Isotonic saline solution, which has the same osmotic pressure as blood, can be prepared by dissolving...
Fantastic news! We've Found the answer you've been seeking!
Question:
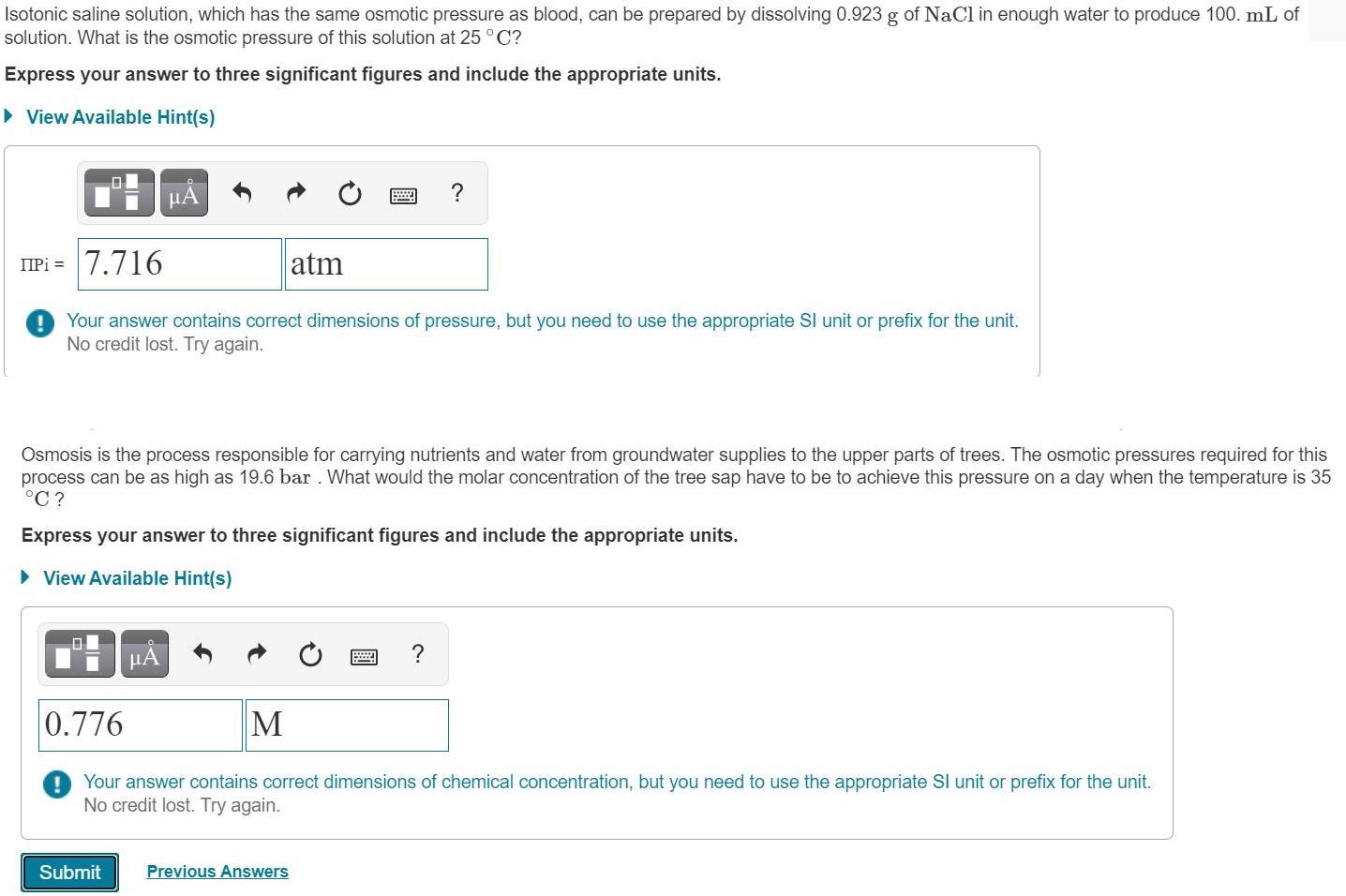
Transcribed Image Text:
Isotonic saline solution, which has the same osmotic pressure as blood, can be prepared by dissolving 0.923 g of NaCl in enough water to produce 100. mL of solution. What is the osmotic pressure of this solution at 25°C? Express your answer to three significant figures and include the appropriate units. • View Available Hint(s) HA ? IPi = 7.716 atm Your answer contains correct dimensions of pressure, but you need to use the appropriate Sl unit or prefix for the unit. No credit lost. Try again. Osmosis is the process responsible for carrying nutrients and water from groundwater supplies to the upper parts of trees. The osmotic pressures required for this process can be as high as 19.6 bar. What would the molar concentration of the tree sap have to be to achieve this pressure on a day when the temperature is 35 °C ? Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) HA ? 0.776 M Your answer contains correct dimensions of chemical concentration, but you need to use the appropriate Sl unit or prefix for the unit. No credit lost. Try again. Submit Previous Answers Isotonic saline solution, which has the same osmotic pressure as blood, can be prepared by dissolving 0.923 g of NaCl in enough water to produce 100. mL of solution. What is the osmotic pressure of this solution at 25°C? Express your answer to three significant figures and include the appropriate units. • View Available Hint(s) HA ? IPi = 7.716 atm Your answer contains correct dimensions of pressure, but you need to use the appropriate Sl unit or prefix for the unit. No credit lost. Try again. Osmosis is the process responsible for carrying nutrients and water from groundwater supplies to the upper parts of trees. The osmotic pressures required for this process can be as high as 19.6 bar. What would the molar concentration of the tree sap have to be to achieve this pressure on a day when the temperature is 35 °C ? Express your answer to three significant figures and include the appropriate units. > View Available Hint(s) HA ? 0.776 M Your answer contains correct dimensions of chemical concentration, but you need to use the appropriate Sl unit or prefix for the unit. No credit lost. Try again. Submit Previous Answers
Expert Answer:

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemistry questions
-
Osmosis is the process responsible for carrying nutrients and water from groundwater supplies to the upper parts of trees. The osmotic pressures required for this process can be as high as 18.1atm....
-
Which of the isomeric C4H10O alcohols can be prepared by hydrogenation of aldehydes? Which can be prepared by hydrogenation of ketones? Which cannot be prepared by hydrogenation of a carbonyl...
-
A compound known as Hagemann's ester can be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid and heat. Write the structure for the product of...
-
Governmental Funds Statement of Revenues Expenditures and Changes in Fund Balance. You have recently started working as the controller for a small county. The county is preparing its financial...
-
A process is in statistical control with x = 202.5 and s = 2.0. Specifications are at LSL = 196 and USL = 206. (a) Estimate the process capability with an appropriate process capability ratio. (b)...
-
The Jimenez Corporations forecasted 2022 financial statements follow, along with some industry average ratios. Calculate Jimenezs 2022 forecasted ratios, compare them with the industry average data,...
-
\(\frac{48}{143} \times \frac{77}{120}\) Perform the indicated operations. Reduce to lowest terms.
-
Danny Badens Verde Vineyards in Oakville, California, produces three varieties of wine: Merlot, Viognier, and Pinot Noir. His winemaster, Russel Hansen, has identified the following activities as...
-
Lindstrom Company produces two fountain pen models. Information about its products follows: Sales revenue Less: Variable costs Contribution margin Total units sold Lindstrom's fixed costs total...
-
Preble Company manufactures one product. Its variable manufacturing overhead is applied to production based on direct labor-hours and its standard cost card per unit is as follows: Direct material: 5...
-
A company seeking investment opportunities has collected the following information: Project A Project B Project C Project D Initial investment $520,000 $360,000 $270,000 $390,000 PV of cash inflows...
-
Explain in your own words - (a) what is the error in the following function definition and (b) how can you fix the function without changing the function header? Your answer should be in plain...
-
Choose another Federal Agency. Make a chart (not a list- a visual exhibit) comparing the FOIA process for that agency and the USCIS. Include the differences and similarities in the process.?
-
The government is considering imposing an import quota on a product that is also produced domestically. Demonstrate that the quota would have different effects on domestic consumers and producers....
-
The diversity of both students and faculty poses important considerations for teaching and learning. Reflect on the characteristic differences in gender, race, and culture, as well as the differences...
-
What is the research objective for the following study? Please read the study....
-
The potential for harm or compromise to the supply chain that arises because of risk from suppliers, their supply chains, and their products or services refers to _____? Supply Chain Vulnerability,...
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
Predict the outcome of the following reactions: (a) (b) CN CI 2 equiv. KNH2 liq. NH3 -33 C NH CH3 2 equiv. NaNH liq. NH2 -33C Cl
-
The following structure is an intermediate in a synthesis of prostaglandins F2a and E2 by E. J. Corey (Harvard University). A Horner-Wadsworth-Emmons reaction was used to form the (E)-alkene. Write...
-
The compounds in each part below have the same (or similar) molecular weights. Which compound in each part would you expect to have the higher boiling point? Explain your answers. (a) (b) (c) OH or...
-
1.5 Moscow to Tokyo. After spending a week in Moscow, you get an email from your friend in Japan. He can get you a very good deal on a plane ticket and wants you to meet him in Tokyo next week to...
-
1.6 Mark Du Plessis in Johannesburg. Mark Du Plessis lives in Johannesburg, South Africa. He can buy a euro for R16.3700 (South African rand). At the same time, Richard Nolan, living in Paris, can...
-
1.7 Mexico's Cada Seis Aos. Mexico was famous-or infamous-for many years for having two things every six years (cada seis aos in Spanish): a presi- dential election and a currency devaluation. This...

Study smarter with the SolutionInn App


