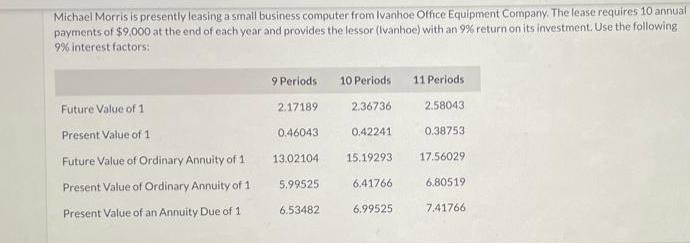
Michael Morris is presently leasing a small business computer from Ivanhoe Office Equipment Company. The lease...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Michael Morris is presently leasing a small business computer from Ivanhoe Office Equipment Company. The lease requires 10 annual payments of $9,000 at the end of each year and provides the lessor (Ivanhoe) with an 9% return on its investment. Use the following 9% interest factors: Future Value of 1 Present Value of 1 Future Value of Ordinary Annuity of 1 Present Value of Ordinary Annuity of 1 Present Value of an Annuity Due of 1 9 Periods 2.17189 0.46043 13.02104 5.99525 6.53482 10 Periods 2.36736 0.42241 15.19293 6.41766 6.99525 11 Periods 2.58043 0.38753 17.56029 6.80519 7.41766 (a) Assuming the computer has a ten-year life and will have no salvage value at the expiration of the lease, what was the original cost of the computer to Ivanhoe? (Round factor values to 5 decimal places, e.g. 1.25124 and final answer to 0 decimal places, e.g. 458,581.) Michael Morris is presently leasing a small business computer from Ivanhoe Office Equipment Company. The lease requires 10 annual payments of $9,000 at the end of each year and provides the lessor (Ivanhoe) with an 9% return on its investment. Use the following 9% interest factors: Future Value of 1 Present Value of 1 Future Value of Ordinary Annuity of 1 Present Value of Ordinary Annuity of 1 Present Value of an Annuity Due of 1 9 Periods 2.17189 0.46043 13.02104 5.99525 6.53482 10 Periods 2.36736 0.42241 15.19293 6.41766 6.99525 11 Periods 2.58043 0.38753 17.56029 6.80519 7.41766 (a) Assuming the computer has a ten-year life and will have no salvage value at the expiration of the lease, what was the original cost of the computer to Ivanhoe? (Round factor values to 5 decimal places, e.g. 1.25124 and final answer to 0 decimal places, e.g. 458,581.)
Expert Answer:
Answer rating: 100% (QA)
SOLUTION To calculate the cost of leasing the computer we can use the present value of an annuity f... View the full answer

Related Book For 

Advanced Financial Accounting
ISBN: 978-0137030385
6th edition
Authors: Thomas Beechy, Umashanker Trivedi, Kenneth MacAulay
Posted Date:
Students also viewed these accounting questions
-
A special metal S conducts electricity without any resistance. A closed wire loop, made of S, does not allow any change in flux through itself by inducing a suitable current to generate a...
-
Defined blip as a pair of integers, written a,b) is the same if a+b= x+y, let B = {(x,y) = x.y = Z} and is defined as the following * operation (a,b) (c,d) = (a+c,b+d) Is B group under the operation...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Herbs Pty Ltd is considering investing in a new herb packaging machine. The machine is estimated to cost $80,000 which can last for 7 years before it becomes too costly to maintain and can be sold...
-
The turbine in Problem 14.23 is adiabatic. Is it reversible, irreversible, or impossible?
-
Assuming you were specifying an AISI 1040 steel for an application where you desired to maximize the ductility, how would you specify it?
-
The Bailey Insurance Agency received the following notes during 2010: Requirements 1. Identifying each note by number, compute interest using a 360-day year, and determine the due date and maturity...
-
The comparative balance sheets for 2011 and 2010 are given below for Surmise Company. Net income for 2011 was $50 million. Required: Prepare the statement of cash flows of Surmise Company for the...
-
What are the challenges associated with virtual teamwork, and what strategies can teams employ to overcome barriers to collaboration, build trust, and maintain cohesion in distributed work...
-
The Buse Sound, Ltd. has three divisions for selling music: Cassette, Vinyl, and CD. The divisions have the following revenues and expenses for June 2022: Cassette Vinyl CD Revenues $82,000 $430,000...
-
Show that the equal payment series compounding factor is i F = A[ (+)-1].
-
Read the article: https://www.umgc.edu/current-students/learning-resources/writing-center/writing-resources/getting-started-writing/writing-for-an-audience View yourself as a speech expert. You have...
-
Data for Hermann Corporation are shown below: Selling price Variable expenses Contribution margin Percent Per Unit of Sales $ 60 39 $ 21 100% 65 35% Fixed expenses are $72,000 per month and the...
-
A 5-cm-diameter cylinder that is 50 cm long is at 55 oC and is to be cooled by crossflow immersion in a 10 oC stream. From Fig. 1.8, if the desired initial cooling rate is 300 W, what should be the...
-
Mason Company provided the following data for this year: Sales Direct labor cost Raw material purchases Selling expenses Administrative expenses Manufacturing overhead applied to work in process...
-
Red Kyoto makes canned carrot puree for use in restaurants and hospitals. They have ascertained that the cost of transporting its products (measured in truck loads) can be reduced by using a...
-
In Exercises 15 through 30, find the derivative dy/dx. In some of these problems, you may need to use implicit differentiation or logarithmic differentiation. y ex + et -2x 1 + e
-
Assume the same facts as in P8- 13 except that Pinware Wholesalers Inc. (Pinware) must order the watches two months in advance of expected delivery. On March 1, 20X3, Pinware ordered Swiss watches...
-
Polar Limited is a Canadian company that recently completed two large transactions with companies based in Germany: 1. On July 1, 20X4, Polar acquired equipment at a cost of 500,000 from Bier Ltd....
-
In general, how would fair values be determined for productive assets?
-
Explain in detail, with neat sketch, the working principle of a fuel cell in producing electrical energy from the chemical energy of a fuel.
-
Under atmospheric conditions the acetone-chloroform azeotrope boils at \(64.6^{\circ} \mathrm{C}\) and contains 33.5 mole per cent acetone. The vapour pressures of acetone and chloroform at this...
-
A system was prepared by partially decomposing \(\mathrm{CaCO}_{3}\) into an evacuated space. What is the number of degrees of freedom \((f)\) for the system?

Study smarter with the SolutionInn App


