One mole of solid silver superheated to 1000 C is allowed to melt at the same...
Fantastic news! We've Found the answer you've been seeking!
Question:
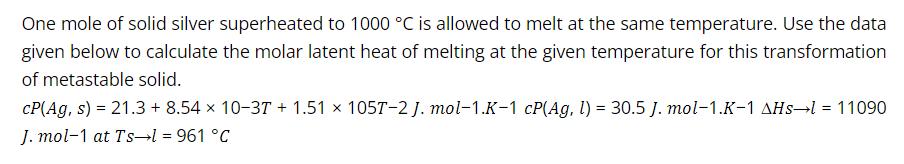
Transcribed Image Text:
One mole of solid silver superheated to 1000 °C is allowed to melt at the same temperature. Use the data given below to calculate the molar latent heat of melting at the given temperature for this transformation of metastable solid. CP(Ag, s) = 21.3 + 8.54 x 10-37 + 1.51 x 105T-2 J. mol-1.K-1 cP(Ag, l) = 30.5 J. mol-1.K-1 AHs-l = 11090 J. mol-1 at Ts l = 961 °C One mole of solid silver superheated to 1000 °C is allowed to melt at the same temperature. Use the data given below to calculate the molar latent heat of melting at the given temperature for this transformation of metastable solid. CP(Ag, s) = 21.3 + 8.54 x 10-37 + 1.51 x 105T-2 J. mol-1.K-1 cP(Ag, l) = 30.5 J. mol-1.K-1 AHs-l = 11090 J. mol-1 at Ts l = 961 °C
Expert Answer:
Answer rating: 100% (QA)
Date 1 20 Page No Guien Molei of Solid delver I mod Meltingpaint of Silber 961C Heat o... View the full answer

Posted Date:
Students also viewed these accounting questions
-
One mole of solid Cr 2 O 3 at 2500 K is dissolved in a large volume of a liquid Raoultian solution of Al 2 O 3 and Cr 2 O 3 in which X Cr2O3 = 0.2 and which is also at 2500 K. Calculate the changes...
-
One mole of solid gold is raised from 25 C to 100 C at constant pressure. Cp/(J/K mol) = 23.7 + 0.00519T. Calculate S for the transformation.
-
Two containers hold an ideal gas at the same temperature and pressure. Both containers hold the same type of gas, but container B has twice the volume of container A. (i) What is the average...
-
It is stated in Section 40.3 that a finite potential well always has at least one bound level, no matter how shallow the well. Does this mean that as U 0 0, E 1 0? Does this violate the Heisenberg...
-
How does the auditor communicate internal control deficiencies to management and the audit committee in a financial statement audit?
-
Consider a linear time-invariant system whose impulse response is real and is given by h[n]. Suppose the responses of the system to the two inputs x[n] and v[n] are, respectively, y[n] and z[n], as...
-
Which data entry application control would detect and prevent entry of alpha betic characters as the price of an item in the inventory master file? a. field check c. completeness check b. limit check...
-
Given the following set of cash flows: Period Cash Flow 1 $ 45,000 2 40,000 3 35,000 4 30,000 5 25,000 a. If your required rate of return is 7% per year, what is the present value of the above cash...
-
What is the compound yield on a Treasury bill that costs $98,920 and will be redeemed for $100,000 after 90 days? Assume 365 days in a year. Round your answer to one decimal place. % How much...
-
What characteristics of a property, plant, and equipment item make it different from other assets, such as accounts receivable or inventory?
-
A checking account bank statement normally includes ______. Multiple select question. the ending balance in the Cash account a list of checks drawn on the account during the period a list of...
-
(a) What is the cost of heating a hot tub containing 1540 kg of water from 10.0C to 40.0C, assuming 75.0% efficiency to take heat loss to surroundings into account? The cost of electricity is...
-
A company sells two products: microwaves and toaster ovens. The expected sales for microwaves were 3,000 units; 4,000 were sold. The budgeted selling price for microwaves was $78.00; however, the...
-
1.How would you define Logistics Management and Supply Chain management? Why is it important to have an effective and agile Supply Chain in our current business climate? 2. Consider the purchase of...
-
A rotating cylinder about 17 km long and 8.4 km in diameter is designed to be used as a Space Colony. Its rotations imitate the gravitational acceleration due to gravity as experienced on the Earth's...
-
Trisha is head of a team of three accountants at a non-profit agency that strives to promote literacy among inner city teenagers who drop out of high school. Trisha has held this position for over...
-
C D 4 kN B 60 A 25 3 kN 2m 5 kN E 55
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
Prove that if \(G\) is an undirected tree with more than one vertex, then \(G\) contains at least two vertices of degree 1 .
-
The graph below shows computer links between an official vote-tallying center at vertex 1 and several precincts. For the sake of secrecy, links can be made secure, but since this is an expensive...
-
Finish the proof of Theorem 1, that is, if \(G\) is a connected graph with \(n\) vertices and \(n-1\) edges, then \(G\) is a tree.

Study smarter with the SolutionInn App

