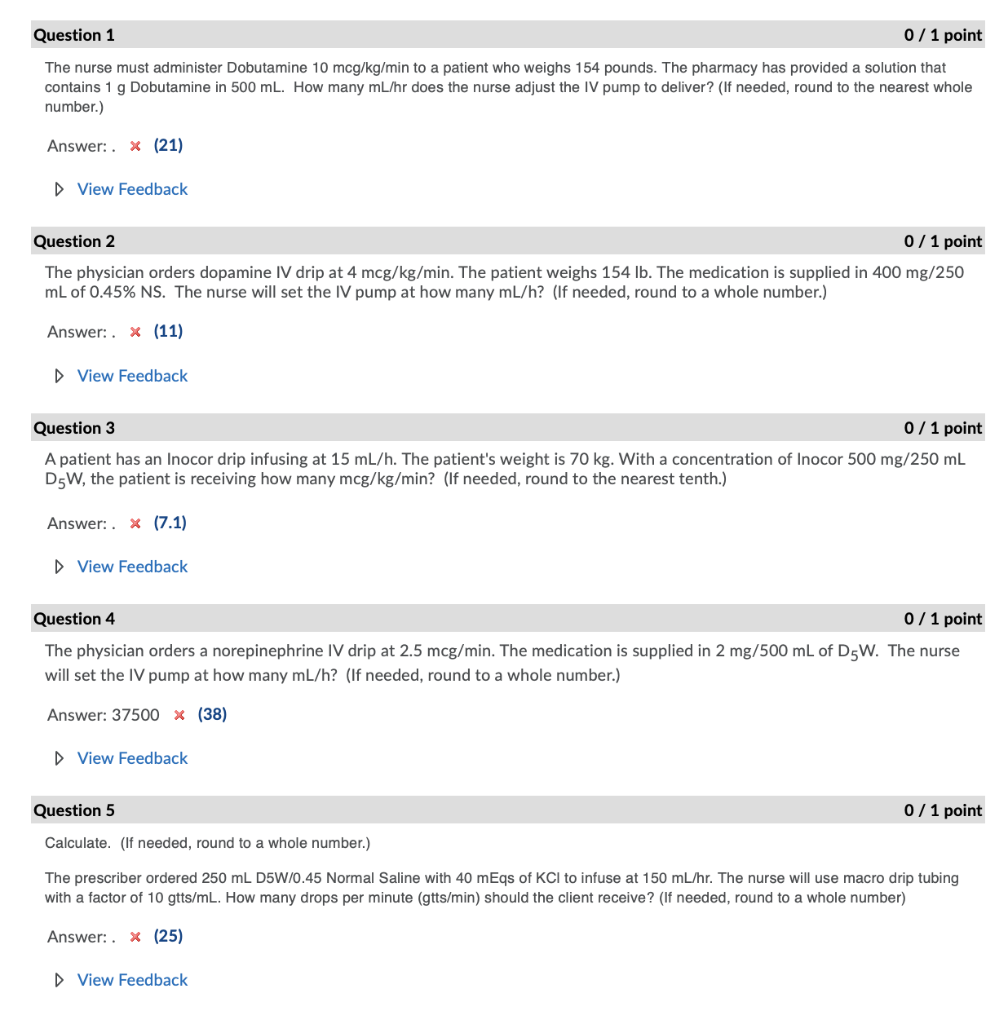
Question 1 0/1 point The nurse must administer Dobutamine 10 mcg/kg/min to a patient who weighs...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question 1 0/1 point The nurse must administer Dobutamine 10 mcg/kg/min to a patient who weighs 154 pounds. The pharmacy has provided a solution that contains 1 g Dobutamine in 500 mL. How many mL/hr does the nurse adjust the IV pump to deliver? (If needed, round to the nearest whole number.) Answer:. x (21) View Feedback Question 2 0/1 point The physician orders dopamine IV drip at 4 mcg/kg/min. The patient weighs 154 lb. The medication is supplied in 400 mg/250 mL of 0.45% NS. The nurse will set the IV pump at how many mL/h? (If needed, round to a whole number.) Answer:. x (11) ▷ View Feedback Question 3 0/1 point A patient has an Inocor drip infusing at 15 mL/h. The patient's weight is 70 kg. With a concentration of Inocor 500 mg/250 mL D5W, the patient is receiving how many mcg/kg/min? (If needed, round to the nearest tenth.) Answer:. x (7.1) ▷ View Feedback Question 4 0/1 point The physician orders a norepinephrine IV drip at 2.5 mcg/min. The medication is supplied in 2 mg/500 mL of D5W. The nurse will set the IV pump at how many mL/h? (If needed, round to a whole number.) Answer: 37500 * (38) ▷ View Feedback Question 5 Calculate. (If needed, round to a whole number.) The prescriber ordered 250 mL D5W/0.45 Normal Saline with 40 mEqs of KCl to infuse at 150 mL/hr. The nurse will use macro drip tubing with a factor of 10 gtts/mL. How many drops per minute (gtts/min) should the client receive? (If needed, round to a whole number) Answer:. x (25) 0/1 point ▷ View Feedback Question 1 0/1 point The nurse must administer Dobutamine 10 mcg/kg/min to a patient who weighs 154 pounds. The pharmacy has provided a solution that contains 1 g Dobutamine in 500 mL. How many mL/hr does the nurse adjust the IV pump to deliver? (If needed, round to the nearest whole number.) Answer:. x (21) View Feedback Question 2 0/1 point The physician orders dopamine IV drip at 4 mcg/kg/min. The patient weighs 154 lb. The medication is supplied in 400 mg/250 mL of 0.45% NS. The nurse will set the IV pump at how many mL/h? (If needed, round to a whole number.) Answer:. x (11) ▷ View Feedback Question 3 0/1 point A patient has an Inocor drip infusing at 15 mL/h. The patient's weight is 70 kg. With a concentration of Inocor 500 mg/250 mL D5W, the patient is receiving how many mcg/kg/min? (If needed, round to the nearest tenth.) Answer:. x (7.1) ▷ View Feedback Question 4 0/1 point The physician orders a norepinephrine IV drip at 2.5 mcg/min. The medication is supplied in 2 mg/500 mL of D5W. The nurse will set the IV pump at how many mL/h? (If needed, round to a whole number.) Answer: 37500 * (38) ▷ View Feedback Question 5 Calculate. (If needed, round to a whole number.) The prescriber ordered 250 mL D5W/0.45 Normal Saline with 40 mEqs of KCl to infuse at 150 mL/hr. The nurse will use macro drip tubing with a factor of 10 gtts/mL. How many drops per minute (gtts/min) should the client receive? (If needed, round to a whole number) Answer:. x (25) 0/1 point ▷ View Feedback
Expert Answer:
Answer rating: 100% (QA)
Answer 1 Dobutamine ordered 10 mcg kg minute Weight of patient 154 lb Formula for weight convert divide the mass value by 2205 Weight in kg 154 2205 6... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these law questions
-
Find all solutions to the homogeneous system Ax = 0 for the coefficient matrix (a) (b) (c) (d) (e) (f) (g) (h) 3 2 3 0 4 1 258 147 130 203 021 0226 2371 1141 1133 3033 1121 003-3 1-0 3 2 -2 5 1-4/
-
Find the general solutions for Problems a-c. Sketch the eigerrvectors and a few typical trajectories.(Show your method) a. b. c. 2 2 6 2 4 2
