "revolution" throughout the world recently? 2. What are the main areas of deficiencies as pointed out by corporate governance reformers? 3. Representing...
-
whiuch oral hypoglycemic agent would be safest for an older patient if hypoglycemia is a mojor concern? tolbutamide glipizide metformin glimepiride
-
You are about to begin a summer internship in Logistics working for a company Express Mover that provides packaging and shipping services to customers moving large amounts of materials from 2,000...
-
Define the characteristics of a Service that distinguishes it from a product. There are two kinds of service companies, machine based and people based, describe and give an example of each.
-
At the beginning of Year 4, Hardin Company had 310,000 shares of $10 par common stock outstanding. During the year, it engaged in the following transactions related to its common stock: March 1...
-
i). How cost accounting is useful where financial accounting could not be answered. Give any example to support your answer. ii) A furniture manufacturing firm with the designing beds and related...
-
Consider the Grossman model of demand for health and answer the following questions: 1. Assume that price of medical care increases. How would this impact the optimal health stock of optimal health...
-
Lily Corporation manufactures a single product. The standard cost per unit of product is shown below. Direct materials-1 pound plastic at $8 per pound $8.00 Direct labor-2.0 hours at $12.50 per hour...
-
Choose a any topic related to last month marketing events. Any marketing events in September. Gather data points related to that event and determine how successful that event was. Share what you...
-
Lesley works for a CPA firm and is working on an audit for a large cattle ranch. She has contracted with an organization who has a drone that can fly over the ranch and count the heads of cattle for...
-
Archangel Corporation prepared the following variance report. Instructions Fill in the appropriate amounts or letters for the question marks in the report. ARCHANGEL CORPORATION Variance...
-
Suppose you paid $ 75 for a ticket to see your universitys football team compete in a bowl game. Some-one offered to buy your ticket for $ 100, but you decided to go to the game. Required: 1. What...
-
Refer to the preceding exercise. The divisional controller at Serendipity Sounds Minneapolis Division has estimated the following cost data for the divisions sound systems. (Assume there are no fixed...
-
Mighty Muffler, Inc. operates an automobile service facility that specializes in replacing mufflers on com-pact cars. The following table shows the costs incurred during a month when 700 mufflers...
-
Calculate the acentric factor for ethanol. The vapour pressure of ethanol can be estimated from the following equation: \[ \log _{10} P^{\text {Sat }}=8.1122-\frac{1592.864}{t+226.184} \] where...
-
For liquid acetone at \(20^{\circ} \mathrm{C}\) and \(1 \mathrm{bar}\), \[ \beta=1.487 \times 10^{-3} /{ }^{\circ} \mathrm{C} \quad \alpha=62 \times 10^{-6} / \mathrm{bar} \quad V=1.287...
-
Determine the molar volume of ammonia vapour and ammonia liquid at \(321.55 \mathrm{~K}\) and 1.95 \(\mathrm{MPa}\). Ammonia is assumed to follow van der Waals equation of state.

Study smarter with the SolutionInn App


