No need to show work for questions with answers already What is the change in enthalpy(H) for
Fantastic news! We've Found the answer you've been seeking!
Question:
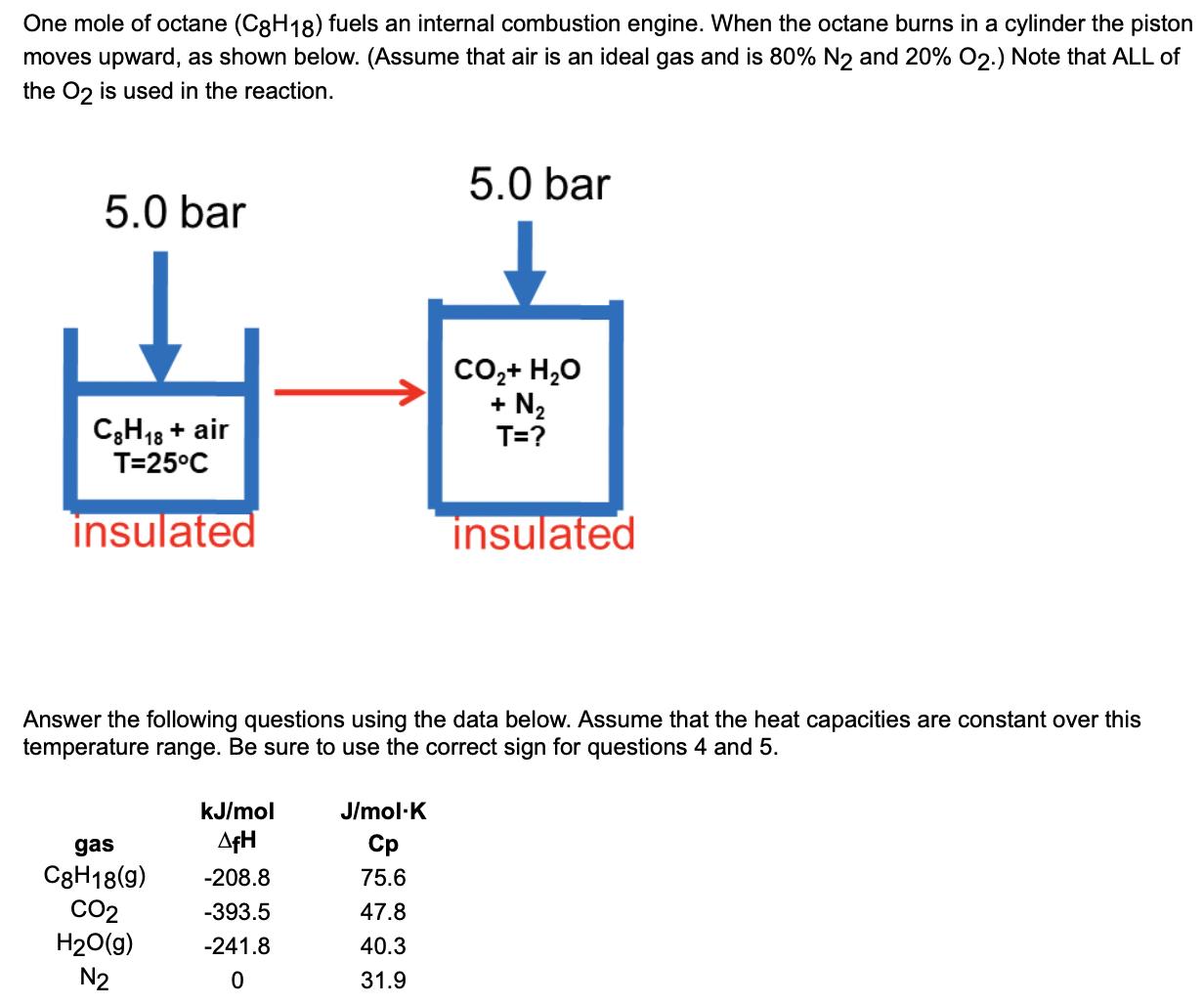
No need to show work for questions with answers already
- What is the change in enthalpy(H) for the contents of the cylinder? (kJ) This is an insulated cylinder and a constant pressure process. (Hint:Enthalpy change is equal to __ for a constant pressure process.)
- ?
- How many moles of oxygen are in the cylinder before reaction? Answer=12.5
- What is the final gas temperature? (K) Answer=2484
- How much work is performed by the gas? (kJ) (Hint: the answer is a negative number)
- ?
- What is the change in internal energy (U) for the gas in the cylinder? (kJ) U=Q+W
Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:




