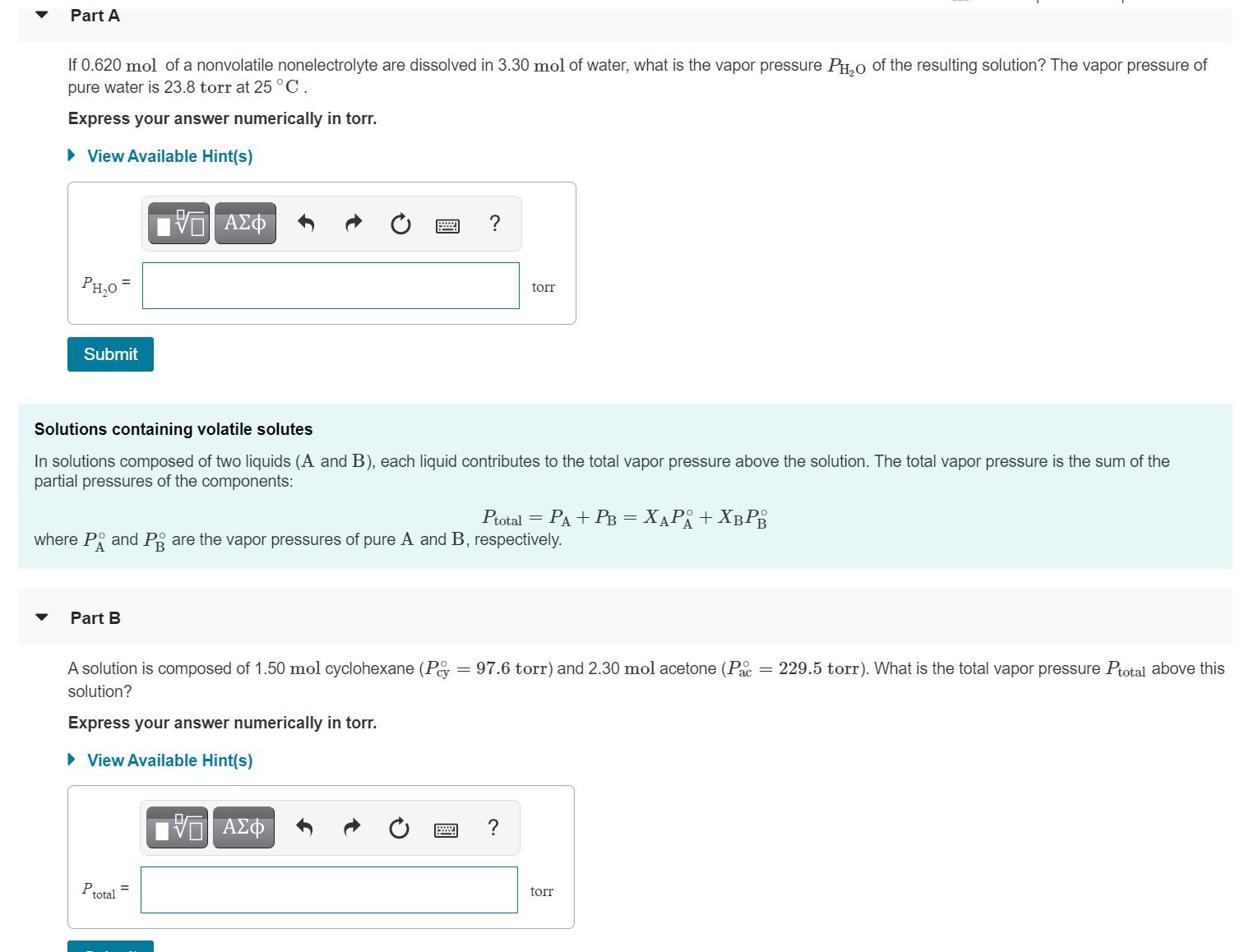
Part A If 0.620 mol of a nonvolatile nonelectrolyte are dissolved in 3.30 mol of water,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Part A If 0.620 mol of a nonvolatile nonelectrolyte are dissolved in 3.30 mol of water, what is the vapor pressure PH,0 of the resulting solution? The vapor pressure of pure water is 23.8 torr at 25 °C. Express your answer numerically in torr. • View Available Hint(s) ? PH,0 = torr Submit Solutions containing volatile solutes In solutions composed of two liquids (A and B), each liquid contributes to the total vapor pressure above the solution. The total vapor pressure is the sum of the partial pressures of the components: Potal 3D Pа + Рв 3 ХАРА + Х в РЕ = where P and Pg are the vapor pressures of pure A and B, respectively. Part B A solution is composed of 1.50 mol cyclohexane (Pey 97.6 torr) and 2.30 mol acetone (P = 229.5 torr). What is the total vapor pressure Ptotal above this solution? Express your answer numerically in torr. • View Available Hint(s) V AZO ? Ptotal torr 圓 Part A If 0.620 mol of a nonvolatile nonelectrolyte are dissolved in 3.30 mol of water, what is the vapor pressure PH,0 of the resulting solution? The vapor pressure of pure water is 23.8 torr at 25 °C. Express your answer numerically in torr. • View Available Hint(s) ? PH,0 = torr Submit Solutions containing volatile solutes In solutions composed of two liquids (A and B), each liquid contributes to the total vapor pressure above the solution. The total vapor pressure is the sum of the partial pressures of the components: Potal 3D Pа + Рв 3 ХАРА + Х в РЕ = where P and Pg are the vapor pressures of pure A and B, respectively. Part B A solution is composed of 1.50 mol cyclohexane (Pey 97.6 torr) and 2.30 mol acetone (P = 229.5 torr). What is the total vapor pressure Ptotal above this solution? Express your answer numerically in torr. • View Available Hint(s) V AZO ? Ptotal torr 圓
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A chemist mixes two liquids A and B to form a homogeneous mixture. The densities of the liquids are 2.0514 g/mL for A and 2.6678 g/mL for B. When she drops a small object into the mixture, she finds...
