Urban Glam Cosmetics made purchases of lipstick in the current year as follows: 81 units e...
Fantastic news! We've Found the answer you've been seeking!
Question:
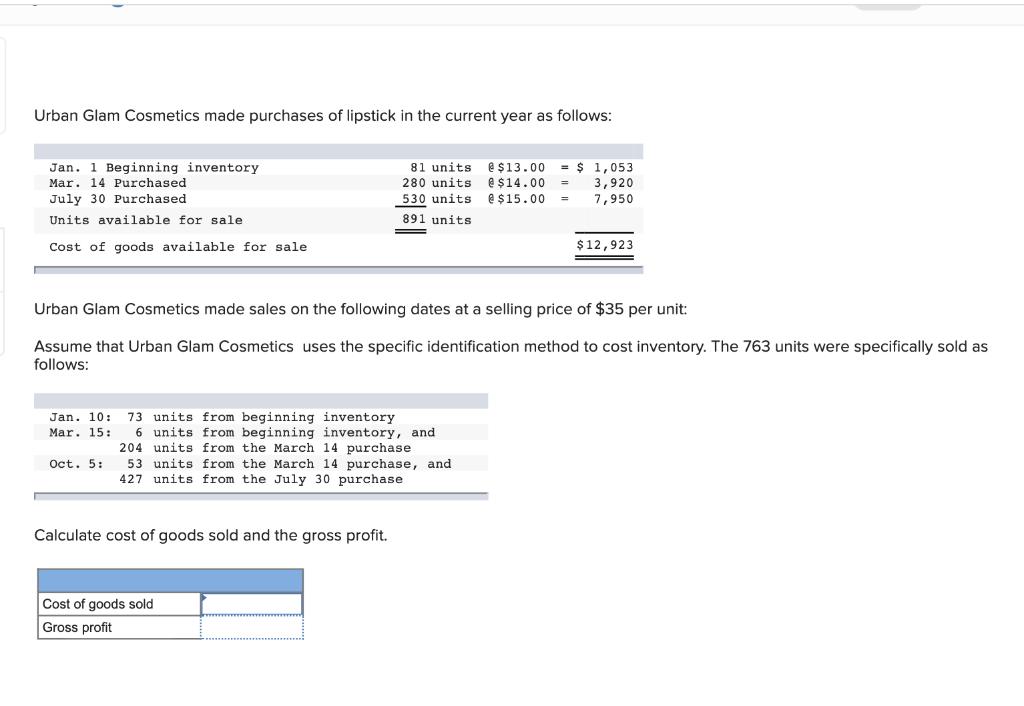
Transcribed Image Text:
Urban Glam Cosmetics made purchases of lipstick in the current year as follows: 81 units e $13.00 $ 1,053 3,920 7,950 Jan. 1 Beginning inventory Mar. 14 Purchased 280 units e$14.00 July 30 Purchased 530 units e $15.00 Units available for sale 891 units Cost of goods available for sale $12,923 Urban Glam Cosmetics made sales on the following dates ta selling price of $35 per unit: Assume that Urban Glam Cosmetics uses the specific identification method to cost inventory. The 763 units were specifically sold as follows: 73 units from beginning inventory 6 units from beginning inventory, and 204 units from the March 14 purchase 53 units from the March 14 purchase, and 427 units from the July 30 purchase Jan. 10: Mar. 15: Oct. 5: Calculate cost of goods sold and the gross profit. Cost of goods sold Gross profit Urban Glam Cosmetics made purchases of lipstick in the current year as follows: 81 units e $13.00 $ 1,053 3,920 7,950 Jan. 1 Beginning inventory Mar. 14 Purchased 280 units e$14.00 July 30 Purchased 530 units e $15.00 Units available for sale 891 units Cost of goods available for sale $12,923 Urban Glam Cosmetics made sales on the following dates ta selling price of $35 per unit: Assume that Urban Glam Cosmetics uses the specific identification method to cost inventory. The 763 units were specifically sold as follows: 73 units from beginning inventory 6 units from beginning inventory, and 204 units from the March 14 purchase 53 units from the March 14 purchase, and 427 units from the July 30 purchase Jan. 10: Mar. 15: Oct. 5: Calculate cost of goods sold and the gross profit. Cost of goods sold Gross profit
Expert Answer:
Answer rating: 100% (QA)
cost of goods sold Cost of goods available for sales 12923 Less ending inven... View the full answer

