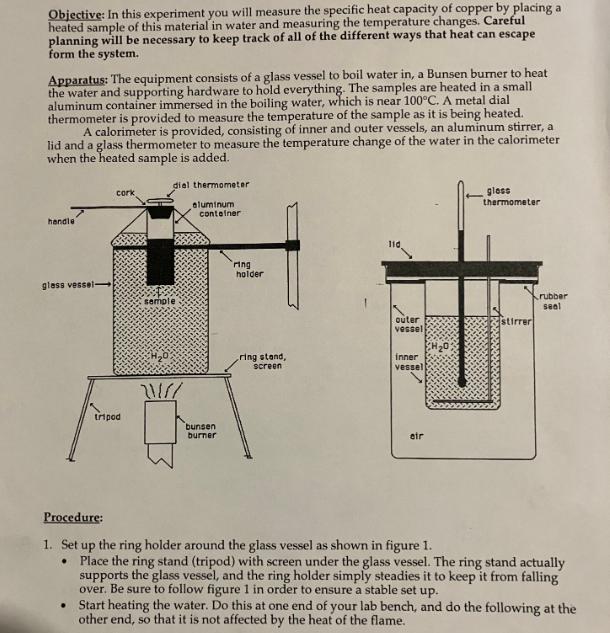
Objective: In this experiment you will measure the specific heat capacity of copper by placing a...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
Objective: In this experiment you will measure the specific heat capacity of copper by placing a heated sample of this material in water and measuring the temperature changes. Careful planning will be necessary to keep track of all of the different ways that heat can escape form the system. Apparatus: The equipment consists of a glass vessel to boil water in, a Bunsen burner to heat the water and supporting hardware to hold everything. The samples are heated in a small aluminum container immersed in the boiling water, which is near 100C. A metal dial thermometer is provided to measure the temperature of the sample as it is being heated. A calorimeter is provided, consisting of inner and outer vessels, an aluminum stirrer, a lid and a glass thermometer to measure the temperature change of the water in the calorimeter when the heated sample is added. handle glass vessel- cork tripod diel thermometer aluminum container semple H0 21/// bunsen burner ring holder ring stand, screen 110 outer vessel inner vessel eir gloss thermometer rubber seal Procedure: 1. Set up the ring holder around the glass vessel as shown in figure 1. Place the ring stand (tripod) with screen under the glass vessel. The ring stand actually supports the glass vessel, and the ring holder simply steadies it to keep it from falling over. Be sure to follow figure 1 in order to ensure a stable set up. Start heating the water. Do this at one end of your lab bench, and do the following at the other end, so that it is not affected by the heat of the flame. 2. Weigh the copper pellets. Make sure you that you do not include any containers. Record the weight of the copper pellets only. Write the mass in table 2. (Use enough copper pellets to fill the aluminum sample container about 1/3 to 1/2 full.) 3. Pour the copper pellets back into the aluminum sample container. Cover the sample with a cork, and insert the metal dial thermometer into the pellets to monitor their temperature. Insert the container into the boiling water in the glass vessel as shown in figure 1. The water level should be approximately as shown. 4. While you are waiting for the pellets to reach 100C, you should weigh and record the various parts of the calorimeter that come in contact with the water in the calorimeter. Refer to figure 2. Record the first two masses in table 1, and the mass of the water in table 2. These include: stirrer inner vessel water c=0.22 kcal/kg C c=0.22 kcal/kgC c=1.00 kcal/kg C The specific heat capacity of each item is listed. The inner vessel should be about 1/2 to 2/3 full of water at room temperature. During and after making the above measurements, keep the calorimeter far from the Bunsen burner so that the heat from the flame doesn't affect the results. The masses of the lid, rubber seal and outer vessel are not needed as they are not in direct contact with the water, and therefore should not absorb heat from it. 5. Once the pellets in (2) have reached 100C (or very near it)... Measure and record the temperature of the hot copper pellets Measure and record the temperature of the water in the calorimeter. Then, quickly dump the sample pellets into the calorimeter, put the lid on and stir with the stirrer. Record the highest temperature reached by the water, and make sure the sample is well- stirred. 6. Record the temperature of the water every two minutes occasionally stirring the pellets. Thoroughly dry off the copper pellets and put them away. Use paper towels and heat to do this. Data and Calculations Before you leave the lab, you need to fill out the information in tables 1 and 2. Be neat and clear as you fill out each table. Watch your units! Mass of Inner vessel Mass of Stirrer Mass of Copper Pellets Mass of Water in Calorimeter Calorimeter Table 1 Copper Sample Initial Temperature of Copper Initial Temperature of Calorimeter Immediate Final Temperature of both Temperature after 2 min. Temperature after 4 min. 45.39 0.0453 kg 12.549-0.012546 Table 2 89. 19570.0841K4 150.654-70.15065K 99.6372, 75k 21.0C -7 294,15K 27.0C 300.15K 25, 7 C-7 298.85 k 25.8 C 296.95 K Analysis of Data 1. Which of the above objects lost heat when the pellets were dumped into the calorimeter? The Copper lost heat when the pellets were dumper into the calorimeter. 2. Which of the above objects gained heat when the pellets were transferred into the calorimeter? The vessel, stirrer, and water gained heat when the pelle were transferred into the calorimeter. 3. Calculate the amount of heat gained using your numbers in the data tables. You will also need to use the values for specific heat given in the previous page. 4. How should the amount of heat lost compare to the amount of heat gained? 