-
What is the vapor pressure of an aqueous solution of 36.4 g of KBr in 199.5 g of H2O if the vapor pressure of H2O at the same temperature is 32.55 torr? What other solute(s) would give a solution...
-
What is the vapor pressure at 23C of a solution of 1.20 g of naphthalene, C10H8, in 25.6 g of benzene, C6H6? The vapor pressure of pure benzene at 23C is 86.0 mmHg; the vapor pressure of naphthalene...
-
Reference frame S is moving along the x axis at 0.6c relative to frame S. A particle that is originally at x = 10 m at t 1 = 0 is suddenly accelerated and then moves at a constant speed of c/3 in...
-
The molecular weight of a particular polymer should fall between 2,100 and 2,350. Fifty samples of this material were analyzed with the results x = 2,275 and s = 60. Assume that molecular weight is...
-
Hagers Home Repair Company, a regional hardware chain, which specializes in do-it-yourself materials and equipment rentals, is considering an acquisition of Lyon Lighting (LL). Doug Zona, Hagers...
-
\(\frac{3}{8} \times\left(\frac{13}{12}-\frac{35}{36} ight)\) Perform the indicated operations. Reduce to lowest terms.
-
The completed financial statement columns of the worksheet for Hubbs Company are shown below. Instructions (a) Prepare an income statement, a retained earnings statement, and a classified balance...
-
ment 3 - Ch 3 Cash Accounts receivable Repair supplies Prepaid rent Office furniture Accounts payable Notes payable Trial Balances September 30, 2023 Unadjusted Trial Balance Saved Adjustments...
-
The balances in the general ledger accounts of Thunder Graphics are shown below. Instructions On the forms provided in your working papers: 1 . Open the accounts in the general ledger with their...
-
If the energy resolution of a scintillator is 9.4% at 662 keV, find the standard deviation (in energy units) of the Gaussian curve that would be a fit to the photopeak at that energy.
-
1. Tesla is beta testing its self-driving car using a select group of people in several different neighborhoods in the country. As a results, problems and unexpected events are very public. Metz...
-
5 Select a commercial, movie, or music video from the last three years. Write an approximately three-page, double- spaced analysis of the movie using Jung's archetypes/psychoanalytic criticism...
-
You are planning a trip on the ocean. On the ocean distance is measured in Nautical Miles (aka "knots"). A Nautical Mile is equal to 1.1508 Regular Miles. You would like to calculate the distance you...
-
Use the given f(x) and g(x) to evaluate each expression. f(x)=x+56, g(x) = x (a) What is the value of (fog)(5)? (fog)(5)= 9 (Simplify your answer.) (b) What is the value of (g of)(-3)? (gof)(-3)=...
-
The trend of making something yourself at home has been helped by the vast amounts of informationavailable on the Internet. The DiVito brothers are curious about whether people making their own...
-
Homework: Chapter 4 Homework Part 1 of 3 O Points: 0 of 1 ave As a real estate speculator, you are planning and able to buy a house that costs $200,000, borrowing the full amount with no money down...
-
Which of the companies has the lowest accounts receivable turnover in the year 20X2? a. Company A. b. Company B. c. Company C. d. CompanyD. 20X1 20X2 Credit Sales Average Receivables Balance $1.0...
-
A sample of green crystals of nickel(II) sulfate heptahydrate was heated carefully to produce the bluish green nickel(II) sulfate hexahydrate. What are the formulas of the hydrates? If 8.753 g of the...
-
Define each of the following: noble-gas core, pseudo-noble-gas core, valence electron.
-
Compounds A and B are known to be stereoisomers of one another. Compound A has a violet color; compound B has a green color. Are they geometric or optical isomers?
-
1.7 Limitations of Comparative Advantage. The key to understanding most theories is what they say and what they don't. Name four or five key limitations to the theory of comparative advantage.
-
1.6 Post-LIBOR Scandal. Why do you think the UK government has resolved against the total elimina- tion of LIBOR benchmarks after the scandal?
-
1.9 Aidan's Globalization. After reading the chapter's description of Aidan's globalization process, how would you explain the distinctions between interna- tional, multinational, and global...

Study smarter with the SolutionInn App