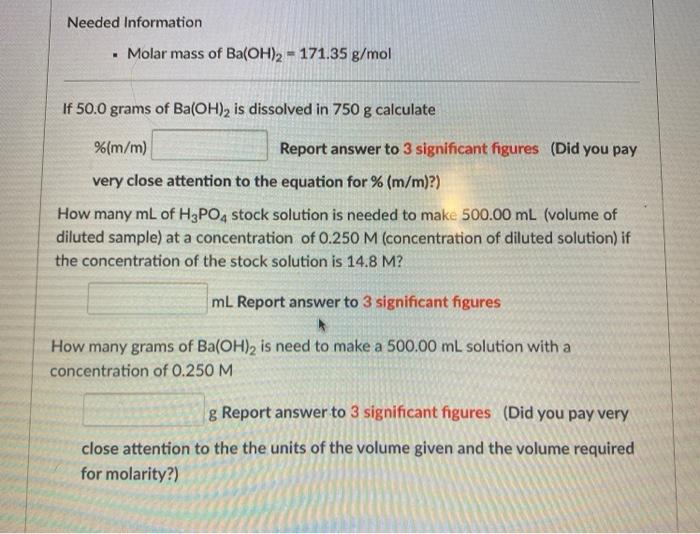
Needed Information Molar mass of Ba(OH)2 = 171.35 g/mol If 50.0 grams of Ba(OH), is...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Needed Information • Molar mass of Ba(OH)2 = 171.35 g/mol If 50.0 grams of Ba(OH), is dissolved in 750 g calculate %(m/m) Report answer to 3 significant figures (Did you pay very close attention to the equation for % (m/m)?) How many mL of H3PO, stock solution is needed to make 500.00 mL (volume of diluted sample) at a concentration of 0.250 M (concentration of diluted solution) if the concentration of the stock solution is 14.8 M? mL Report answer to 3 significant figures How many grams of Ba(OH)2 is need to make a 500.00 mL solution with a concentration of 0.250 M g Report answer to 3 significant figures (Did you pay very close attention to the the units of the volume given and the volume required for molarity?) Needed Information • Molar mass of Ba(OH)2 = 171.35 g/mol If 50.0 grams of Ba(OH), is dissolved in 750 g calculate %(m/m) Report answer to 3 significant figures (Did you pay very close attention to the equation for % (m/m)?) How many mL of H3PO, stock solution is needed to make 500.00 mL (volume of diluted sample) at a concentration of 0.250 M (concentration of diluted solution) if the concentration of the stock solution is 14.8 M? mL Report answer to 3 significant figures How many grams of Ba(OH)2 is need to make a 500.00 mL solution with a concentration of 0.250 M g Report answer to 3 significant figures (Did you pay very close attention to the the units of the volume given and the volume required for molarity?)
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these accounting questions
-
A thin, 50.0-cm-Iong metal bar with mass 750 g rests n, but is not attached to, two metallic supports in a uniform 0.450-Tmagnetic field, as shown v R in Fig. A battery and a 25.0-Ω resistor in...
-
Calculate the mass in grams of the following. a. 0.15 mol Na b. 0.594 mol S c. 2.78 mol CH2Cl2 d. 38 mol (NH4)2S
-
Calculate the mass in grams of the following. a. 0.205 mol Fe b. 0.79 mol F c. 5.8 mol CO2 d. 48.1 mol K2CrO4
-
Two tiny particles having charges of 5.76 C and -7 C are placed along the y-axis. The 5.76 C particle is at y = 0 cm, and the other particle is at y = 48.74 cm. Where must a third charged particle be...
-
Mack and Myer, LLP, a law firm, is considering the replacement of its old accounting system with new software that should save $10,000 per year in net cash operating costs. The old system has zero...
-
1. State the 4 major challenges or themes for IT leaders when it comes to managing IT innovation. 2. State some of the guiding principles for facilitating IT innovation. 3. What are some of the...
-
Explain the different quality management methods that are applicable to a project organization.
-
Jennings operated a courier service to collect and deliver money. The contract with his customers allowed him a day or so to deliver the money that had been collected. Instead of holding collections...
-
7. Relative Velocity 10 points): Two airplanes fly between the same two cities. The destination city is due north of the origin, and both planes experience the same west-to-east crosswind but travel...
-
Bruce also requires a calculation of taxable income for the most recent year ended December 31, 2019. The calculation should be prepared in Excel. He has also requested that you prepare the year-end...
-
PART C (5 Marks) Rumba Pty Ltd is a resident company that was incorporated on 1 October 2016 and commenced manufacturing and selling rum. The company incurred losses in its first three years of...
-
Explain why Enron had so much debt from the time of its inception onward and explain why it was under pressure to hide this debt.
-
Which of the following methods of fictitious accounting was not identified by the SEC in its Enforcement Action against Rite Aid? (a) Understatement of bad debt expense. (b) Failure to write off...
-
FCPA violations often consist of bribery, (a) In which the amounts involved are not in themselves material. (b) Which can have serious consequences. (c) Which is difficult to identify in financial...
-
It is important for users of financial statements to be aware of different ____________ methods and to question why one method was ____________ rather than another.
-
Which of the following was not a signal in Enrons financial statements of its financial problems? (a) Receivables increased as a percentage of sales. (b) Its debt-to-equity ratio decreased over time....
-
4. (a) Please draw a cubic unit cell and show planes such as (110), (011), (001), (112) (b) Please write the crystal directions given in the cubic unit cell. X 132 C 1/2 N a b 1|2 133 y
-
What are the principal differences among asset liquidity management, liability management, and balanced liquidity management?
-
Find the number of half-lives required to reach the following levels of completion: (a) 10%. (b) 90%. (c) 99.9%.
-
Consider the following sets of replicate measurements: Calculate the mean and the standard deviation for each of these six data sets. Calculate the 95% confidence interval for each set of data. What...
-
Calculate the initial potential needed for a current of 0.065 A in the cell Co|Co2+ (5.90 ( 10-3 M) ||Zn2+ (2.95 3 10-3 M) |Zn if this cell has a resistance of 4.50 V.
-
Determine the natural frequency of vibration, in bending, of the system shown in Figs. 2.90 (c) by modeling the system as a single-degree-of-freedom system. Assume that the mass is \(m=50...
-
Determine the natural frequency of vibration, in bending, of the system shown in Figs. 2.90 (a) by modeling the system as a single-degree-of-freedom system. Assume that the mass is \(m=50...
-
Determine the natural frequency of vibration, in bending, of the system shown in Figs. 2.90 (b) by modeling the system as a single-degree-of-freedom system. Assume that the mass is \(m=50...

Study smarter with the SolutionInn App



