On January 1, 2017 Pucket Co. issued a $950,000, 4-yr, zero- interest bearing note to Key...
Fantastic news! We've Found the answer you've been seeking!
Question:
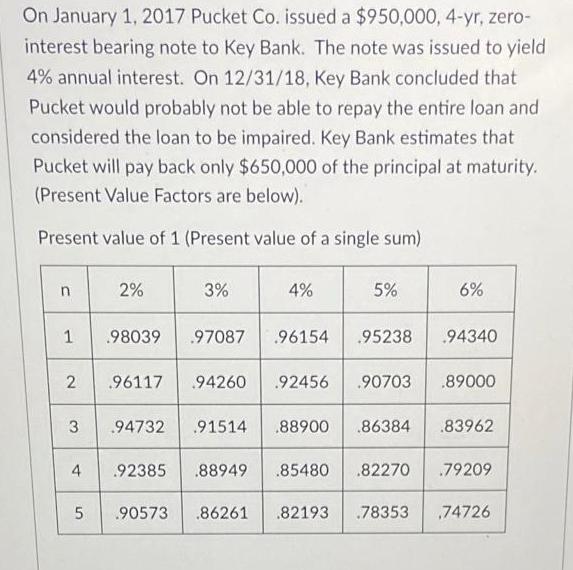

Transcribed Image Text:
On January 1, 2017 Pucket Co. issued a $950,000, 4-yr, zero- interest bearing note to Key Bank. The note was issued to yield 4% annual interest. On 12/31/18, Key Bank concluded that Pucket would probably not be able to repay the entire loan and considered the loan to be impaired. Key Bank estimates that Pucket will pay back only $650,000 of the principal at maturity. (Present Value Factors are below). Present value of 1 (Present value of a single sum) n 1 2 3 4 5 2% .98039 3% 4% .97087 .96154 96117 94260 .92456 .92385 .88949 .85480 5% .90573 .86261 .82193 .95238 94732 .91514 .88900 .86384 .83962 6% .90703 .89000 .82270 .94340 .78353 .79209 74726 On January 1, 2017 Pucket Co. issued a $950,000, 4-yr, zero- interest bearing note to Key Bank. The note was issued to yield 4% annual interest. On 12/31/18, Key Bank concluded that Pucket would probably not be able to repay the entire loan and considered the loan to be impaired. Key Bank estimates that Pucket will pay back only $650,000 of the principal at maturity. (Present Value Factors are below). Present value of 1 (Present value of a single sum) n 1 2 3 4 5 2% .98039 3% 4% .97087 .96154 96117 94260 .92456 .92385 .88949 .85480 5% .90573 .86261 .82193 .95238 94732 .91514 .88900 .86384 .83962 6% .90703 .89000 .82270 .94340 .78353 .79209 74726
Expert Answer:
Answer rating: 100% (QA)
Calculate the initial discount on the zerointerestbear... View the full answer

Related Book For 

Intermediate Accounting
ISBN: 978-0132162302
1st edition
Authors: Elizabeth A. Gordon, Jana S. Raedy, Alexander J. Sannella
Posted Date:
Students also viewed these accounting questions
-
On May 1, 2020, Jadeja Corporation, a publicly listed corporation, issued $200,000 of five-year, 8% bonds, with interest payable semi-annually on November 1 and May 1. The bonds were issued to yield...
-
On January 1, 2020, Offshore Corporation erected a drilling platform at a cost of $5,460,000. Offshore is legally required to dismantle and remove the platform at the end of its six-year useful life,...
-
Buchanan Corporation issues $500,000 of 11% bonds that are due in 10 years and pay interest semi-annually. At the time of issue, the market rate for such bonds is 10%. Calculate the bonds' issue...
-
MW Canada developed a 90-hour technical training program that was delivered over two months. Because it enabled the loom mechanics to act more independently with less downtime if the total cost was...
-
A firms current profits are $550,000. These profits are expected to grow indefinitely at a constant annual rate of 5 percent. If the firms opportunity cost of funds is 8 percent, determine the value...
-
In the chi-square independence test, the expected frequency for each class must always be _______.
-
A 6061-T6 aluminum alloy solid circular rod of length \(4 \mathrm{~m}\) is pinned at one end while fixed at the other end. If it is subjected to an axial load of \(15 \mathrm{kN}\) and F.S. \(=2\)...
-
During the last week of August, Apache Arts Companys owner approaches the bank for an $80,000 loan to be made on September 2 and repaid on November 30 with annual interest of 12%, for an interest...
-
What is the 32-bit version of the following signed number: 1000 0000 0001 0101
-
The data shown in Table 3E.2 are chemical process yield readings on successive days (read down, then across). Construct a histogram for these data. Comment on the shape of the histogram. Does it...
-
Two independent harmonic oscillators of equal mass are oscillating about the origin with angular frequencies and 2 and have total energies E, and E2, respectively. The variations of their momenta p...
-
Cash Budget For the Year Ended December 31, 2013 Increase in Cash Decrease in Cash Total Cash receipts $9,172,846 Cash payments for materials $(5,338,660) Wages and commissions paid $(1,616,693)...
-
Greer Utsey earned $47,500 in 2022 for a company in Kentucky. Greer is single with one dependent under 17 and is paidweekly. The FUTA rate in Kentucky for 2022 is 0.6 percent on the first $7,000 of...
-
You will continue to help M&M family in building a successful financial plan. Once again, if applicable, please consider apply the same concept to you own financial planning to build healthy...
-
Imagine you have just assumed the role of CFO at Canova Manufacturing, a prominent company poised for dynamic expansion. The Board of Directors has adamantly conveyed that dilution of the current...
-
What is a budget? How does revenue relate to a budget? Explain two consequences of having revenue being too far from budget. Why is it better to use percentages for revenue performance instead of...
-
Record the December 31 adjusting entries for the following transactions and events in general journal form. Assume that December 31 is the end of the annual accounting period. (5 p.) a. The Prepaid...
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
What factors may indicate that a promise to deliver a good or service is not separate from other promises?
-
Indicate which rate and time period you would use in order to select the correct interest factor for the following situations. In addition, indicate the interest factor that would be used. a. Using...
-
Complete the requirements of E22-10 using the direct method. In E22-10 Star land Corporation provided the following comparative balance sheets and income statement. Star land Corporation Income...
-
The freezing point of pure benzene is \(5.44^{\circ} \mathrm{C}\) and that of the solution containing \(2.092 \mathrm{~g}\) of benzaldehyde in \(100 \mathrm{~g}\) of benzene is \(4.44^{\circ}...
-
The molality of dissolved gases in water at \(0^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\) is \(1.29 \times 10^{-3}\). The decrease in volume during melting of ice is \(0.0907 \mathrm{cc} /...
-
Calculate the mass of methyl alcohol which, when dissolved in \(100 \mathrm{~g}\) of water, would just prevent the formation of ice at \(-10^{\circ} \mathrm{C}\), given that \(K_{\mathrm{f}}\) is...

Study smarter with the SolutionInn App