Related Book For 

Federal Taxation 2016 Comprehensive
ISBN: 9780134104379
29th edition
Authors: Thomas R. Pope, Timothy J. Rupert, Kenneth E. Anderson
Posted Date:
Students also viewed these accounting questions
-
Paddington Gifts made purchases of a particular product in the current year as follows: Required: The business uses a periodic inventory system. Ending inventory consists of 120 units. Calculate the...
-
Jasper & Williams made purchases of a particular product in the current year as follows: Required: Ending inventory consists of 155 units. Assuming a periodic system, determine the costs to be...
-
Parfour made purchases of a particular product in the current year as follows: Parfour made sales on the following at a selling price of $35 per unit: Required The business uses p perpetual inventory...
-
(a) Alison has been self-employed for many years. Her profit for tax year 2020-21 is 6,380. Is she liable to pay Class 2 NICs for the year? (b) Amanda starts a business on 25 May 2020 and she is...
-
Implement the linear optimization model that you developed for the investment scenario use Solver to find an optimal solution. Save the Answer and Sensitivity reports in your Excel workbook....
-
1. Place yourself in Kevin's and Jeff's shoes. How do you respond to Reid concerning this delicate situation? Jeff and Kevin's first and only priority is the system automation projects, but if there...
-
Form a small group (four to six people) and carry out an interview role play for this position. Half of the team should represent the company and the other half act as applicants.
-
Lavage Rapide is a Canadian company that owns and operates a large automatic carwash facility near Montreal. The following table provides data concerning the companys costs: For example, electricity...
-
As a new digital designer, your job is to develop a calculator for a number system. In this particular task, given a 4-bit number X3X2X1X, in a 2's complement system, design a circuit that takes the...
-
The Community Youth Center (CYC) Corporation failed to pay its annual registration fee in 2000 and was automatically dissolved by the State Corporation Commission. CYC continued to operate as a...
-
Durden Soap manufactures Bar soap and Liquid soap. Certain ingredients are shared, and combined before going to each department. A batch of 100 gallons of pre-mix results in 40 gallons to the Bar...
-
What are the common characteristics of a heat engine?
-
Comment on the statement: "Heat cannot be easily converted into work".
-
Is it possible to achieve a reversible heat engine in practice?
-
What is entropy? Give an example.
-
One mol of nitrogen at \(25^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\) is allowed to expand reversibly to a volume of \(50 \mathrm{dm}^{3}\). If the gas is assumed to be ideal, calculate the final...
-
explain the historical events leading to the development of Equity - Key historical events that led to the creation of equity and common law - The significance of Earl of Oxford's case - The...
-
Describe the general ways that the revised Form 990, applicable for tax year 2008 and beyond, is different from previous versions.
-
Phil, a cash-basis taxpayer, sells the following marketable securities, which are capital assets during 2015. Determine whether the gains or losses are long-term or short-term. Also determine the net...
-
Jackie purchases equipment during the current year for $800,000 that has a seven-year MACRS recovery period. She expects to sell the property after three years. Jackie anticipates that her marginal...
-
Alabama Corporation conducts a copper mining business. During the current year, it reports regular taxable income of $400,000, which includes a $100,000 deduction for percentage depletion. The...
-
A system, consisting of \(2 \mathrm{~mol}\) of \(\mathrm{N}_{2}, 5 \mathrm{~mol}\) of \(\mathrm{H}_{2}\) and \(2 \mathrm{~mol}\) of \(\mathrm{NH}_{3}\) initially, is undergoing the following...
-
The following reaction takes place in a system consisting of \(3 \mathrm{~mol} \mathrm{CH}_{4}, 5 \mathrm{~mol} \mathrm{H}_{2} \mathrm{O}\), \(1 \mathrm{~mol} \mathrm{CO}\) and \(4 \mathrm{~mol}...
-
The study of thermodynamics enables us to understand (a) Whether the transformation of energy is feasible or not (b) To what extent the transformation will take place (c) In which direction the...

Study smarter with the SolutionInn App


