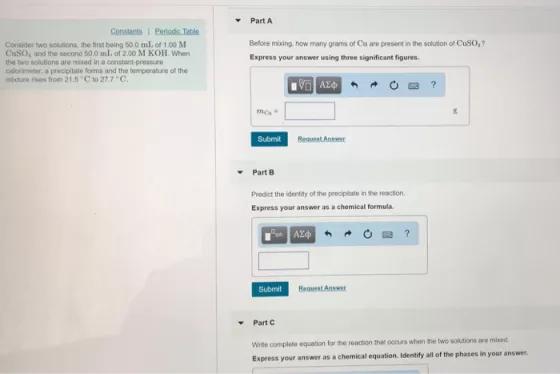
Part A Constant Petode Tatle Conder two soona he t being so o ml. of 1.00...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part A Constant Petode Tatle Conder two soona he t being so o ml. of 1.00 M CSO, nd the secono 00 ml. of 2.00 M KOH When thewolutons are med a cnstart pre r.a precipiute oma and the bemperature of the ure fo 21 C 217 C. Beflore mising how matny gras of Cu are presentin the scuton or CuS0, Expreas your anewer uning three significant figures AZO Submit RenatAntr Part 8 Prediet the identity of the preciptate n the acton. Espress your answer asa chemical formula Submit HemtAn Part C Wite complete equation tar he seaction that ocns when the two wtions misd Express your answer as a chemical equation. Identify all of the phases in your answer Part A Constant Petode Tatle Conder two soona he t being so o ml. of 1.00 M CSO, nd the secono 00 ml. of 2.00 M KOH When thewolutons are med a cnstart pre r.a precipiute oma and the bemperature of the ure fo 21 C 217 C. Beflore mising how matny gras of Cu are presentin the scuton or CuS0, Expreas your anewer uning three significant figures AZO Submit RenatAntr Part 8 Prediet the identity of the preciptate n the acton. Espress your answer asa chemical formula Submit HemtAn Part C Wite complete equation tar he seaction that ocns when the two wtions misd Express your answer as a chemical equation. Identify all of the phases in your answer
Expert Answer:
Answer rating: 100% (QA)
A 500 mL of 10 M of cusoy molculay mass 9756 mass X100 o Molanity is defined as ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
A solution of 100.0 mL of 0.200 M KOH is mixed with a solution of 200.0 mL of 0.150 M NiSO4. (a) Write the balanced chemical equation for the reaction that occurs. (b) What precipitate forms? (c)...
-
How many grams of KOH are present in 35.0 mL of a 5.50 M solution?
-
How do players purchase virtual goods? Identify three virtual currencies and their value in U.S. dollars. Who would pay $330,000 for a virtual space station? Or $100,000 for an asteroid space resort?...
-
PIM Industries, Inc., manufactures electronics components. Each unit costs $30 before the final test. The final test rejects, on average, 5 percent of the 50,000 units manufactured per year. The...
-
How are foreign operations defined under SFAS No. 131 [ASC 280]?
-
Add the given vectors by components. The displacement vectors in Fig. 9.29. 25.0 22.3 km 62.6 41.5 km 42.2 Fig. 9.29 X 51.6 km
-
The feed (equimolar A and B) to a reactor is heated from \(100^{\circ} \mathrm{F}\) to \(500^{\circ} \mathrm{F}\) in a \(1-2\) parallel-counterflow heat exchanger with a mean overall heat-transfer...
-
A recent annual report for FedEx contained the following data: Required: 1. Determine the accounts receivable turnover ratio and average days sales in receivables for the current year. 2. Explain the...
-
When owning and operating any business, whether it's a caf or a pool cleaning business, it's very important that you know exactly what your income and expenditure is. Is your business making money or...
-
Montreal Electronics Company manufactures two large-screen television models: the Nova, which has been produced for 10 years and sells for $900, and the Royal, a new model introduced in early 20x0,...
-
TOPIC: STRATEGIES TO CONNECT AT YOUR WORKPLACE Give the following: 1. Introduction - What the topic is all about, its main objectives, importance, and general ideas. - Increase the interest of the...
-
Acceptance is timely if it is made before an offer terminates. (True/False)
-
An adhesion contract will never be deemed unconscionable. (True/False)
-
An illegal contract is valid unless it is executory. (True/False)
-
The tort of defamation does not occur unless a defamatory statement is made in writing. (True/False)
-
Legitimate competitive behavior does not constitute wrongful interference with a contractual relationship. (True/False)
-
A new Community Center is being built in Hadleyville. The perimeter of the rectangular playing field is 434 yards. The length of the field is 7 yards less than triple the width. What are the...
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
Four of the boxes in the following periodic table are colored. Which of these are metals and which are nonmetals? Which one is an alkaline earth metal? Which one is a noble gas?
-
(a) Define atomic number and mass number. (b) Which of these can vary without changing the identity of the element?
-
In CF3Cl the C-Cl bond-dissociation energy is 339 kJ / mol. In CCl4 the bond-dissociation energy is 293 kJ / mol. What is the range of wavelengths of photons that can cause C-Cl bond rupture in one...
-
Tesco plc is one of the worlds largest food retailers. Fiscal year 2014 (the year ended February 28, 2015) was a rocky year for the retailer. The companys sales and margins had come under pressure as...
-
Exhibit P2 displays a selected set of financial ratios for the years 20152017 of three fashion retailers: USbased Gap, UK-based Next, and Japan-based Uniqlo (Fast Retailing). Using this set of...
-
In 2009, following the worldwide credit crisis, several US-based car manufacturers, such as Chrysler and General Motors, approached bankruptcy and needed to be bailed out by the US government and...

Study smarter with the SolutionInn App