905+= -9 gained 5. Use your answer to questions (1) and (4) to solve for the specific heat of copper. 6. Calculate the percent error of the specific heat of copper. The accepted value for copper 0.0923kcal/kg.C. is percen+ Error = Cmeasured-0.0923 0.0923 x 100% 7. Discuss sources of error in your conclusion. You will need to write about the different ways heat loss/gain could have affected your results. Objective: In this experiment you will measure the specific heat capacity of copper by placing a heated sample of this material in water and measuring the temperature changes. Careful planning will be necessary to keep track of all of the different ways that heat can escape form the system. Apparatus: The equipment consists of a glass vessel to boil water in, a Bunsen burner to heat the water and supporting hardware to hold everything. The samples are heated in a small aluminum container immersed in the boiling water, which is near 100C. A metal dial thermometer is provided to measure the temperature of the sample as it is being heated. A calorimeter is provided, consisting of inner and outer vessels, an aluminum stirrer, a lid and a glass thermometer to measure the temperature change of the water in the calorimeter when the heated sample is added. handle glass vessel- cork tripod diel thermometer aluminum container semple H0 21/// bunsen burner ring holder ring stand, screen 110 outer vessel inner vessel eir gloss thermometer rubber seal Procedure: 1. Set up the ring holder around the glass vessel as shown in figure 1. Place the ring stand (tripod) with screen under the glass vessel. The ring stand actually supports the glass vessel, and the ring holder simply steadies it to keep it from falling over. Be sure to follow figure 1 in order to ensure a stable set up. Start heating the water. Do this at one end of your lab bench, and do the following at the other end, so that it is not affected by the heat of the flame. 2. Weigh the copper pellets. Make sure you that you do not include any containers. Record the weight of the copper pellets only. Write the mass in table 2. (Use enough copper pellets to fill the aluminum sample container about 1/3 to 1/2 full.) 3. Pour the copper pellets back into the aluminum sample container. Cover the sample with a cork, and insert the metal dial thermometer into the pellets to monitor their temperature. Insert the container into the boiling water in the glass vessel as shown in figure 1. The water level should be approximately as shown. 4. While you are waiting for the pellets to reach 100C, you should weigh and record the various parts of the calorimeter that come in contact with the water in the calorimeter. Refer to figure 2. Record the first two masses in table 1, and the mass of the water in table 2. These include: stirrer inner vessel water c=0.22 kcal/kg C c=0.22 kcal/kgC c=1.00 kcal/kg C The specific heat capacity of each item is listed. The inner vessel should be about 1/2 to 2/3 full of water at room temperature. During and after making the above measurements, keep the calorimeter far from the Bunsen burner so that the heat from the flame doesn't affect the results. The masses of the lid, rubber seal and outer vessel are not needed as they are not in direct contact with the water, and therefore should not absorb heat from it. 5. Once the pellets in (2) have reached 100C (or very near it)... Measure and record the temperature of the hot copper pellets Measure and record the temperature of the water in the calorimeter. Then, quickly dump the sample pellets into the calorimeter, put the lid on and stir with the stirrer. Record the highest temperature reached by the water, and make sure the sample is well- stirred. 6. Record the temperature of the water every two minutes occasionally stirring the pellets. Thoroughly dry off the copper pellets and put them away. Use paper towels and heat to do this. Data and Calculations Before you leave the lab, you need to fill out the information in tables 1 and 2. Be neat and clear as you fill out each table. Watch your units! Mass of Inner vessel Mass of Stirrer Mass of Copper Pellets Mass of Water in Calorimeter Calorimeter Table 1 Copper Sample Initial Temperature of Copper Initial Temperature of Calorimeter Immediate Final Temperature of both Temperature after 2 min. Temperature after 4 min. 45.39 0.0453 kg 12.549-0.012546 Table 2 89. 19570.0841K4 150.654-70.15065K 99.6372, 75k 21.0C -7 294,15K 27.0C 300.15K 25, 7 C-7 298.85 k 25.8 C 296.95 K Analysis of Data 1. Which of the above objects lost heat when the pellets were dumped into the calorimeter? The Copper lost heat when the pellets were dumper into the calorimeter. 