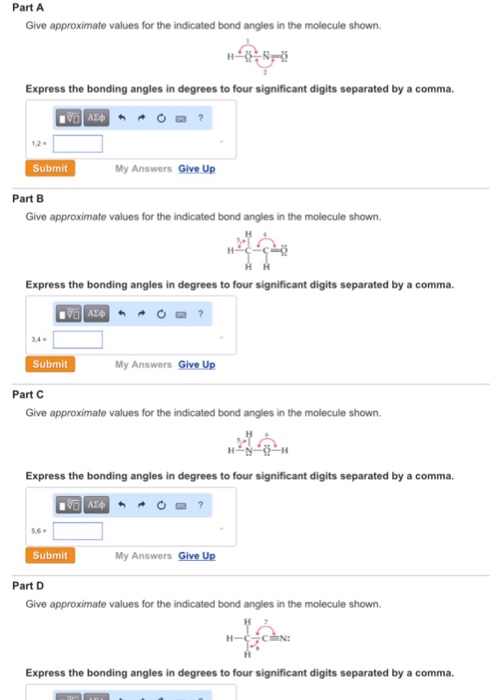
Part A Give approximate values for the indicated bond angles in the molecule shown. SA Express...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part A Give approximate values for the indicated bond angles in the molecule shown. „ŠSA Express the bonding angles in degrees to four significant digits separated by a comma. 180 ΑΣΦΑ 1,2- Submit Part B Give approximate values for the indicated bond angles in the molecule shown. Express the bonding angles in degrees to four significant digits separated by a comma. VAX ? 3,4- Submit My Answers Give Up Part C Give approximate values for the indicated bond angles in the molecule shown. 5,6- Express the bonding angles in degrees to four significant digits separated by a comma. Submit My Answers Give Up AXO My Answers Give Up Part D Give approximate values for the indicated bond angles in the molecule shown. Já Express the bonding angles in degrees to four significant digits separated by a comma. Part A Give approximate values for the indicated bond angles in the molecule shown. „ŠSA Express the bonding angles in degrees to four significant digits separated by a comma. 180 ΑΣΦΑ 1,2- Submit Part B Give approximate values for the indicated bond angles in the molecule shown. Express the bonding angles in degrees to four significant digits separated by a comma. VAX ? 3,4- Submit My Answers Give Up Part C Give approximate values for the indicated bond angles in the molecule shown. 5,6- Express the bonding angles in degrees to four significant digits separated by a comma. Submit My Answers Give Up AXO My Answers Give Up Part D Give approximate values for the indicated bond angles in the molecule shown. Já Express the bonding angles in degrees to four significant digits separated by a comma.
Expert Answer:
Answer rating: 100% (QA)
Bond angle depends on hybridisation and electronegetivity of central atom and presence of lone pairs ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
Give the approximate values for the indicated bond angles in the following molecules: (a) (b) (c) (d) 4 3 6 H-COCH 7 H
-
Give approximate values for the indicated bond angles in the following molecules: a. b. c. d. 1 H- 2.
-
Approximate values for the radius of a gold atom and the radius of a gold nucleus are 10 10 m and 10 15 m, respectively. Estimate the ratio of the volume of a gold atom to the volume of a gold...
-
How would your answers in Problem 48 change if partnership revenues were $100,000 instead of $150,000? Data From Problem 48: The KL Partnership is owned equally by Kayla and Lisa. Kaylas basis is...
-
Manitoba Paper Company packages paper for photocopiers. The company has developed standard over-head rates based on a monthly practical capacity of 90,000 direct-labor hours as follows: Standard...
-
What is the magnitude of the electric field produced by a charge of magnitude 4.50 C at a distance of (a) 1.00 m (b) 3.00 m?
-
Nielsen Media Research earns money by charging television stations and networks for its services. For example, NBC pays Nielsen to provide ratings for its television shows. Why doesnt NBC simply do...
-
Logan B. Taylor is a widower whose wife, Sara, died on June 6, 2011. He lives at 4680 Dogwood Lane, Springfield, MO 65801. He is employed as a paralegal by a local law firm. During 2013, he had the...
-
Adaptability is the predominate tension for a college in these days of intense competition for "customers" (students) and faculty, right?But, be adaptable to what?Wouldn't the college have to develop...
-
To keep track of office furniture, computers, printers, and so on, the FOUNDIT Company uses the table structure shown in Table P6.5. a. Given that information, write the relational schema and draw...
-
Compare modified accrual to accrual accounting basis with regard to encumbrance accounting 23. How expenditures, encumbrances, and transfers affect fund balance
-
consists of an The 2000 mm long composite bar shown in Figure aluminum bar having a modulus of elasticity EA = 70 GPa and length LAI= = 500 mm, which is securely fastened to a steel bar having...
-
Q3. According to the schematic diagram and experimental results shown below. determine the radius of gyration (k), and the moment of inertia about G. Knowing that motion is clearly simple harmonic...
-
A system of force is applied on the structure on the figure shown. Determine the following: 14. Equivalent force of the force system. 15. Distance where the line of action intersects member AB,...
-
3. A vector has a magnitude of 50 kN and points away from the origin in the 2nd quadrant making an angle of 30 degrees from the positive y axis. Determine the vertical vector which, when added to...
-
A uniform bar of length L = 1 m is moving in a zero gravity, zero atmosphere field at a uniform speed of 10 m/s as shown by the arrows. Initially parallel to the X direction, the bar is not rotating...
-
Suppose a speculator has $100,000 available for speculative purposes. The current spot exchange rate is $1.70/1. A 3-months call option on the pound is available to purchase at $1.80/1, and the...
-
In the circuit shown in Figure 4, a battery supplies a constant voltage of 40 V, the inductance is 2 H, the resistance is 10, and l(0) = 0. (a) Find l(t). (b) Find the current after 0.1s.
-
Which type (or types) of crystalline solid is characterized by each of the following: (a) High mobility of electrons through out the solid; (b) Softness, relatively low melting point; (c) High...
-
Propane, C3H8, liquefies under modest pressure, allowing a large amount to be stored in a container. (a) Calculate the number of moles of propane gas in a 110-L container at 3.00 atm and 27oC. (b)...
-
The hydrocarbons acetylene (C2H2) and benzene (C6H6) have the same empirical formula. Benzene is an "aromatic" hydrocarbon, one that is unusually stable because of its structure. (a) By using the...
-
Brandywine Homecare, a not-for-profit business, had revenues of \($12\) million in 2015. Expenses other than depreciation totaled 75 percent of revenues, and depreciation expense was \($1.5\)...
-
Bright Horizons Skilled Nursing Facility, an investor-owned company, constructed a new building to replace its outdated facility. The new building was completed on January 1, 2015, and Bright...
-
What is the difference between long-term debt and current portions of long-term debt?

Study smarter with the SolutionInn App


