Determine the amount of heat removed from the reactor per mole of CH4 fed to the reactor.
Fantastic news! We've Found the answer you've been seeking!
Question:
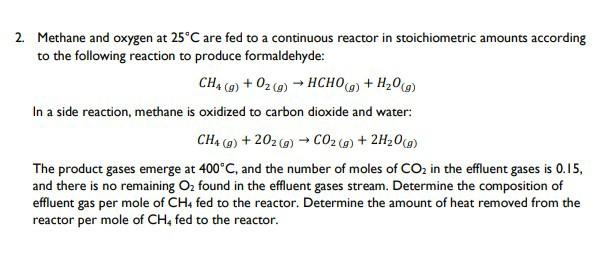
Determine the amount of heat removed from the reactor per mole of CH4 fed to the reactor.
Transcribed Image Text:
2. Methane and oxygen at 25°C are fed to a continuous reactor in stoichiometric amounts according to the following reaction to produce formaldehyde: CH4 (g) + O2(g) → HCHO(g) + H₂O(g) In a side reaction, methane is oxidized to carbon dioxide and water: CH4 (g) +202 (g) → CO₂ (g) + 2H₂O(g) The product gases emerge at 400°C, and the number of moles of CO₂ in the effluent gases is 0.15, and there is no remaining O₂ found in the effluent gases stream. Determine the composition of effluent gas per mole of CH4 fed to the reactor. Determine the amount of heat removed from the reactor per mole of CH4 fed to the reactor. 2. Methane and oxygen at 25°C are fed to a continuous reactor in stoichiometric amounts according to the following reaction to produce formaldehyde: CH4 (g) + O2(g) → HCHO(g) + H₂O(g) In a side reaction, methane is oxidized to carbon dioxide and water: CH4 (g) +202 (g) → CO₂ (g) + 2H₂O(g) The product gases emerge at 400°C, and the number of moles of CO₂ in the effluent gases is 0.15, and there is no remaining O₂ found in the effluent gases stream. Determine the composition of effluent gas per mole of CH4 fed to the reactor. Determine the amount of heat removed from the reactor per mole of CH4 fed to the reactor.
Expert Answer:
Answer rating: 100% (QA)
0 CHY 20 25C CHyzl with Imole CO with 015 mole cos 102385 HO 210 4100C 3 I mole o... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these mechanical engineering questions
-
Balance this redox reaction by inspection. C18H38 + O2 ( CO2 + H2O
-
Determine the mole fraction of sodium that ionizes according to the reaction Na Na+ + e - at 2000 K and 0.8 atm (KP = 0.668 for this reaction).
-
Determine the mole fraction of argon that ionizes according to the reaction Ar Ar+ + e- at 10,000 K and 0.35 atm (KP = 0.00042 for this reaction).
-
What other advice can you offer owners of seasonal businesses about coping with the effects of their companies highly irregular sales patterns? About managing cash flow in general?
-
In 1994, the U.S. postal service put a picture of rodeo rider Ben Pickett, not the rodeo star, Bill Pickett, whom it meant to honor, on a stamp. It printed 150,000 sheets. Recognizing its error, it...
-
The cone-and-plate viscometer (see Fig. 2B.11). A cone-and-plate viscometer consists of a stationary flat plate and an inverted cone, whose apex just contacts the plate. The liquid whose viscosity is...
-
Does Fairmont have any contactor personnel whose have terminated but are being paid through payroll after termination (e.g., ghost employees)?
-
A job cost sheet of Sandoval Company is given below. Instructions (a) Answer the following questions. (1) What are the source documents for direct materials, direct labor, and manufacturing overhead...
-
2. An engineer is planning for a 15-year retirement. In order to supplement his pension and offset the anticipated effects of inflation, he intends to withdraw $6000 at the end of the first year, and...
-
Wilhelmina is a trader whose financial year ends on 31 March. Her trial balance at 31 March 2004 was as follows. Further information 1. Stock at 31 March 2004 cost $16.000. 2. The loan was received...
-
! Required information [The following information applies to the questions displayed below.] Andrea would like to organize SHO as either an LLC (taxed as a sole proprietorship) or a C corporation. In...
-
Identify and discuss the social factors of organizational behavior that influence power and negotiations.
-
Find the Vision, Mission, and Values for a company of your choice and provide those to the class here. Discuss the importance of a company Vision, Mission, and Values and how that relates to the...
-
What are the main elements or parts of the Australian IR system
-
learned how training and development are integrated into an organization's career development program. Reflect on your experience with career development programs. As a training and development...
-
Individual incentive plans are less preferable than group incentives and company-wide incentives." Support and explain your answer citing credible academic/business sources.
-
On June 1, 2020, the Alma Corp. acquired a piece of land with a building on it for $1,400,000. The land was worth $250,000, and the building was worth $1,150,000. Alma paid for this purchase by...
-
Write the general quadratic equation y2 - 8y - 4x + 28 = 0 in standard form. Determine the vertex, focus, and directrix of the parabola defined by this equation. Sketch a graph.
-
Assume we fill a spherical balloon from a bottle of helium gas. The helium gas provides work PdV that stretches the balloon material S dA and pushes back the atmosphere Po dV. Write the...
-
A piston/cylinder assembly contains 1 kg of liquid water at 20oC and 300 kPa. There is a linear spring mounted on the piston such that when the water is heated the pressure reaches 1 MPa with a...
-
The throttle process in Example 6.5 is an irreversible process. Find the reversible work and irreversibility assuming an ambient temperature at 25C.
-
On January 2, 2018, The Register, a science and technology news site based in Britain, published an article revealing the existence of two major bugs leaving virtually all computers and smartphones...
-
In the 1970s, Special Electric Company brokered the sale of crocidolite asbestos, which is the most toxic form of asbestos, to Johns- Manville Corporation. Special Electric never held possession of...
-
Plaintiffs W. O. and J. C. Lucy had wanted to purchase Ferguson Farm from the Zehmers for at least eight years. One night, Lucy stopped by the establishment the Zehmers operated and said that he bet...

Study smarter with the SolutionInn App