2. Which of the above objects gained heat when the pellets were transferred into the calorimeter? The vessel, stirrer, and water gained heat when the pelle were transferred into the calorimeter. 3. Calculate the amount of heat gained using your numbers in the data tables. You will also need to use the values for specific heat given in the previous page. 4. How should the amount of heat lost compare to the amount of heat gained? 905+= -9 gained 5. Use your answer to questions (1) and (4) to solve for the specific heat of copper. 6. Calculate the percent error of the specific heat of copper. The accepted value for copper 0.0923kcal/kg.C. is percen+ Error = Cmeasured-0.0923 0.0923 x 100% 7. Discuss sources of error in your conclusion. You will need to write about the different ways heat loss/gain could have affected your results.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
1. Why is it said that electronic commerce is a catalyst of fundamental changes in organizations? Search the Ashford Online Library for an article that addresses change in organizations as a result...
-
George operates a business that generated adjusted gross income of $250,000 and taxable income of $170,000 this year (before the domestic production activities deduction). Included in income was...
-
Suppose Hunter Valley is deciding whether to purchase new accounting software. The payback for the $30,050 software package is two years, and the software's expected life is three years. Hunter...
-
What is the difference between the spot, forward, and swap markets? Illustrate each description with an example.
-
Use the following information from the records of Packen Partners to prepare an income statement under the periodic inventory system for the year ended 30 June 2026. Purchases Inventory, 1 July 2025...
-
Presented below are two independent situations: 1. Potomac Inc. acquired 10% of the 500,000 shares of common stock of Maryland Corporation at a total cost of $11 per share on June 17, 2010. On...
-
a) Market refers to the mechanism through which all goods and services are voluntarily exchanged among different owners. Through price, markets allocate scarce resources among competing uses. Discuss...
-
Analyzes the evidence of discriminatory effects and provides specific examples of connections to the rule, policy, or process in EEOC V ABERCROMBIE court case
-
Scenario you have been taking public speaking and you learned about Informative Speech, Persuasive Speech, and Applying Ethos and Pathos. answered a few questions. Your feelings when you began the...
-
Case Study: DARLENE This scenario is based on the real-life human rights case of Noffke v. McClaskinHot House . As part of a government program, Darlene, a grade 12 graduate, got a job with a local...
-
provide a brief recommendation for your client. (my client is tim hortons ) i want answer based on tim hortons Describe 3 areas of concern about your client's customer service that you will tackle...
-
Three Peer Reviews In this forum, you have two duties. 1. Post a copy of your rough draft in the text box of this forum You will be forgiven for any formatting issues this creates. 2. Write three...
-
Interview the person you chose about your differences with the goal of learning more about their point of view. Remember that you're not here to argue with someone or convince them that they're...
-
A circle of radius r is a curve that consists of all points at distance r from the center of the circle. A disk of radius r is the set of all points whose distance from its center is less than or...
-
Arlington Merchants reported the following on its income statement for the fiscal years ending December 31, 2016 and 2015. 2016 2015 Sales $4,857,500 $4,752,900 Cost of goods sold 3,258,950 3,207,000...
-
Your instructor hands you a closed flask partly filled with room-temperature water. When you hold it, the heat transfer between your bare hands and the flask causes the water to boil. Quite...
-
In Chapter 26, we learned that light travels more slowly in glass than in air. Does this contradict Einstein's second postulate?
-
What is the weight of a cubic meter of cork? Could you lift it? (For the density of cork, use 400 kg/m3.)
-
Flaherty Ltd sells two types of shoes, mens shoes and womens shoes. During the financial year ended 30 June 2019, fixed costs were $460 800 and sales were in the ratio of three units (pairs) of mens...
-
Newbery and Newstead is a public accounting firm specialising in auditing local medium-sized businesses. Fees charged for each audit are determined on the basis of identifiable hours worked on an...
-
Hotbox Ltd produces pizza boxes using two processes cutting and packaging. The production budget for the year ending 30 June 2019 estimated raw materials use of $400 000, factory overhead of $270...

Study smarter with the SolutionInn App