-
Find the general solutions for Problems a, and c handsketch the phase portraits from nullclines and/or vector fields. (You can check your results by using a CAS, a solver. or /DE Matrix Element...
-
Which statement about the Java variable and literal is NOT true? a. A global variablewill be initalised by the complier automatically. b. Literals can also be used as the lvalue in an express. c. An...
-
Why would a company such as Target Corporation prefer a quick response inventory policy?
-
Consider the transportation problem having the following parameter table: Destination 3 4 Supply Source Demand
-
On January 1, 2020, lessor Marcy and lessee Lenox contract for the lease of a machine for five payments of \(\$ 7,000\) each. The \(\$ 7,000\) payments are to be paid at the end of each year. They...
-
Flexible budget Brabham enterprises manufactures tires for the Formula l motor racing circuit. For August 2009, it budgeted to manufacture and sell 3000 tires at a variable cost of $ 74 per tire and...
-
338. 3 eBook 4. A-Z Break-even Point S Schweser Satellites Inc. produces satellite earth stations that sell for $95,000 each. The firm's fixed costs, F, are $2 million, 50 earth stations are 6. O...
-
Locate the court case Ellis v. Jarvis et al., 117 AFTR 2d 2016-1932. Using only the head notes, answer the following questions: a. What issue(s) did the court address? b. What was the ruling of the...
-
Franklin Corporation incurs the following annual fixed costs. Item Depreciation Officers' salaries Long-term lease Property taxes Cost $ 63,000 160,000 50,000 13,000 Required Determine the total...
-
If 1 cup of 3 6 oC water is mixed with 2 cups of 9 o C water, what is the temperature of the final mixture, in o C ?
-
A scuba diving school has been given the opportunity to purchase a sunken shipwreck for just $25,000, giving trainee scuba divers the opportunity to navigate through the ship as part of their...
-
Two charges of +8.0 * 10^-9 C and +4.0 * 10^-9 C are held a distance of 0.20 m A. Calculate the magnitude and direction of the electric field strength at the midpoint between the B. Calculate the...
-
Is linux and Ubuntu the same or is Ubuntu an distributor of linux ? Can I use Ubuntu just i would linux ?
-
If the initial angular velocity is - 3 . 1 rad / s , then what is the angular velocity, in radians per second, at time 2 3 . 7 5 s ? A detailed graph of angular acceleration versus time is shown....
-
There are a few misconceptions about peer reviewed research that learners have when beginning a doctoral program. Many learners believe things such as the following: A published research article is...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
Complete and balance the following equations: (a) NaH(s) + H2O(l) (b) Fe(s) + H2SO4(aq) (c) H2(g) + Br2(g) (d) Na(l) + H2(g) (e) PbO(s) + H2(g)
-
Hard water contains Ca2+, Mg2+, and Fe2+, which interfere with the action of soap and leave an insoluble coating on the insides of containers and pipes when heated. Water softeners replace these ions...
-
Lauryl alcohol is obtained from coconut oil and is used to make detergents. A solution of 5.00 g of lauryl alcohol in 0.100 kg of benzene freezes at 4.1 oC. What is the approximate molar mass of...
-
For the coordinate transformation (12.1) and (12.2), develop the second-order finite difference approximations of \(u_{y}, u_{x x}\), and \(u_{x y}\) at \(\left(\xi_{i}, \eta_{j} ight)\). Apply...
-
For the coordinate transformation (12.1) and (12.2), derive the formula for \(u_{x y}\).
-
If your course involves exercises with a finite volume CFD software, study the software manual to determine which types of cell shapes are available for unstructured grids. Does the software allow...

Study smarter with the SolutionInn App


