Part A What is the width of the slit? a = You have been asked to measure
Fantastic news! We've Found the answer you've been seeking!
Question:
Part A
What is the width of the slit?
| a = |
Transcribed Image Text:
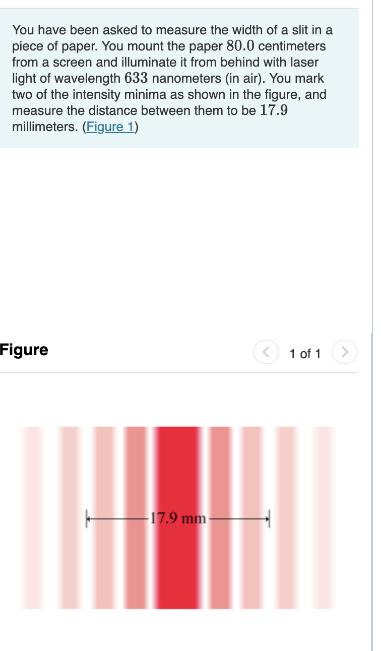
You have been asked to measure the width of a slit in a piece of paper. You mount the paper 80.0 centimeters from a screen and illuminate it from behind with laser light of wavelength 633 nanometers (in air). You mark two of the intensity minima as shown in the figure, and measure the distance between them to be 17.9 millimeters. (Figure 1) Figure -17.9 mm- 1 of 1 You have been asked to measure the width of a slit in a piece of paper. You mount the paper 80.0 centimeters from a screen and illuminate it from behind with laser light of wavelength 633 nanometers (in air). You mark two of the intensity minima as shown in the figure, and measure the distance between them to be 17.9 millimeters. (Figure 1) Figure -17.9 mm- 1 of 1
Expert Answer:
Answer rating: 100% (QA)
A the diagram given below H 24 Slit width a MDX y a 3x08 x 633x159 W 2 2DX 895103 a 1697... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A single slit 1.0mm wide is illuminated by 450-nm light, what is the width of the central maximum (in cm) in the diffraction pattern on a screen 5.0m away?
-
A slit of width 0.20 mm is illuminated with monochromatic light of wavelength 480 nm, and a diffraction pattern is formed on a screen 1.0 m from the slit. (a) What is the width of the central...
-
The diffraction pattern from a single slit is viewed on a screen. Using blue light, the width of the central maximum is 2.0 cm. (a) Would the central maximum be narrower or wider if red light is used...
-
During the year ended 30 June 20X5, Gem Stone Ltd acquired two areas of interest Site A and Site B. The exploration and evaluation activities at Site A amounted to $64454401. The costs relate to the...
-
Costco Wholesale is the national membership-warehouse retailer. It has 58 million members and 147,000 employees. Foot Locker sells athletic footwear and clothes in its 3,500 stores in the United...
-
Spring scale A is attached to the ceiling. A 10.0-kg mass is suspended from the scale. A second spring scale, B, is hanging from a hook at the bottom of the 10.0-kg mass and a 4.0-kg mass hangs from...
-
Refer to Exercise 12. Can you reject H0 at the = 0.05 level? State a conclusion. Exercise 12 A simple random sample of 17 business majors from a certain university had a mean GPA of 2.81 with a...
-
Color Florists, a retail business, had the following cash receipts during January 20--. The sales tax is 5%. Jan. 1 Received payment on account from Ray Boyd, $880. 3 Received payment on account from...
-
Porter Corporation reports the following components of stockholders' equity on January 1. Common stock-$10 par value, 110,000 shares authorized, 40,000 shares issued and outstanding Paid-in capital...
-
At December 31, 20X0, Oettinger Corporation, a premium kitchen cabinetmaker for the home remodeling industry, reported the following accounts receivable information on its year-end balance sheet:...
-
4. In the months leading up to Groupons IPO, the SEC posed a number of questions regarding Groupons choice of accounting principles for revenue recognition. Specifically, the SEC referred to the...
-
What are the two types of files that auditors review in preparation for an audit? What are the contents of each file?
-
What are the five objectives of internal control systems?
-
How does the Standards describe the scope (in general terms) of an internal audit?
-
Outline the key aspects of the preliminary communication for an audit.
-
What three types of audit approval are given for audits? Discuss each briefly.
-
PCB Electronics manufactures small custom electronic assemblies. Units must be processed through four stations: assembly, soldering, painting, and inspection. Orders inter-arrival times follow an...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
The lightest solid material is silica aerogel, which has a typical density of only about .010 g/cm3. The molecular structure of silica aerogel is typically 95% empty space. What is the mass of 1 m3...
-
The spout heights in the container in Fig. 9.43 are 10 cm, 20 cm, 30 cm, and 40 cm. The water level is maintained at a 45-cm height by an outside supply. (a) What is the speed of the water out of...
-
The engines of most rockets produce a constant thrust (forward force). However, when a rocket is fired, its acceleration increases with time as the engine continues to operate. Is this situation a...
-
The heat of vaporization of ether is \(25.98 \mathrm{~kJ} / \mathrm{mol}\) at its boiling point, \(34.5^{\circ} \mathrm{C}\). (a) Calculate the rate of change of vapour pressure with temperature...
-
Show that the internal energy of an ideal gas is a function only of temperature.
-
For a gas obeying the equation of state \(V=B+\frac{R T}{P}\), the Joule-Thomson coefficient is given by \[ \mu_{\mathrm{JT}}=-\frac{1}{C_{P}}\left(T \frac{d B}{d T}-B ight) \]

Study smarter with the SolutionInn App


