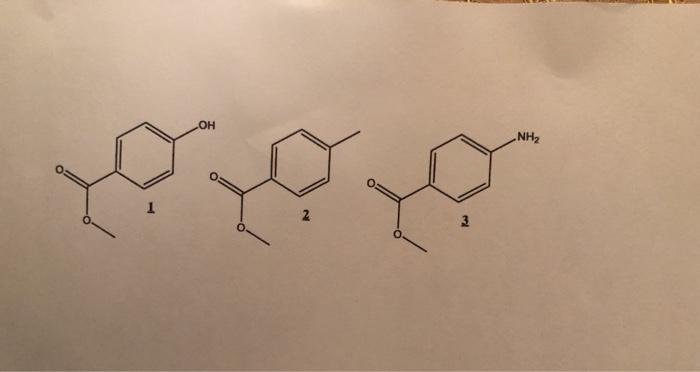
Organics, 1, 2 and 3 were mixed (500 mg of each of the products). You are in
Question:
Organics, 1, 2 and 3 were mixed (500 mg of each of the products). You are in a chemistry lab. You have at your disposal all the necessary equipment (glassware, filter etc.), all the aqueous solutions (acids, bases etc.) required, the usual organic solvents (Et2O, ACOET, acetone, hexanes etc.) and the usual chemicals. (desiccants etc.). Develop a strategy for separating these three products by liquid extraction. At all times, justify the response in terms of the pH of the solution used, pKa involved, chemical reaction involved, equilibrium constant of the reaction. At the end of the protocol, the three products are in their neutral form (not in the protonated form or in the deprotonated form).

Advanced Financial Accounting
ISBN: 978-0078025624
10th edition
Authors: Theodore E. Christensen, David M. Cottrell, Richard E. Baker




