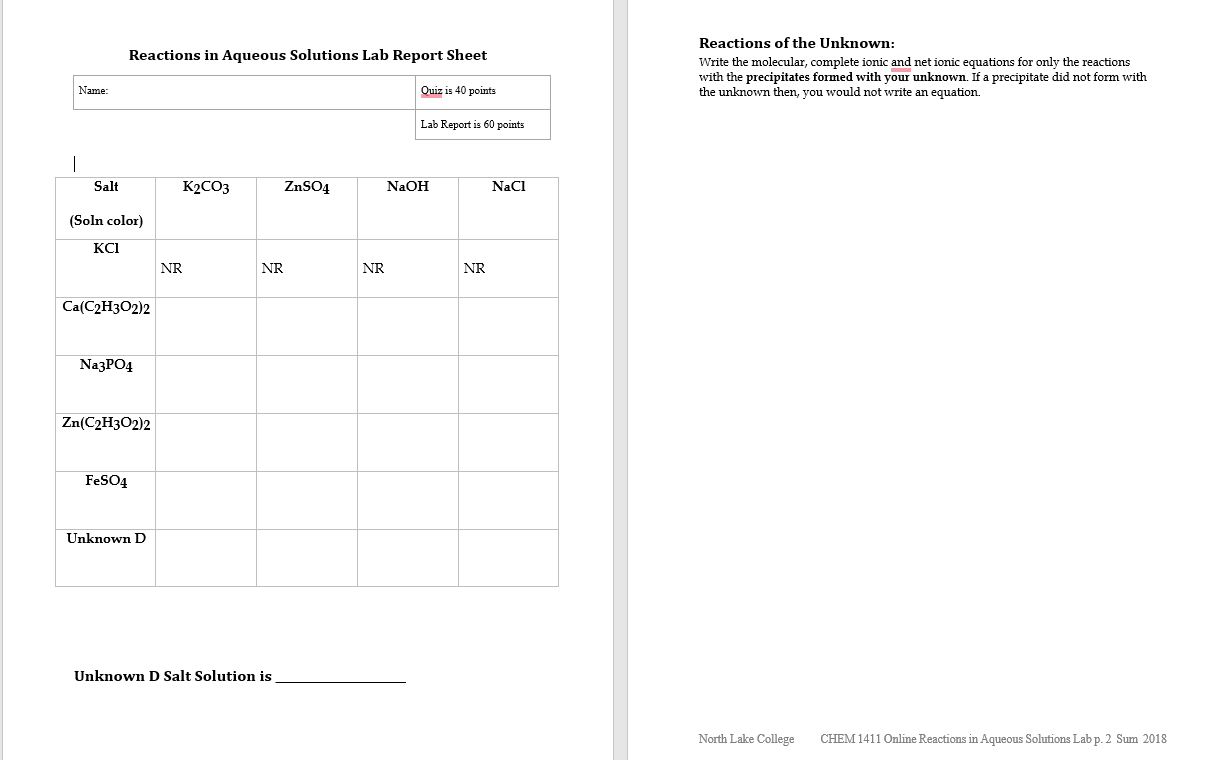
Name: | Salt (Soln color) KCI Reactions in Aqueous Solutions Lab Report Sheet Ca(C2H3O2)2 Na3PO4 Zn(C2H3O2)2...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Name: | Salt (Soln color) KCI Reactions in Aqueous Solutions Lab Report Sheet Ca(C2H3O2)2 Na3PO4 Zn(C2H3O2)2 FeSO4 Unknown D NR K2CO3 ZnSO4 NR Unknown D Salt Solution is NR Quiz is 40 points Lab Report is 60 points NaOH NR NaCl Reactions of the Unknown: Write the molecular, complete ionic and net ionic equations for only the reactions with the precipitates formed with your unknown. If a precipitate did not form with the unknown then, you would not write an equation. North Lake College CHEM 1411 Online Reactions in Aqueous Solutions Lab p. 2 Sum 2018 Name: | Salt (Soln color) KCI Reactions in Aqueous Solutions Lab Report Sheet Ca(C2H3O2)2 Na3PO4 Zn(C2H3O2)2 FeSO4 Unknown D NR K2CO3 ZnSO4 NR Unknown D Salt Solution is NR Quiz is 40 points Lab Report is 60 points NaOH NR NaCl Reactions of the Unknown: Write the molecular, complete ionic and net ionic equations for only the reactions with the precipitates formed with your unknown. If a precipitate did not form with the unknown then, you would not write an equation. North Lake College CHEM 1411 Online Reactions in Aqueous Solutions Lab p. 2 Sum 2018
Expert Answer:
Answer rating: 100% (QA)
K2CO3 Molecular equation K2CO3 aq H2O l 2KOH aq CO2 g H2O l Complete ionic equation 2K aq CO3 2 aq 2H aq 2OH aq 2K aq 2OH aq CO2 g H2O l Net ionic equ... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Please help with the following questions below... 1) Based on your SWOT of Southwest Airlines, explain which one of the 5 generic business strategies you think Southwest is following and why. Explain...
-
Cabana Cruise Line offers cruise ship service to various tropical locations throughout the globe. The company is considering the addition of a new cruise ship to its fleet in order to expand service...
-
Given the network in Figure P8.2 (a) write the equations for va (t) and vb (t) and (b) write the equations for vc (t) and vd (t). vat)w(t) Figure P8.2
-
The adjusted trial balance for Ray Corporation at July 31, 2017, the corporation's fiscal year end, contained the following: Of the lease liability amount, $16,250 is due within the next year. Total...
-
Entre Preneur found a site for his new haute cuisine restaurant. The site has a vacant gasoline station. He purchased the property for $ 900,000 and had the station demolished at a cost of $ 30,000....
-
Two projects, A and B, are analyzed using ranking present worth analysis with MARR at \(i \%\). It is found that \(\mathrm{PW}(\mathrm{A})>\mathrm{PW}(\mathrm{B})\). If \(M A R R\) is changed to...
-
Develop the appropriate primary research question to be associated with this design. Develop a hypothetical research scenario that would necessitate the use of the Action Research Approach and a...
-
The comparative balance sheets of Menachem Corporation at the beginning and end of the year 2012 appear below. Net income of $34,000 was reported, and dividends of $23,000 were paid in 2012. New...
-
Is Mongodb more efficient or faster in scaling than NoSQL?
-
Amy Lloyd is interested in leasing a new Honda and has contacted three automobile dealers for pricing information. Each dealer offered Amy a closed-end 36-month lease with no down payment due at the...
-
Assume the variable s has been assigned a value and my_set refers to a set. Write code that determines whether s is in my_set. If so, assign True to the found variable. Otherwise, assign False to...
-
Which statement is the most accurate? a) Our economy was at full employment in 2012. b) Our economy operated outside its production possibilities curve in 2012. c) Our economy is currently operating...
-
You purchased a machine for $1.09 million three years ago and have been applying straight-line depreciation to zero for a seven-year life. Your tax rate is 21%. If you sell the machine today (after...
-
Karl Stick is president of Stock Company. He also owns 100 percent of its stock. Karl's salary is $220,000. At the end of the year, Karl was paid a bonus of $100,000 because the firm had a good year....
-
Lets consider the effects of inflation in an economy composed of only two people: Bob, a bean farmer, and Rita, a rice farmer. Bob and Rita both always consume equal amounts of rice and beans. In...
-
You are planning to invest $1000 in an account earning 7% per year for retirement. a. If you put the $1000 in an account at age 23, and withdraw it 34 years later, how much will you have? b. If you...
-
What are reading frames?
-
Why do markets typically lead to an efficient outcome for buyers and sellers?
-
Frank Willingham has the following transactions during the year: Sale of office equipment on March 15 that cost $20,000 when purchased on July 1, 2010. Frank has claimed $5,000 in depreciation and...
-
Harold Conners (Social Security number 785-23-9873) lives at 13234 DeMilo Drive, Houston, TX 77052, and is self-employed for 2012. He estimates his required annual estimated tax payment for 2012 to...
-
Marty is a sales consultant. Marty incurs the following expenses related to entertainment of his clients in 2012: Dues to a country club......................................................$4,500...
-
Beginning in the 1920s, Russian physicist Pyotr Kapitza or Kapitsa (18941984, Nobel laureate in physics 1978) measured the Paschen-Back effect to an accuracy of 1 percent to 3 percent in various...
-
Consider transitions from a \({ }^{2} D\) state to a \(2 P\) state in the strong field PaschenBack regime. List all allowed transitions and show that there are only three different spectral lines.
-
What is the longest wavelength of the Paschen series spectrum? Would it be visible to the human eye?

Study smarter with the SolutionInn App


